Optimizing Cefsulodin Sodium Production: Advanced Purification Strategies for Global Pharmaceutical Supply Chains
Optimizing Cefsulodin Sodium Production: Advanced Purification Strategies for Global Pharmaceutical Supply Chains
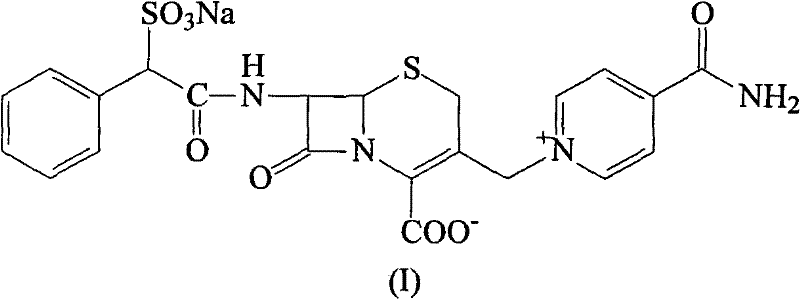
The landscape of third-generation cephalosporin manufacturing is constantly evolving, driven by the imperative for higher purity standards and more robust supply chains. Patent CN101891755B introduces a transformative methodology for the refinement and purification of Cefsulodin Sodium, a critical antibiotic used extensively in treating Pseudomonas aeruginosa infections. This intellectual property outlines a sophisticated three-stage process that leverages acid-alkali reactions, activated carbon adsorption, and preparative chromatography to overcome historical limitations in yield and quality. For R&D directors and procurement specialists, understanding this technology is paramount, as it represents a shift towards more reliable API intermediate supplier capabilities. The method not only addresses the intrinsic difficulties associated with the synthesis of complex side chains but also ensures that the final bulk drug meets stringent pharmacopoeial requirements. By integrating this purification logic, manufacturers can significantly mitigate the risks associated with batch-to-batch variability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of Cefsulodin has been plagued by significant technical hurdles, primarily stemming from the complexity of its side chain synthesis. The conventional route typically involves an acylation reaction between 7-ACA and alpha-sulfo phenyllacetyl chloride, followed by quaternary ammoniation with isonicotinamide. However, the preparation of alpha-sulfo phenyllacetyl chloride is notoriously difficult, characterized by challenging purification steps and low overall yields. This bottleneck often results in a crude bulk material laden with unreacted starting materials, isomeric impurities, and degradation products that are structurally similar to the target molecule. Traditional recrystallization techniques frequently fail to remove these persistent impurities, leading to a final product that may struggle to meet the rigorous purity specifications required for injectable antibiotics. Furthermore, the reliance on harsh reaction conditions in older methods can compromise the stability of the beta-lactam ring, resulting in reduced potency and potential safety concerns for clinical applications.
The Novel Approach
In stark contrast, the novel approach detailed in the patent data offers a streamlined downstream processing strategy that bypasses the synthetic bottlenecks by focusing on high-efficiency purification. Instead of attempting to perfect the difficult synthesis of the side chain precursor, this method accepts the crude bulk material and subjects it to a rigorous refinement protocol. The process initiates with a controlled acidification step to precipitate the target compound, effectively separating it from highly soluble impurities. This is followed by a targeted adsorption phase using activated carbon, which removes colored bodies and organic trace contaminants without degrading the sensitive antibiotic structure. Finally, the implementation of preparative high-performance liquid chromatography (HPLC) provides a resolution capability far superior to standard crystallization, ensuring that even trace level impurities are eliminated. This holistic approach transforms a complex manufacturing challenge into a manageable, scalable operation suitable for industrial production.
Mechanistic Insights into Acid-Base Precipitation and Chromatographic Separation
The core of this purification technology lies in the precise manipulation of physicochemical properties, specifically solubility and polarity, to isolate Cefsulodin Sodium. The initial step involves dissolving the bulk material in water and carefully adjusting the pH to a range of 1.5 to 2.5 using acids such as hydrochloric acid or phosphoric acid. At this specific acidic pH, the zwitterionic nature of the molecule shifts, causing the Cefsulodin to precipitate out of the aqueous solution while many polar impurities remain dissolved. This acid-base reaction is critical; if the pH is too high, precipitation is incomplete, and if too low, degradation of the beta-lactam ring may occur. The subsequent filtration yields a crude solid that is significantly enriched in the target compound, setting the stage for the final polishing steps. This mechanism demonstrates a deep understanding of the molecule's ionization constants and allows for a cost reduction in pharmaceutical manufacturing by minimizing solvent usage compared to extensive solvent-based extractions.
Following precipitation, the process employs preparative chromatography, which acts as the ultimate gatekeeper for product quality. The mobile phase consists of a specific volume ratio of 1:3 acetone to an alkaline solution with a pH between 8 and 10. This alkaline environment ensures the molecule remains in its soluble sodium salt form during elution, while the acetone modifies the polarity to optimize interaction with the stationary phase, which can be silica gel or aluminum oxide. The flow velocity is tightly controlled between 3.8 and 5.6 ml/min at temperatures of 30-35°C to maximize resolution. This mechanistic precision ensures that the commercial scale-up of complex pharmaceutical intermediates is feasible without sacrificing purity. By strictly controlling these parameters, the process effectively separates the Cefsulodin from any remaining isomers or hydrolysis products, delivering a high-purity API intermediate that is ready for formulation.
