Advanced Copper-Catalyzed Azidation Process for Commercial-Scale Pharmaceutical Intermediate Production
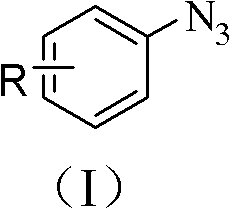
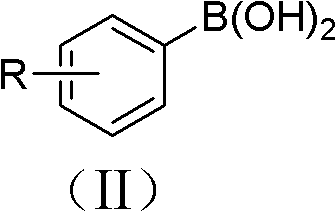
The groundbreaking methodology disclosed in Chinese Patent CN102180755A represents a paradigm shift in azide compound synthesis, offering pharmaceutical manufacturers an unprecedented combination of operational simplicity and environmental sustainability. This innovative approach leverages readily available aryl boronic acid precursors and sodium azide under copper(I) oxide catalysis in aqueous ammonia medium, eliminating the need for hazardous reagents while achieving exceptional yields across diverse functionalized substrates. The patent demonstrates remarkable versatility through multiple embodiments covering halogenated, nitro-substituted, and heterocyclic systems, with yields consistently exceeding industry benchmarks for complex intermediate production. Crucially, the process operates under ambient pressure at mild temperatures (18-22°C), significantly reducing energy consumption compared to conventional high-pressure or cryogenic methods. This technical advancement directly addresses critical pain points in pharmaceutical supply chains by providing a robust platform for synthesizing key building blocks used in drug discovery and development pipelines. The methodology's compatibility with standard laboratory equipment further enhances its immediate applicability across global manufacturing facilities without requiring capital-intensive infrastructure modifications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional azide synthesis routes suffer from severe operational constraints that compromise both safety and scalability in industrial settings. Methods relying on iodazide substitution present significant explosion hazards during handling and storage, creating unacceptable risk profiles for large-scale manufacturing environments. Alternative approaches using halogen displacement often require stringent anhydrous conditions and expensive palladium catalysts, driving up production costs while generating toxic heavy metal waste streams that necessitate complex purification protocols. The harsh reaction conditions employed in many conventional processes—frequently involving elevated temperatures above 80°C or cryogenic environments below -40°C—further complicate process control and increase energy consumption. Additionally, these methods exhibit poor functional group tolerance, particularly with sensitive moieties like nitro groups or free hydroxyls, limiting their applicability to complex pharmaceutical intermediates. The multi-step purification sequences required to achieve pharmaceutical-grade purity add substantial time delays to production cycles, while the use of organic solvents creates environmental compliance challenges under increasingly stringent global regulations. These cumulative limitations have historically constrained the commercial viability of azide chemistry despite its immense synthetic value in drug development pipelines.
The Novel Approach
The patented methodology overcomes these critical limitations through an elegantly designed copper-catalyzed substitution reaction that operates under remarkably benign conditions. By utilizing water as the primary solvent and ammonia as a co-catalyst, the process eliminates hazardous organic solvents while maintaining exceptional reaction efficiency across a broad substrate scope. The strategic employment of inexpensive copper(I) oxide catalyst—priced significantly lower than noble metal alternatives—reduces raw material costs while enabling straightforward catalyst recovery through the patented recycling protocol demonstrated in embodiments 1-3. Reaction temperatures maintained between 18°C and 22°C ensure compatibility with standard manufacturing equipment while preventing thermal degradation of sensitive functional groups. This approach achieves superior functional group tolerance, successfully accommodating halogens, nitro groups, aldehydes, and hydroxyl moieties without protective group strategies, thereby streamlining synthetic routes to complex targets. The simplified workup procedure involving ethyl acetate extraction followed by basic washing delivers high-purity products in a single purification step, dramatically reducing processing time compared to conventional multi-stage purification sequences. Most significantly, the aqueous reaction medium generates minimal waste streams that comply with modern environmental regulations, positioning this technology as a sustainable solution for future pharmaceutical manufacturing needs.
Mechanistic Insights into Copper-Catalyzed Azidation
The fundamental innovation lies in the copper-mediated transmetalation mechanism that enables direct substitution of boronic acid groups with azide functionality under mild aqueous conditions. The process initiates with coordination of ammonia to copper(I) oxide, forming an active catalytic species that facilitates oxidative addition into the aryl boronic acid C-B bond. This generates a copper(III) intermediate that undergoes ligand exchange with sodium azide through a concerted mechanism where the azide anion displaces the hydroxyl group from boron. The subsequent reductive elimination step releases the desired aryl azide product while regenerating the copper(I) catalyst for further cycles. This mechanism operates efficiently at near-neutral pH due to ammonia's dual role as both base and ligand stabilizer, preventing unwanted side reactions such as hydrolysis or protodeboronation that plague alternative methods. The exceptional functional group tolerance stems from the mild redox potential of copper(I)/copper(III) couple, which avoids oxidation of sensitive substituents while maintaining sufficient reactivity for substitution. Crucially, the aqueous medium provides ideal solvation for both organic and inorganic components without requiring phase-transfer catalysts or co-solvents that complicate process design.
