Optimizing Ivabradine Production: A Technical Breakthrough for Commercial Scale-Up
Optimizing Ivabradine Production: A Technical Breakthrough for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic pathways for critical cardiovascular medications, and the synthesis of Ivabradine represents a significant area of innovation. Patent CN101723897A introduces a refined methodology for producing Ivabradine and its addition salts, specifically addressing the inefficiencies found in earlier generations of synthetic routes. This technology focuses on the construction of the core benzazepine scaffold through a strategic N-alkylation sequence, bypassing the problematic hydrogenation and reduction steps that have historically plagued manufacturing efforts. By leveraging stable intermediates and optimizing reaction sequences, this approach offers a compelling solution for achieving high-purity Ivabradine intermediates with improved operational efficiency. The structural integrity of the final molecule, as depicted below, relies on the precise coupling of the benzazepinone core with the benzocyclobutane moiety, a challenge this patent effectively resolves.
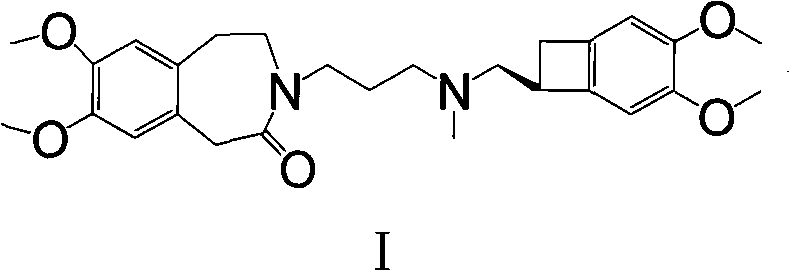
For R&D directors and process chemists, the significance of this patent lies in its ability to streamline the production workflow while maintaining stringent quality standards. The method described involves synthesizing an N-(3-substituted propyl)-7,8-dimethoxy-1,3,4,5-tetrahydro-2H-3-benzazepin-2-one intermediate, which serves as a pivotal junction in the synthesis. This intermediate is subsequently reacted with (1S)-4,5-dimethoxy-1-(methylaminomethyl)benzocyclobutane to generate the final active pharmaceutical ingredient. The technical elegance of this route is not merely in the bond formation but in the selection of reagents and conditions that minimize side reactions, ensuring that the impurity profile remains within acceptable limits for regulatory submission. This level of control is essential for any reliable pharmaceutical intermediate supplier aiming to support global drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical approaches to Ivabradine synthesis have been fraught with technical and economic hurdles that hinder efficient commercialization. For instance, the route disclosed in US5296482 involves a final hydrogenation step of a double bond that suffers from a disappointingly low yield of approximately 40%. Furthermore, the preparation of the key amine precursor, (1S)-4,5-dimethoxy-1-(methylaminomethyl)benzocyclobutane, is notoriously difficult and costly, creating a bottleneck in the supply chain. Another prior art method, detailed in patents like CN1683341, relies on expensive 2-haloacetals and necessitates a reduction step using reducing agents, which not only inflates the cost of goods but also introduces safety and waste disposal concerns incompatible with green chemistry principles.
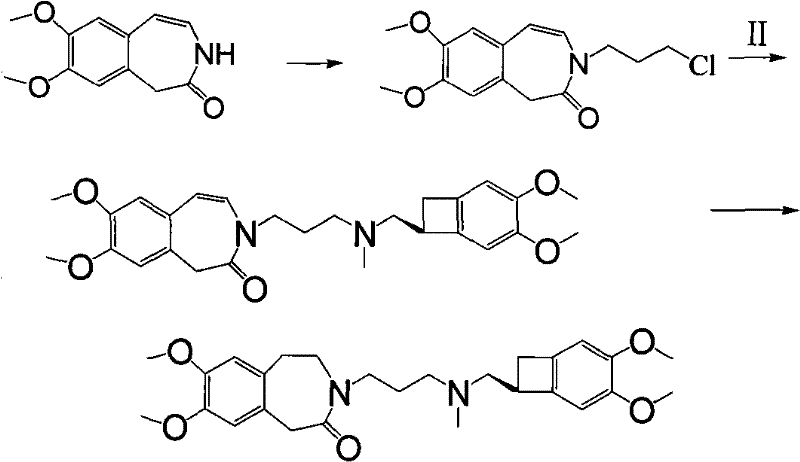
Additionally, other existing methodologies, such as the one found in CN101284813, attempt to mitigate some costs but introduce new complexities. These routes often involve the formation of N-chloropropyl intermediates followed by reduction, a sequence that is chemically unreasonable and prone to side reactions involving the chloroalkane group. Moreover, the free base form of the benzocyclobutane amine used in these older methods is unstable and cannot be stored for long periods, forcing manufacturers to rely on less convenient salt forms or immediate usage, which complicates inventory management. These cumulative inefficiencies result in higher production costs and lower overall throughput, making cost reduction in API manufacturing a critical priority for adopting newer technologies.
