Scaling Advanced Optoelectronic Materials: A Technical Breakdown of Novel Boron Compound Synthesis
The landscape of advanced optoelectronic materials is undergoing a significant transformation driven by the demand for high-performance organic luminescent systems. Patent CN108912152B introduces a groundbreaking beta-dicarbonyl fluoroboron compound, specifically 2,2-difluoro-(N-ethylcarbazole-3)-1-(6-bromonaphthalene-2)-2H-1,3,2-dioxaborane, which represents a major leap forward in the design of pure organic room temperature phosphorescence systems. This novel molecule is not merely a theoretical construct but a tangible chemical entity that bridges the gap between small-molecule fluorescence and functional nanomaterial assembly. By leveraging the unique photophysical properties of beta-dicarbonyl fluoroboron cores, this technology enables the creation of organic nanospheres and nanorods that exhibit exceptional stability and sensitivity in biological and electronic environments. The strategic integration of carbazole and naphthalene moieties within the fluoroboron framework enhances the conjugation system, resulting in superior light absorption and emission characteristics that are critical for next-generation bioimaging and display technologies. 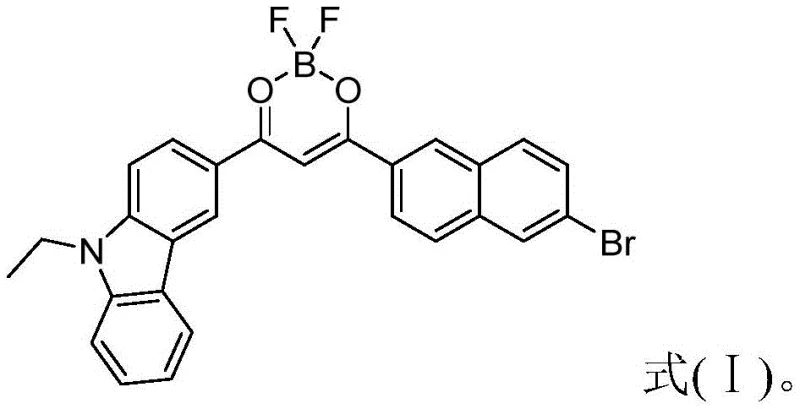
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of organic luminescent materials has been plagued by significant challenges related to aggregation-caused quenching and the reliance on expensive heavy metal complexes to achieve phosphorescence. Traditional fluorophores often suffer from rapid decay of excited states in the solid state or aggregated forms, severely limiting their utility in high-concentration applications such as solid-state lighting or dense biological labeling. Furthermore, conventional methods for inducing room temperature phosphorescence typically require cryogenic conditions or the incorporation of toxic transition metals like iridium or platinum, which introduce substantial regulatory hurdles and environmental liabilities for commercial manufacturers. The synthesis of these traditional materials often involves multi-step pathways with low atom economy, generating significant chemical waste and driving up the cost of goods sold. Additionally, controlling the morphology of these materials at the nanoscale has been notoriously difficult, leading to batch-to-batch inconsistencies that compromise the reliability of supply chains for high-tech industries requiring precise optical specifications.
The Novel Approach
The methodology outlined in the patent data presents a paradigm shift by utilizing a metal-free organic system that achieves room temperature phosphorescence through supramolecular assembly rather than heavy metal coordination. This novel approach capitalizes on the intrinsic ability of the beta-dicarbonyl fluoroboron compound to self-assemble into well-defined nanostructures via weak intermolecular interactions when transferred from organic solvents to aqueous environments. By manipulating the solvent conditions, specifically the presence or absence of surfactants, manufacturers can precisely tune the morphology between nanospheres and nanorods, offering a level of control that is absent in conventional precipitation methods. This solvent-driven assembly process eliminates the need for complex templating agents or harsh reaction conditions, thereby simplifying the production workflow and reducing the overall energy consumption of the manufacturing process. The resulting materials exhibit robust photostability and large two-photon absorption cross-sections, making them ideally suited for deep-tissue imaging applications where traditional dyes fail to penetrate effectively or degrade under prolonged irradiation.
