Advanced Copper-Catalyzed Synthesis of 1,2,4-Tricarbonyl Sulfoxide Ylides for Commercial Pharmaceutical Manufacturing
Advanced Copper-Catalyzed Synthesis of 1,2,4-Tricarbonyl Sulfoxide Ylides for Commercial Pharmaceutical Manufacturing
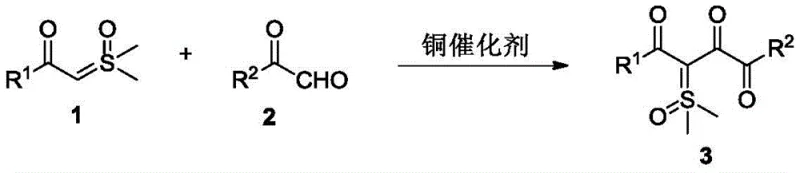
The landscape of organic synthesis for complex pharmaceutical intermediates is constantly evolving, driven by the need for more efficient, selective, and scalable methodologies. A significant breakthrough in this domain is documented in Chinese Patent CN111269156B, which discloses a robust method for synthesizing 1,2,4-tricarbonyl sulfoxide ylide compounds. These compounds are not merely academic curiosities; they serve as critical structural units in drug discovery, exhibiting remarkable biological activities including anticancer, antiviral, and neuroprotective properties. The patented technology utilizes a copper-catalyzed cross-coupling reaction between substituted formyl sulfoxide ylides and alpha-carbonyl aldehyde compounds. This approach represents a paradigm shift from traditional self-coupling methods, offering a pathway to access diverse molecular architectures under remarkably mild conditions. For R&D directors and procurement specialists alike, understanding the nuances of this synthesis is vital for securing a reliable supply chain of high-purity pharmaceutical intermediates.
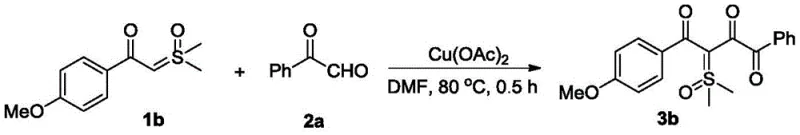
The core innovation lies in the ability to construct the 1,2,4-tricarbonyl framework with high precision and minimal environmental impact. By leveraging a divalent copper catalyst, such as copper(II) acetate, the reaction proceeds efficiently in common organic solvents like N,N-dimethylformamide (DMF). The process operates effectively at temperatures ranging from 60°C to 100°C and tolerates an air atmosphere, which drastically simplifies the operational requirements compared to inert gas protocols. This technical advancement addresses long-standing challenges in the field, particularly the difficulty in synthesizing asymmetric derivatives. As we delve deeper into the technical specifics, it becomes clear why this method is poised to become a standard for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this invention, the synthesis of 1,2,4-tricarbonyl sulfoxide ylides was predominantly achieved through self-coupling reactions of acyl sulfoxide ylides. While functional, these legacy methods suffered from severe intrinsic limitations that hindered their utility in modern drug development. The most significant drawback was the lack of structural diversity; self-coupling inherently produces symmetrical products where the substituents on either side of the carbonyl backbone are identical. This symmetry restricts the chemical space available for medicinal chemists exploring structure-activity relationships (SAR). Furthermore, conventional processes often required prolonged reaction times, sometimes extending up to 24 hours or more, which negatively impacts throughput and energy consumption in a manufacturing setting. Low product yields were another persistent issue, often necessitating extensive purification steps that erode overall process efficiency and increase the cost of goods sold (COGS).
The Novel Approach
The methodology described in Patent CN111269156B overcomes these historical bottlenecks by introducing a cross-coupling strategy. By reacting a substituted formyl sulfoxide ylide with a distinct alpha-carbonyl aldehyde, the process enables the construction of asymmetric structures where R1 is not equal to R2. This capability is transformative for generating libraries of novel compounds for biological screening. The reaction conditions are notably mild, avoiding the need for harsh acidic or basic additives that can degrade sensitive functional groups or complicate waste treatment. Experimental data from the patent demonstrates that high yields can be achieved in significantly shorter timeframes, often within 0.5 hours under optimized conditions. For instance, specific examples in the patent show isolated yields reaching up to 90%, showcasing the high efficiency and selectivity of this copper-catalyzed system. This transition from symmetrical self-coupling to asymmetric cross-coupling marks a substantial upgrade in synthetic capability.
Mechanistic Insights into Copper-Catalyzed Cross-Coupling
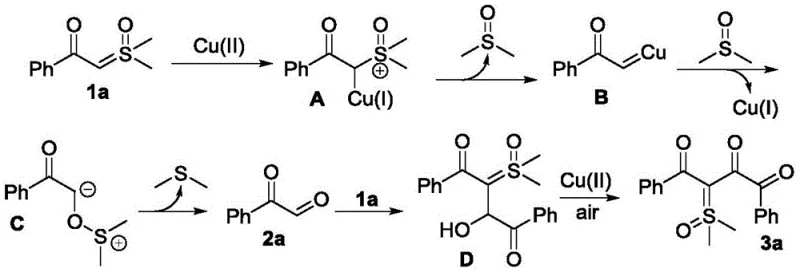
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to adapt this chemistry for specific API intermediates. The proposed mechanism involves the activation of the sulfoxide ylide by the copper(II) catalyst. Initially, the copper species coordinates with the ylide, facilitating the formation of a reactive copper-carbenoid or metal-ylide intermediate. This activated species then undergoes nucleophilic attack or insertion into the alpha-carbonyl aldehyde. The presence of the copper catalyst is pivotal, as it lowers the activation energy barrier for the C-C bond formation. Crucially, the reaction environment allows for the smooth progression of the catalytic cycle without the accumulation of stable off-cycle species that typically plague transition metal catalysis. The compatibility of the catalyst with various functional groups—such as halogens, alkoxy groups, and heterocycles—suggests a robust tolerance that is essential for late-stage functionalization in drug synthesis.