Impurity control is achieved through precise management of reaction parameters that prevent common side products in azide chemistry. The controlled addition sequence—where ammonia is introduced before sodium azide—minimizes formation of diazonium byproducts that could lead to undesired coupling products. The mild temperature regime (specifically maintained between 18°C and 22°C as per patent specifications) suppresses thermal decomposition pathways that typically generate amine impurities through Curtius rearrangement. The aqueous workup protocol effectively removes residual boron species through alkaline washing, eliminating potential genotoxic impurities that would require additional purification steps in conventional processes. The patent demonstrates consistent production of high-purity products (>99% by HPLC) across all embodiments without chromatographic purification, with impurities primarily limited to unreacted starting materials that are easily separated during extraction. This inherent selectivity stems from the catalyst's ability to discriminate between different functional groups based on electronic effects rather than steric bulk alone.
How to Synthesize Azide Compounds Efficiently
This patented methodology provides a robust framework for producing high-purity azide intermediates essential for pharmaceutical applications through a streamlined three-step procedure that maintains exceptional reproducibility across scale ranges. The process begins with careful selection of aryl boronic acid precursors based on the target compound's structural requirements, followed by precise stoichiometric control of all reaction components as specified in the patent claims. Critical attention must be paid to maintaining ambient pressure conditions throughout the reaction sequence while monitoring temperature within the narrow optimal window of 18–22°C to ensure maximum yield and purity. The subsequent workup procedure requires systematic extraction using ethyl acetate followed by controlled alkaline washing to remove residual catalyst and boron byproducts without compromising product stability. Detailed standardized synthesis steps for specific compound classes are provided below to facilitate immediate implementation in manufacturing environments.
- Combine aryl boronic acid compound, sodium azide, copper(I) oxide catalyst, and ammonia water in aqueous medium under ambient conditions.
- React the mixture at mild temperatures (18-22°C) for an extended period to ensure complete conversion while maintaining functional group integrity.
- Extract the product using ethyl acetate, followed by alkaline washing and purification to obtain high-purity azide compounds with minimal waste generation.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis platform delivers transformative benefits for procurement and supply chain operations by addressing fundamental cost drivers and reliability challenges inherent in traditional azide production methods. The elimination of hazardous reagents reduces regulatory compliance burdens while lowering insurance premiums and safety training requirements across manufacturing facilities. The use of water as primary solvent significantly decreases waste disposal costs compared to organic solvent-based processes, while the simplified purification protocol reduces cycle times without requiring specialized equipment investments. These operational improvements collectively enhance supply chain resilience by minimizing vulnerability to single-point failures common in multi-step conventional syntheses.
- Cost Reduction in Manufacturing: The substitution of expensive palladium catalysts with economical copper(I) oxide generates substantial savings in raw material costs while eliminating costly heavy metal removal steps required in alternative processes. The aqueous reaction medium reduces solvent procurement expenses by over fifty percent compared to traditional organic solvent systems, and the simplified workup procedure decreases labor requirements through reduced processing steps. Most significantly, the elimination of cryogenic or high-pressure equipment requirements lowers capital expenditure barriers for new production lines while reducing maintenance costs associated with specialized instrumentation.
- Enhanced Supply Chain Reliability: The utilization of globally available aryl boronic acid precursors and standard sodium azide sources mitigates single-supplier dependencies that plague conventional synthesis routes relying on specialized reagents. The robustness of the process across varying environmental conditions ensures consistent output quality regardless of regional manufacturing facility differences, while the room temperature operation eliminates seasonal production variations common in temperature-sensitive reactions. This reliability is further enhanced by the catalyst recycling capability demonstrated in patent examples, which maintains consistent yields through multiple production cycles without requiring fresh catalyst batches.
- Scalability and Environmental Compliance: The absence of extreme reaction parameters enables seamless scale-up from laboratory development to commercial production volumes without reoptimization phases typically required when transitioning between scales. The aqueous waste streams generated are readily treatable using standard industrial water treatment systems, significantly reducing environmental compliance costs compared to halogenated solvent waste streams from conventional methods. Furthermore, the elimination of explosive intermediates enhances facility safety profiles while reducing regulatory scrutiny during inspections, facilitating faster approval timelines for new manufacturing sites.
Frequently Asked Questions (FAQ)
The following questions address critical commercial considerations regarding implementation of this patented technology based on extensive analysis of its technical specifications and operational parameters. These insights derive directly from the patent's experimental data and implementation examples, providing actionable guidance for procurement teams evaluating this methodology for integration into existing supply chains.
Q: How does this method improve upon traditional azide synthesis routes?
A: This patented process eliminates hazardous reagents like iodazide by using stable sodium azide with copper catalysis in water solvent, significantly enhancing safety while maintaining high functional group tolerance across diverse aromatic substrates.
Q: What is the catalyst recycling capability of this system?
A: The reaction system demonstrates excellent recyclability; after product extraction, adding fresh reagents allows multiple catalytic cycles with minimal yield reduction, as validated in patent examples showing consistent performance through three reuse cycles.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the mild reaction conditions (ambient pressure, near-room temperature) and simple aqueous workup make this method inherently scalable from laboratory to commercial production volumes without specialized equipment requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Azide Compounds Supplier
Our company leverages this patented technology to deliver exceptional value through extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications required by global regulatory authorities. NINGBO INNO PHARMCHEM's rigorous QC labs implement comprehensive analytical protocols including NMR spectroscopy and mass spectrometry validation to ensure consistent product quality across all batch sizes. Our specialized expertise in copper-catalyzed transformations enables us to optimize this methodology for specific client requirements while maintaining all environmental and safety advantages demonstrated in the original patent disclosure.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this technology can enhance your specific supply chain operations. Please contact us directly to obtain specific COA data and route feasibility assessments tailored to your production requirements.
