Advanced Synthesis of 1-Substituted Aminomethyl Sinomenine Derivatives for Commercial API Manufacturing
The pharmaceutical industry continuously seeks novel scaffolds to enhance therapeutic efficacy while minimizing toxicity, particularly in the realm of anti-inflammatory and analgesic agents. Patent CN1948291A introduces a significant advancement in this field by disclosing a class of 1-substituted aminomethyl sinomenine derivatives and their preparation methods. Sinomenine, a morphinan alkaloid extracted from Sinomenium acutum, is well-known for its immunosuppressive and anti-arrhythmic properties, yet its clinical utility is often hampered by side effects such as histamine release. This patent addresses these limitations by strategically modifying the A-ring at the 1-position carbon, creating a library of derivatives with potentially improved pharmacological profiles. The disclosed compounds feature a core morphinan structure where the 1-position is functionalized with various aminomethyl groups, offering a versatile platform for drug discovery. By leveraging a robust synthetic pathway that avoids extreme conditions, this technology provides a reliable foundation for developing next-generation rheumatoid arthritis treatments and other therapeutic applications requiring high-purity intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for modifying the sinomenine skeleton often involve multi-step sequences that require harsh reaction conditions, such as strong acids or bases, which can degrade the sensitive morphinan core. Conventional approaches to introducing nitrogen-containing side chains frequently rely on过渡 metal catalysts or protecting group strategies that increase both the cost and the environmental footprint of the synthesis. Furthermore, earlier attempts at structural modification were often limited in scope, failing to provide a generalized method for introducing diverse aromatic or aliphatic amines at the C-1 position. These limitations result in lower overall yields, complex purification processes, and difficulties in scaling up for commercial production. The reliance on specialized reagents and the generation of significant chemical waste pose substantial challenges for procurement managers aiming to reduce costs in API manufacturing. Consequently, there has been a persistent need for a more efficient, atom-economical, and versatile synthetic route that can accommodate a wide range of substituents without compromising the integrity of the parent molecule.
The Novel Approach
The methodology described in CN1948291A overcomes these historical barriers through a streamlined two-step strategy centered on a Mannich-type reaction followed by nucleophilic substitution. This novel approach utilizes sinomenine hydrochloride as the starting material, reacting it directly with formaldehyde and a secondary amine under neutral conditions to install a reactive aminomethyl handle. This initial transformation is remarkably mild, operating effectively at temperatures ranging from -40°C to reflux, allowing for precise control over the reaction kinetics. The resulting intermediate, typically a 1-dimethylaminomethyl derivative, serves as a versatile precursor that can undergo further substitution with a vast array of aromatic amines or heterocycles like morpholine. This modularity enables the rapid generation of diverse chemical space around the sinomenine core, facilitating structure-activity relationship (SAR) studies. The use of common solvents such as ethanol, methanol, or dichloromethane ensures that the process is not only chemically efficient but also operationally simple, making it an ideal candidate for industrial adoption.