Furthermore, the mechanism accounts for the observed selectivity and impurity profile. The absence of strong acids or bases prevents unwanted side reactions such as aldol condensations or hydrolysis of the ylide moiety, which are common pitfalls in carbonyl chemistry. The oxidative nature of the copper catalyst, potentially assisted by air or oxygen, ensures the regeneration of the active catalytic species, sustaining the reaction turnover. This mechanistic clarity provides confidence in the reproducibility of the process. For quality control purposes, the clean reaction profile means that the crude product often contains fewer byproducts, simplifying the downstream purification process. This level of mechanistic control is what distinguishes a laboratory curiosity from a viable industrial process capable of delivering high-purity pharmaceutical intermediates consistently.
How to Synthesize 1,2,4-Tricarbonyl Sulfoxide Ylide Efficiently
Implementing this synthesis in a practical setting requires adherence to the optimized parameters identified in the patent data. The general procedure involves combining the sulfoxide ylide and the alpha-carbonyl aldehyde in a polar aprotic solvent, with DMF being the preferred choice due to its ability to solubilize both reactants and stabilize the transition states. The addition of the copper catalyst, typically Cu(OAc)2, initiates the reaction upon heating. It is important to note that while the reaction can proceed under air, controlling the atmosphere can further optimize yields for sensitive substrates. The detailed standardized synthesis steps, including precise stoichiometric ratios and workup procedures, are outlined below to ensure successful replication and scale-up.
- Combine substituted formyl sulfoxide ylide and alpha-carbonyl aldehyde compound in an organic solvent such as DMF with a divalent copper catalyst like Cu(OAc)2.
- Heat the reaction mixture to a temperature between 60°C and 100°C under an air or oxygen atmosphere for approximately 0.5 to 24 hours depending on substrate reactivity.
- Quench the reaction with saturated brine, extract with ethyl acetate, dry over anhydrous sodium sulfate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical elegance. The primary advantage lies in the significant cost reduction in pharmaceutical intermediate manufacturing. By eliminating the need for expensive ligands or exotic catalysts and relying on commodity copper salts, the raw material costs are kept low. Additionally, the shortened reaction times directly translate to increased reactor occupancy rates, allowing facilities to produce more batches within the same timeframe. The simplicity of the workup—often requiring only extraction and crystallization—reduces the consumption of solvents and silica gel, further driving down operational expenses. These factors combine to create a highly economical process that enhances the competitiveness of the final drug product.
Supply chain reliability is another critical area where this technology excels. The starting materials, substituted formyl sulfoxide ylides and alpha-carbonyl aldehydes, are generally accessible and can be sourced from multiple suppliers, mitigating the risk of single-source dependency. The robustness of the reaction conditions means that the process is less susceptible to minor fluctuations in temperature or atmospheric composition, ensuring consistent batch-to-batch quality. This stability is paramount for maintaining continuous production schedules and meeting strict delivery deadlines. Moreover, the scalability of the reaction has been demonstrated across a wide range of substrates, from simple phenyl groups to complex heterocycles, indicating that the process can be adapted for various commercial scale-up of complex pharmaceutical intermediates without extensive re-optimization.
Environmental compliance and waste management are increasingly important metrics for modern chemical enterprises. This synthesis method aligns well with green chemistry principles by avoiding the use of corrosive acids and bases, which simplifies wastewater treatment and reduces the hazard profile of the manufacturing site. The high atom economy and selectivity minimize the generation of hazardous byproducts, lowering the burden on waste disposal systems. For companies aiming to reduce their carbon footprint and adhere to stringent environmental regulations, adopting this cleaner synthetic route offers a strategic advantage. It facilitates the production of high-purity pharmaceutical intermediates while maintaining a sustainable operational model that satisfies both regulatory bodies and corporate social responsibility goals.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a reliable foundation for decision-making. Understanding these details helps stakeholders assess the feasibility of integrating this chemistry into their existing manufacturing workflows.
Q: What are the optimal reaction conditions for this cross-coupling synthesis?
A: The patent specifies that using N,N-dimethylformamide (DMF) as the solvent with Cu(OAc)2 as the catalyst at temperatures between 60°C and 100°C yields the best results, often achieving high purity without significant impurities.
Q: Does this method support asymmetric structure synthesis?
A: Yes, unlike prior art which was limited to symmetrical structures, this cross-coupling method effectively synthesizes asymmetric 1,2,4-tricarbonyl sulfoxide ylides where R1 is not equal to R2, significantly expanding structural diversity.
Q: Is the use of strong acids or bases required for this transformation?
A: No, a key advantage of this protocol is that the synthesis proceeds smoothly without the need for additional alkali or acid additives, simplifying the workup process and reducing corrosive waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,4-Tricarbonyl Sulfoxide Ylide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug development timelines. Our team of expert chemists has thoroughly analyzed the potential of the copper-catalyzed cross-coupling route described in Patent CN111269156B. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 1,2,4-tricarbonyl sulfoxide ylide meets the highest standards required for pharmaceutical applications. We are committed to delivering consistency and quality in every shipment.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis can be tailored to your specific project needs. Whether you require a Customized Cost-Saving Analysis for your current supply chain or need to evaluate the feasibility of new asymmetric derivatives, we are here to support your goals. Please contact us to request specific COA data and route feasibility assessments. By collaborating with us, you gain access to a reliable partner dedicated to optimizing your supply of high-value pharmaceutical intermediates through cutting-edge chemical technology.