Mechanistic Insights into Beta-Dicarbonyl Fluoroboron Assembly
The core mechanism driving the functionality of this material lies in the electronic structure of the beta-dicarbonyl fluoroboron unit, which acts as a rigid planarizer for the conjugated system. The coordination of the boron atom with the oxygen atoms of the diketone ligand creates a highly rigidified structure that restricts intramolecular rotation, a primary non-radiative decay pathway in flexible organic molecules. This rigidification is crucial for enhancing the quantum yield of fluorescence and enabling the intersystem crossing required for phosphorescence. The presence of the bromine atom on the naphthalene ring further facilitates spin-orbit coupling, promoting the transition from singlet to triplet excited states without the need for external heavy atoms. When these molecules are dispersed in aqueous media, the hydrophobic effect drives them to aggregate, but the specific steric hindrance provided by the N-ethylcarbazole group prevents无序 precipitation, instead guiding the formation of ordered crystalline or semi-crystalline nanostructures. This precise molecular packing is what unlocks the room temperature phosphorescence, as the rigid environment suppresses vibrational relaxation and protects the triplet excitons from quenching by molecular oxygen.
Impurity control in this synthesis is inherently managed through the selectivity of the Claisen condensation and the subsequent boron complexation steps. The use of sodium hydride as a strong base ensures the complete deprotonation of the acetyl intermediate, driving the condensation reaction towards the desired 1,3-dicarbonyl product with minimal formation of mono-substituted byproducts. Subsequent purification via column chromatography allows for the removal of unreacted starting materials and inorganic salts, ensuring that the final fluoroboron compound meets the stringent purity requirements necessary for electronic applications. The self-assembly process itself acts as a secondary purification step, as only molecules with the correct structural integrity and amphiphilic balance will successfully incorporate into the growing nanostructures. Defective molecules or impurities that disrupt the weak interaction network are excluded from the assembly, resulting in nanomaterials with highly uniform optical properties. This dual-layer quality control, combining chemical synthesis selectivity with physical self-purification during assembly, provides a robust mechanism for maintaining high product consistency across large production batches.
How to Synthesize 2,2-Difluoro-(N-ethylcarbazole-3)-1-(6-bromonaphthalene-2)-2H-1,3,2-dioxaborane Efficiently
The synthesis of this high-value optoelectronic intermediate follows a logical three-step sequence that balances yield with operational simplicity, making it highly attractive for industrial scale-up. The process begins with the functionalization of the carbazole core, followed by the extension of the conjugated system through carbon-carbon bond formation, and concludes with the critical boron complexation that locks the molecular geometry. Each step utilizes reagents and solvents that are readily available in the global chemical supply chain, mitigating the risk of procurement bottlenecks. The detailed standardized synthesis steps see the guide below for specific reaction parameters and workup procedures that ensure maximum recovery and purity. 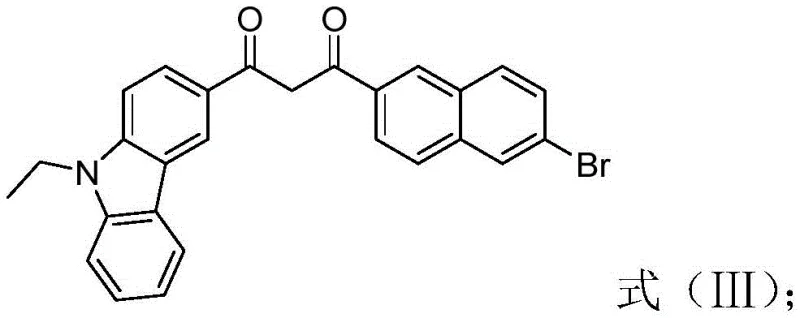
- Perform Friedel-Crafts acylation of N-ethylcarbazole with acetyl chloride using aluminum trichloride catalyst to form N-ethyl-3-acetylcarbazole.
- Execute Claisen condensation between the acetylcarbazole intermediate and methyl 6-bromo-2-naphthoate using sodium hydride base to generate the diketone precursor.
- Complex the diketone precursor with boron trifluoride etherate in the presence of triethylamine to finalize the beta-dicarbonyl fluoroboron structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers substantial strategic advantages regarding cost stability and operational flexibility. The elimination of precious metal catalysts removes a significant source of price volatility from the bill of materials, as the cost of palladium or iridium can fluctuate wildly based on geopolitical factors and mining output. By relying on abundant main-group elements like boron and aluminum, manufacturers can secure long-term pricing agreements with raw material suppliers, ensuring predictable cost structures for multi-year projects. Furthermore, the synthetic pathway avoids the use of highly toxic reagents that require specialized waste treatment facilities, thereby reducing the overhead costs associated with environmental compliance and hazardous material handling. This streamlined chemical profile allows for production in a wider range of facilities, increasing the potential supplier base and enhancing supply chain resilience against regional disruptions.