How to Synthesize Cefsulodin Sodium Efficiently
Implementing this purification protocol requires strict adherence to the operational parameters defined in the patent to ensure reproducibility and safety. The process is designed to be robust, allowing for variations in the starting bulk material quality while still delivering a consistent final product. Operators must pay close attention to the temperature controls during the activated carbon treatment phase, maintaining the solution at 50°C to maximize adsorption efficiency without thermal degradation. The transition from laboratory scale to pilot plant requires careful calibration of the HPLC systems to maintain the critical flow rates and pressure limits described. Below is the structured guide for executing this synthesis and purification workflow effectively.
- Dissolve Cefsulodin bulk material in water and adjust pH to 1.5-2.5 using acid to precipitate crude Cefsulodin.
- Dissolve the precipitate in organic solvent, add 0.1-0.5% activated carbon, stir at 50°C, and filter to remove impurities.
- Perform preparative HPLC separation using an acetone and alkaline solution mobile phase to isolate the highly purified final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this purification technology translates directly into enhanced operational resilience and cost efficiency. The simplification of the process flow eliminates the need for multiple, labor-intensive recrystallization cycles that are common in legacy methods. By streamlining the production pathway, manufacturers can reduce the overall cycle time, thereby improving the responsiveness of the supply chain to market demands. The high yield reported in the patent embodiments suggests that raw material utilization is optimized, which is a key driver for reducing the cost of goods sold (COGS). Furthermore, the use of common, industrially available solvents and reagents ensures that the supply chain is not vulnerable to shortages of exotic or highly regulated chemicals. This reliability makes the manufacturer a preferred partner for long-term contracts where continuity of supply is non-negotiable.
- Cost Reduction in Manufacturing: The elimination of complex synthetic steps for the side chain precursor and the replacement with a robust purification train significantly lowers the barrier to entry for production. By avoiding the expensive and low-yield preparation of alpha-sulfo phenyllacetyl chloride, the overall material costs are drastically reduced. Additionally, the high recovery rates achieved through the precipitation and chromatography steps mean that less starting material is wasted, contributing to substantial cost savings over large production volumes. The process avoids the use of precious metal catalysts or hazardous reagents that would require expensive disposal protocols, further enhancing the economic viability of the method.
- Enhanced Supply Chain Reliability: The reliance on standard unit operations such as filtration, adsorption, and chromatography ensures that the manufacturing process can be easily replicated across different facilities. This flexibility reduces the risk of supply disruptions caused by equipment failure at a single site. The raw materials required, such as acetonitrile, acetone, and common acids, are commodity chemicals with stable global supply networks. This accessibility ensures that production schedules can be maintained even during periods of market volatility, providing a secure source of high-purity Cefsulodin Sodium for downstream formulation partners.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory optimization to multi-ton commercial production. The use of activated carbon for decolorization and impurity removal is an environmentally friendly alternative to solvent-heavy extraction methods, reducing the volume of hazardous waste generated. Moreover, the aqueous nature of the initial precipitation step minimizes the release of volatile organic compounds (VOCs) into the atmosphere. This alignment with green chemistry principles not only simplifies regulatory compliance but also enhances the corporate sustainability profile of the manufacturing entity, a factor increasingly important to global pharmaceutical buyers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and sourcing of Cefsulodin Sodium using this advanced purification methodology. These insights are derived directly from the patent specifications and are intended to clarify the capabilities and limitations of the process for potential partners. Understanding these details is crucial for making informed decisions about supplier qualification and process integration.
Q: What is the primary advantage of the patented purification method for Cefsulodin Sodium?
A: The primary advantage is the significant enhancement of product purity and content through a combination of acid-alkali reaction, active carbon adsorption, and preparative chromatography, ensuring safety for clinical application while maintaining a simple and low-cost industrial process.
Q: Why is the preparation of alpha-sulfo phenyllacetyl chloride considered a bottleneck in traditional synthesis?
A: Traditional synthesis relies on acylation with alpha-sulfo phenyllacetyl chloride, which is notoriously difficult to prepare and purify. This complexity often leads to lower yields and higher impurity profiles in the final antibiotic product, necessitating robust downstream purification strategies.
Q: How does the preparative HPLC step contribute to the final quality of the API?
A: The preparative HPLC step utilizes a specific mobile phase ratio of acetone to alkaline solution (1:3) at controlled pH levels (8-10) to effectively separate the target Cefsulodin molecule from closely related structural impurities and byproducts that standard crystallization cannot remove.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefsulodin Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize that the technical potential of a patent is only realized through flawless execution at scale. As a premier CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate balance of pH and temperature required for Cefsulodin purification is maintained with absolute precision. Our facilities are equipped with state-of-the-art preparative chromatography systems and rigorous QC labs capable of verifying stringent purity specifications down to the ppm level. We understand that for a critical antibiotic like Cefsulodin, consistency is not just a metric but a commitment to patient safety, and our quality management systems are designed to uphold this standard without compromise.
We invite global pharmaceutical companies to leverage our expertise to optimize their supply chains and reduce time-to-market for essential therapies. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that evaluates how this specific purification route can be integrated into your existing portfolio to maximize efficiency. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your volume requirements. Let us demonstrate how our commitment to innovation and quality can become the cornerstone of your antibiotic supply strategy.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