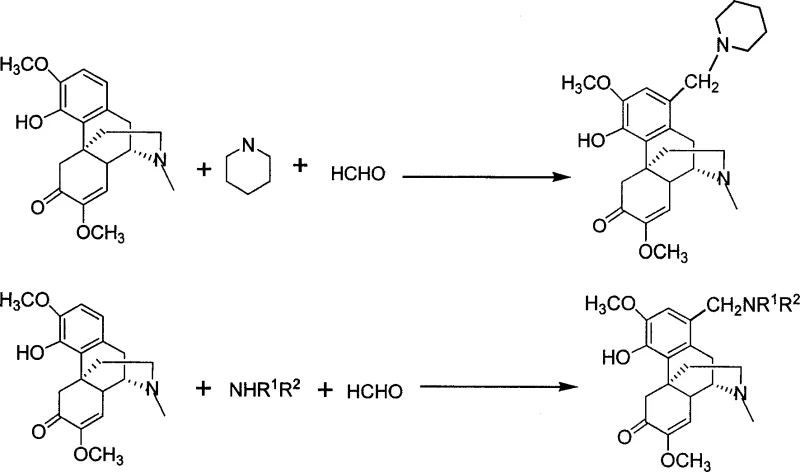
Mechanistic Insights into Mannich Reaction and Nucleophilic Substitution
The core of this synthetic innovation lies in the regioselective functionalization of the sinomenine A-ring. The first step involves the formation of an iminium ion species generated in situ from the reaction of formaldehyde and the secondary amine. This electrophilic species then attacks the electron-rich aromatic ring of sinomenine at the C-1 position, which is activated by the adjacent phenolic hydroxyl and methoxy groups. This electrophilic aromatic substitution proceeds smoothly under neutral conditions, avoiding the need for external Lewis acids. The resulting intermediate possesses a tertiary amine group at the benzylic position, which acts as an excellent leaving group in the subsequent step. In the second stage, the intermediate reacts with a primary aromatic amine or a cyclic amine like morpholine. This nucleophilic displacement occurs via an SN1 or SN2-like mechanism depending on the specific substrate and conditions, effectively swapping the dimethylamino group for the desired substituent. The patent demonstrates that this substitution is highly tolerant of various functional groups on the incoming amine, including halogens, alkyl chains, and alkoxy groups, thereby preserving the chemical diversity required for medicinal chemistry optimization.
Impurity control is inherently managed by the specificity of the reaction conditions. The use of stoichiometric amounts of formaldehyde and amine minimizes the formation of poly-substituted byproducts. Furthermore, the reaction mixture can be easily worked up using standard liquid-liquid extraction techniques, followed by purification via silica gel column chromatography or recrystallization. The patent data indicates that the final products are obtained as white solids with sharp melting points and high purity, as confirmed by NMR and mass spectrometry. The absence of heavy metal residues is a critical advantage, as it eliminates the need for expensive scavenging steps often required in transition-metal catalyzed couplings. This clean reaction profile ensures that the resulting pharmaceutical intermediates meet stringent regulatory standards for residual impurities, significantly reducing the burden on quality control laboratories during the scale-up phase.
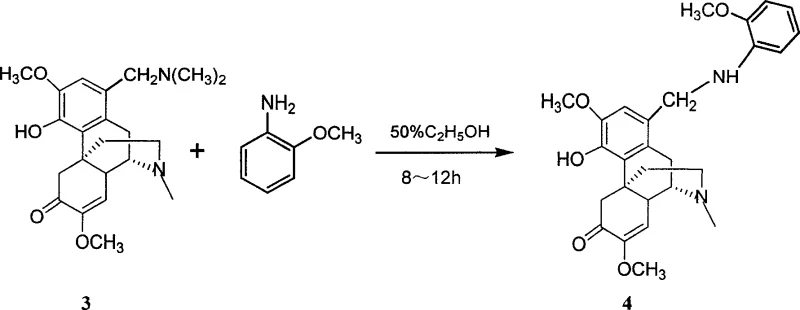
How to Synthesize 1-Substituted Aminomethyl Sinomenine Derivatives Efficiently
To implement this synthesis effectively, operators should begin by dissolving sinomenine hydrochloride in a suitable organic solvent such as ethanol or methanol. The addition of formaldehyde (typically as formalin) and the chosen secondary amine should be conducted under controlled temperature conditions, often utilizing an ice bath to manage exotherms initially before allowing the reaction to proceed at ambient or slightly elevated temperatures. The reaction progress can be monitored via TLC or HPLC to ensure complete conversion to the intermediate. Once the intermediate is isolated and purified, it is subjected to the second substitution step by heating with the target aromatic amine in an alcoholic solvent. Detailed standardized synthesis steps are provided below to guide process development teams in replicating these results with high fidelity.
- Perform a Mannich reaction by reacting sinomenine hydrochloride with formaldehyde and a secondary amine (e.g., dimethylamine or piperidine) in an organic solvent at temperatures ranging from -40°C to reflux.
- Isolate the intermediate 1-dimethylaminomethyl sinomenine derivative through extraction, drying, and purification via silica gel column chromatography or recrystallization.