The Novel Approach
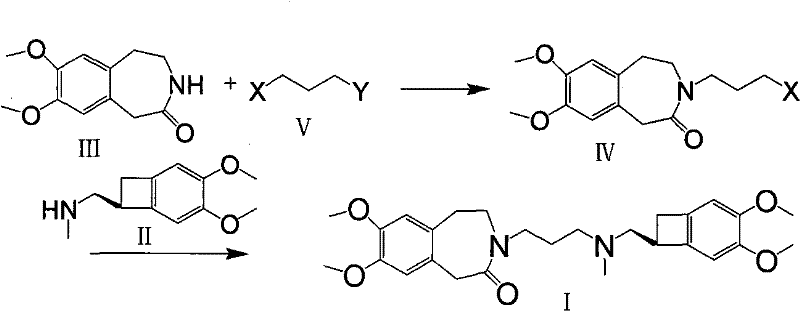
In stark contrast to these legacy methods, the novel approach outlined in CN101723897A presents a streamlined and chemically logical pathway that maximizes yield and minimizes waste. The core innovation lies in the direct alkylation of the benzazepinone nitrogen with a dihalopropane derivative, such as 1-bromo-3-chloropropane, under mild basic conditions. This generates a stable N-substituted intermediate that can be isolated and purified with ease, providing a solid foundation for the subsequent coupling reaction. The reaction sequence is designed to avoid the unstable free base issues by allowing the use of the amine component in its salt form, specifically the hydrochloride salt, which is far more stable and commercially available. This shift in strategy fundamentally alters the economic landscape of the synthesis, offering substantial cost savings through simplified processing and higher material throughput.
The versatility of this new route is further enhanced by the option to perform an in-situ halogen exchange, converting a chloro-group to a more reactive iodo-group using sodium iodide. This Finkelstein-type modification significantly accelerates the final coupling step, ensuring high conversion rates without the need for extreme temperatures or pressures. The result is a process that is not only chemically superior but also operationally simpler, requiring standard equipment and common solvents like acetone or ethyl acetate. For procurement managers, this translates to a more predictable supply chain with reduced dependency on exotic reagents. The ability to produce high-purity Ivabradine intermediates with fewer purification steps directly impacts the bottom line, making this technology a preferred choice for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into N-Alkylation and Halogen Exchange
The mechanistic foundation of this synthesis rests on the nucleophilic character of the lactam nitrogen within the 7,8-dimethoxy-1,3,4,5-tetrahydro-2H-3-benzazepin-2-one ring system. In the presence of a strong base such as potassium tert-butoxide in an aprotic polar solvent like DMSO, the acidic proton on the nitrogen is abstracted to form a resonance-stabilized anion. This anionic species acts as a potent nucleophile, attacking the terminal carbon of the dihalopropane chain. The choice of solvent is critical here; DMSO facilitates the dissolution of both the organic substrate and the inorganic base, creating a homogeneous reaction medium that promotes efficient collision frequency. The use of a dihalopropane with different halogens, such as bromine and chlorine, allows for selective reactivity, where the more labile bromine is displaced first, leaving the chlorine atom intact for the subsequent coupling stage. This selectivity is key to preventing polymerization or bis-alkylation side products.
Following the formation of the N-(3-chloropropyl) intermediate, the process employs a strategic halogen exchange to enhance reactivity for the final bond formation. Chlorine is a moderate leaving group, but in the context of coupling with a sterically hindered amine like the benzocyclobutane derivative, its reactivity might be insufficient for high yields. By introducing sodium iodide into the reaction mixture, the chlorine atom is displaced by iodide via an SN2 mechanism, generating the corresponding N-(3-iodopropyl) intermediate in situ. Iodine is a superior leaving group due to its larger atomic radius and weaker bond strength with carbon, which drastically lowers the activation energy for the nucleophilic attack by the amine. This mechanistic nuance ensures that the final coupling proceeds smoothly even at moderate temperatures, preserving the stereochemical integrity of the chiral benzocyclobutane center and minimizing the formation of degradation byproducts.
How to Synthesize Ivabradine Efficiently
The implementation of this synthesis route requires careful attention to reaction parameters to maximize the benefits of the patented method. The process begins with the activation of the benzazepinone core, followed by the controlled addition of the alkylating agent to ensure mono-substitution. Once the intermediate is secured, the focus shifts to the coupling reaction, where the choice of base and solvent plays a pivotal role in driving the equilibrium towards the product. Detailed procedural guidelines are essential for reproducibility, particularly regarding the stoichiometry of the halogen exchange reagents and the temperature profiles for reflux conditions. The following guide outlines the standardized steps derived from the patent examples to assist technical teams in replicating this high-efficiency pathway.