- Cost Reduction in Manufacturing: The synthetic route achieves cost optimization primarily through the simplification of the purification process and the avoidance of expensive transition metal scavengers. Traditional phosphorescent materials often require rigorous post-reaction treatment to remove trace metals that could poison downstream electronic devices or cause toxicity in biological applications. This metal-free fluoroboron system inherently bypasses the need for such costly purification steps, significantly reducing the consumption of specialized resins and solvents. Additionally, the high atom economy of the Claisen condensation step ensures that a larger proportion of the starting mass is converted into the final product, minimizing waste disposal fees. The overall reduction in processing time and material consumption translates directly into a lower cost per kilogram, providing a competitive edge in price-sensitive markets like consumer electronics and disposable diagnostic devices.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as acetyl chloride, sodium hydride, and boron trifluoride etherate ensures that the supply chain is not dependent on niche vendors with limited production capacity. These reagents are produced by multiple major chemical manufacturers globally, creating a robust network of potential sources that can be activated in case of supply interruptions. The synthesis does not require custom-synthesized ligands or proprietary catalysts that might be subject to intellectual property restrictions or single-source dependencies. This openness in the supply chain allows procurement teams to negotiate better terms and maintain safety stock levels without incurring prohibitive costs. The stability of the intermediate compounds also allows for semi-finished goods to be stored and transported safely, providing flexibility in production scheduling and inventory management.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the use of standard unit operations such as liquid-liquid extraction and distillation, which are well-understood by chemical engineers. The reaction conditions, primarily involving room temperature or moderate reflux, do not require high-pressure reactors or cryogenic cooling systems, reducing the capital expenditure required for plant expansion. From an environmental perspective, the absence of heavy metals simplifies the wastewater treatment process, allowing for easier compliance with increasingly strict environmental regulations in major manufacturing hubs. The organic solvents used, such as dichloromethane and tetrahydrofuran, can be efficiently recovered and recycled through standard distillation columns, further minimizing the environmental footprint and reducing the purchase volume of fresh solvents. This alignment with green chemistry principles enhances the corporate sustainability profile of the end product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this beta-dicarbonyl fluoroboron technology in industrial settings. These answers are derived directly from the patent specifications and practical considerations for scaling organic electronic materials. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this material into existing product lines and for procurement officers assessing the long-term viability of the supply source. 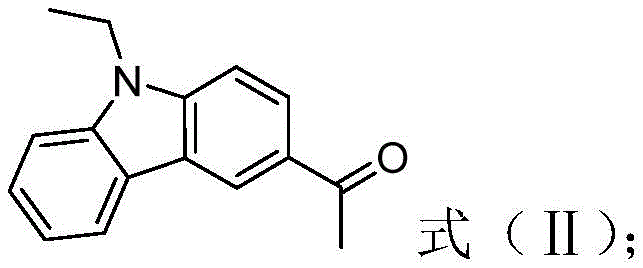
Q: What are the primary photophysical advantages of this beta-dicarbonyl fluoroboron compound?
A: The compound exhibits large molar absorption coefficients, high luminescence quantum yields, and significant two-photon absorption cross-sections. Crucially, it demonstrates pure organic room temperature phosphorescence when assembled into nanostructures, offering superior stability and sensitivity compared to traditional solution-state fluorophores.
Q: How does the morphology of the nanomaterial affect its application in bioimaging?
A: The compound self-assembles into distinct nanospheres and nanorods via weak interactions in aqueous solutions. Nanospheres show dual yellow fluorescence-phosphorescence emission suitable for deep-tissue imaging, while nanorods offer specific morphological advantages for targeted cellular uptake and enhanced signal-to-noise ratios in complex biological environments.
Q: Is this synthesis route scalable for commercial optoelectronic material production?
A: Yes, the synthetic pathway utilizes standard organic transformations like Friedel-Crafts acylation and Claisen condensation which are well-established in industrial settings. The use of common solvents like dichloromethane and tetrahydrofuran, along with accessible reagents, facilitates straightforward scale-up from laboratory grams to multi-ton commercial production without requiring exotic catalysts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Optoelectronic Material Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating innovative patent chemistry into reliable commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial application is seamless and efficient. We understand that high-purity optoelectronic materials require stringent purity specifications and rigorous QC labs to guarantee performance consistency in sensitive electronic and biological applications. Our state-of-the-art facilities are equipped to handle the specific solvent systems and reaction conditions required for beta-dicarbonyl fluoroboron synthesis, maintaining the highest standards of quality and safety throughout the manufacturing process.
We invite you to collaborate with us to optimize your supply chain for advanced luminescent materials. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage potential partners to contact us to request specific COA data and route feasibility assessments that demonstrate our capability to deliver this complex intermediate at a competitive price point. By leveraging our expertise in process chemistry and supply chain management, we can help you secure a stable source of high-performance materials that drive innovation in your end products.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