- React the intermediate with a substituted aromatic amine or morpholine in an alcoholic solvent under neutral conditions to achieve the final 1-substituted aminomethyl diversine derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers compelling economic and logistical benefits. The primary advantage stems from the use of commodity chemicals; sinomenine hydrochloride is a commercially available natural product extract, while formaldehyde and simple amines are produced on a massive global scale, ensuring a stable and cost-effective supply chain. The elimination of precious metal catalysts removes a significant cost driver and mitigates supply risks associated with rare earth elements. Additionally, the mild reaction conditions translate to lower energy consumption, as the process does not require cryogenic cooling or high-pressure equipment, further driving down operational expenditures. The simplicity of the workup procedure, involving basic extraction and crystallization, reduces solvent usage and waste disposal costs, aligning with modern green chemistry initiatives.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by bypassing the need for expensive protecting groups and transition metal catalysts. The direct functionalization of the sinomenine core reduces the number of synthetic steps, which cumulatively lowers labor, material, and overhead costs. By utilizing high-yielding reactions that produce minimal byproducts, the overall material throughput is maximized, leading to substantial savings in raw material procurement. The ability to use standard laboratory glassware and reactors means that capital expenditure for new equipment is minimized, allowing existing facilities to be repurposed for this production line with little modification.
- Enhanced Supply Chain Reliability: Sourcing reliability is greatly improved due to the reliance on widely available bulk chemicals. Unlike specialized reagents that may have long lead times or single-source suppliers, the inputs for this synthesis are stocked by numerous global vendors. This redundancy ensures continuity of supply even during market fluctuations. Furthermore, the robustness of the reaction conditions means that the process is less susceptible to variations in raw material quality, reducing the risk of batch failures. This stability allows for more accurate production planning and inventory management, ensuring that downstream API manufacturing schedules are met without interruption.
- Scalability and Environmental Compliance: The synthetic route is inherently scalable, having been demonstrated effectively from milligram to gram scales in the patent examples. The absence of hazardous reagents and the use of common organic solvents simplify the permitting process for large-scale production. Waste streams are easier to treat, as they primarily consist of aqueous alcohol solutions and organic extracts that can be distilled and recycled. This environmental compatibility reduces the regulatory burden and potential fines associated with hazardous waste disposal. The process design supports a continuous improvement model, where solvent recovery and recycling can be integrated to further enhance sustainability metrics and reduce the overall carbon footprint of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these sinomenine derivatives. The answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these details is crucial for R&D teams evaluating the feasibility of incorporating these intermediates into their drug discovery pipelines.
Q: What are the primary advantages of this sinomenine derivative synthesis route?
A: The patented method utilizes mild reaction conditions (neutral pH, moderate temperatures) and readily available reagents like formaldehyde and common amines, significantly simplifying the process compared to traditional harsh modifications.
Q: Can this process be scaled for industrial API intermediate production?
A: Yes, the process avoids expensive transition metal catalysts and uses standard organic solvents (ethanol, dichloromethane), making it highly suitable for commercial scale-up with straightforward workup procedures.
Q: What is the structural diversity achievable with this method?
A: The method allows for extensive diversification at the 1-position by varying the amine component, enabling the creation of derivatives with alkyl, halogen, alkoxy, or hydroxyl substituted aromatic rings for optimized biological activity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Substituted Aminomethyl Sinomenine Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic pathways outlined in CN1948291A for developing advanced anti-inflammatory therapeutics. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to market supply is seamless. Our state-of-the-art facilities are equipped to handle the specific solvent systems and purification requirements of these morphinan alkaloids, guaranteeing stringent purity specifications for every batch. With rigorous QC labs employing advanced analytical techniques, we validate the structural integrity and purity of each derivative, providing you with the confidence needed to advance your clinical programs.
We invite you to collaborate with us to optimize this chemistry for your specific needs. Our technical team can perform a Customized Cost-Saving Analysis to identify further efficiencies in your supply chain. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your project timelines. By partnering with us, you gain access to a dedicated resource committed to accelerating your development goals while maintaining the highest standards of quality and compliance.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