- Alkylate 7,8-dimethoxy-1,3,4,5-tetrahydro-2H-3-benzazepin-2-one with a dihalopropane in DMSO using potassium tert-butoxide to form the N-substituted intermediate.
- React the resulting intermediate with (1S)-4,5-dimethoxy-1-(methylaminomethyl)benzocyclobutane (free base or salt) in a solvent like acetone, optionally using sodium iodide to facilitate halogen exchange.
- Purify the crude product via column chromatography or recrystallization and convert to the desired pharmaceutically acceptable salt, such as the hydrochloride.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis methodology offers transformative benefits for supply chain stability and cost management. Traditional routes often suffer from volatile raw material costs due to the reliance on specialized reagents like 2-haloacetals or unstable free bases that require cold chain logistics. By shifting to a process that utilizes stable, commodity-grade chemicals and robust intermediates, manufacturers can significantly de-risk their supply chains. The ability to store intermediates without degradation allows for batch planning flexibility, reducing the pressure on just-in-time delivery models. Furthermore, the simplified workup procedures, which often involve straightforward filtration and evaporation rather than complex chromatographic separations, reduce the consumption of silica gel and solvents, leading to a leaner and more cost-effective operation.
- Cost Reduction in Manufacturing: The elimination of low-yield hydrogenation steps and expensive reducing agents directly lowers the variable cost per kilogram of the final product. By avoiding the need for high-pressure hydrogenation equipment and the associated safety protocols, capital expenditure is also optimized. The use of catalytic amounts of sodium iodide to drive the reaction to completion ensures that raw material utilization is maximized, reducing waste disposal costs. Additionally, the higher purity of the crude product means less resource intensity is required for final purification, contributing to substantial cost savings in the overall manufacturing budget.
- Enhanced Supply Chain Reliability: The stability of the key intermediates allows for decoupling the synthesis stages, meaning the N-substituted benzazepinone can be produced and stockpiled independently of the final coupling step. This modularity enhances supply continuity, as disruptions in the supply of the benzocyclobutane amine do not halt the entire production line. Moreover, the preference for using the hydrochloride salt of the amine component aligns with standard market availability, removing the need for custom synthesis of unstable free bases. This alignment with existing market standards ensures a reliable pharmaceutical intermediate supplier can maintain consistent delivery schedules.
- Scalability and Environmental Compliance: The reaction conditions are inherently scalable, utilizing solvents like acetone and ethyl acetate which are widely accepted in large-scale pharmaceutical manufacturing. The absence of heavy metal catalysts or hazardous reducing agents simplifies the environmental compliance landscape, reducing the burden on wastewater treatment facilities. The process generates fewer byproducts, which translates to a lower E-factor (environmental factor), a key metric for sustainable chemistry. This environmental friendliness not only meets regulatory standards but also appeals to stakeholders prioritizing green manufacturing practices, facilitating smoother regulatory approvals for commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis route is vital for stakeholders evaluating its potential for integration into their production portfolios. The following questions address common inquiries regarding the practical application, scalability, and quality implications of the patented method. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What are the primary advantages of this synthesis route over prior art methods?
A: This method eliminates the need for difficult hydrogenation steps with low yields (often around 40% in older routes) and avoids expensive 2-haloacetals. It utilizes stable intermediates that are easier to purify, leading to higher overall purity and yield suitable for industrial production.
Q: How does the process handle the instability of the benzocyclobutane amine component?
A: The process allows for the use of the benzocyclobutane amine in its salt form, specifically the hydrochloride salt. This significantly enhances stability during storage and handling compared to the free base, which is prone to degradation, thereby simplifying logistics and raw material management.
Q: Is this synthesis route scalable for commercial manufacturing?
A: Yes, the reaction conditions are mild (temperatures between 30°C and 120°C) and utilize common industrial solvents like DMSO and acetone. The avoidance of sensitive reduction steps and the use of stable intermediates make the process highly robust and adaptable for large-scale commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ivabradine Supplier
The technical superiority of the synthesis route described in CN101723897A positions it as a cornerstone for modern Ivabradine production, yet realizing its full potential requires a partner with deep process engineering expertise. NINGBO INNO PHARMCHEM stands ready to leverage this technology, bringing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that ensure every batch meets the exacting standards required for cardiovascular therapeutics. We understand that consistency is key in the pharmaceutical supply chain, and our infrastructure is designed to deliver high-purity Ivabradine intermediates with unwavering reliability.
We invite global partners to engage with our technical procurement team to explore how this optimized synthesis route can benefit your specific project needs. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that quantifies the economic advantages of switching to this efficient methodology. We encourage you to request specific COA data and route feasibility assessments to validate the performance metrics in your own context. Together, we can drive down costs and accelerate the availability of life-saving medications, ensuring a healthier future through chemical innovation.
