Scalable Synthesis of N-Difluoromethylthio Phthalimide for Advanced Drug Discovery
Scalable Synthesis of N-Difluoromethylthio Phthalimide for Advanced Drug Discovery
The introduction of fluorine-containing functional groups remains a cornerstone strategy in modern medicinal chemistry, particularly for enhancing metabolic stability and modulating lipophilicity in active pharmaceutical ingredients (APIs). Among these, the difluoromethylthio (SCF2H) group has emerged as a critical pharmacophore, offering unique electronic properties distinct from trifluoromethyl or methylthio groups. However, the widespread adoption of SCF2H-containing molecules has historically been hindered by the complexity and cost of introducing this moiety. A groundbreaking solution is presented in Chinese Patent CN107540598B, which details a highly efficient, two-step method for preparing N-difluoromethylthio phthalimide compounds. This electrophilic reagent serves as a versatile building block for late-stage functionalization, addressing the urgent demand for reliable pharmaceutical intermediate suppliers capable of delivering complex fluorinated scaffolds at scale.
The significance of this technology extends beyond mere academic interest; it represents a tangible shift towards more sustainable and economically viable manufacturing processes for high-value fine chemicals. By utilizing inexpensive starting materials such as benzyl mercaptan and avoiding precious metal catalysts, the disclosed method lowers the barrier to entry for incorporating SCF2H groups into drug candidates. For R&D teams exploring new chemical space, access to such robust synthetic routes is essential for accelerating lead optimization programs without being constrained by supply chain bottlenecks or prohibitive reagent costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
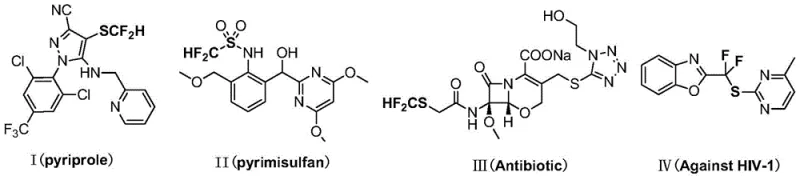
Prior to this innovation, the synthesis of electrophilic difluoromethylthiolating agents was fraught with significant technical and economic challenges. Traditional approaches often relied on the use of stoichiometric amounts of expensive organometallic reagents, such as the silver-based complex SIPrAgSCF2H reported by the Shen group. While effective for specific transformations like Sandmeyer reactions, these silver-mediated protocols generate substantial quantities of metallic waste and require rigorous purification steps to remove residual silver from the final product, which is unacceptable for GMP manufacturing. Furthermore, alternative methods involving xenon difluoride (XeF2) or difluorocarbene insertion into thiols frequently suffer from harsh reaction conditions, poor functional group tolerance, and safety concerns related to the handling of volatile or toxic intermediates.
Another historical bottleneck involved the direct preparation of the key reagent, N-difluoromethylthiophthalimide (Compound 3). Previous literature described its synthesis via nucleophilic substitution using the aforementioned silver reagent, resulting in moderate yields around 66% but at a prohibitively high cost due to the silver component. Additionally, the instability of difluoromethylthio chloride (HCF2SCl), a potential precursor with a low boiling point of 25-35°C, made its isolation and storage hazardous. These factors collectively limited the availability of high-purity difluoromethylthio reagents, creating a supply gap for agrochemical and pharmaceutical manufacturers seeking to exploit the unique lipophilicity profile of the SCF2H group.
The Novel Approach
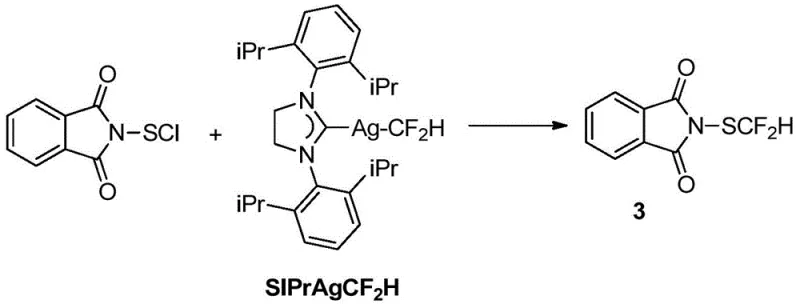
The methodology disclosed in Patent CN107540598B fundamentally reimagines the synthetic landscape by decoupling the production of the difluoromethylthio group from precious metal chemistry. The core innovation lies in a streamlined two-step sequence that begins with the chlorination of difluoromethyl-substituted benzyl thiol (Compound 2) to generate HCF2SCl in situ. Instead of isolating this volatile and potentially dangerous intermediate, the process immediately subjects the reaction mixture to nucleophilic attack by a phthalimide salt (Compound 1). This telescoped approach not only mitigates safety risks associated with handling HCF2SCl but also drives the reaction to completion through immediate consumption of the electrophile, thereby maximizing atom economy and overall yield.
Crucially, this new route utilizes commodity chemicals that are readily available on the global market, such as benzyl mercaptan, chlorine gas, and potassium phthalimide. The reaction conditions are remarkably mild, typically proceeding between -20°C and 30°C in common organic solvents like chloroform or dichloromethane. This operational simplicity translates directly into reduced capital expenditure for manufacturing facilities, as it does not require specialized high-pressure or cryogenic equipment. By eliminating the need for silver catalysts and simplifying the workup procedure to basic filtration and chromatography, this method offers a compelling value proposition for cost reduction in pharmaceutical intermediate manufacturing, ensuring a stable supply of this critical building block.
Mechanistic Insights into Electrophilic Difluoromethylthiolation
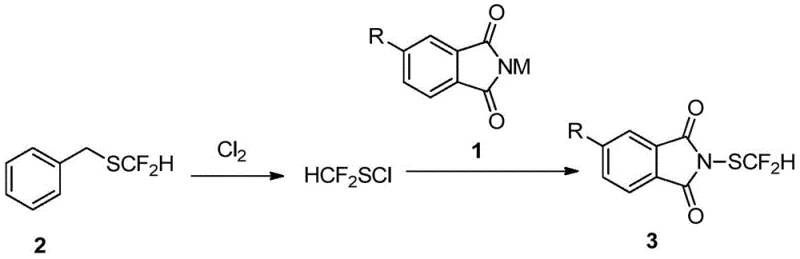
The chemical elegance of this process is rooted in the controlled generation and immediate utilization of the electrophilic sulfur species. In the first stage, chlorine gas acts as an oxidant and chlorinating agent, reacting with the sulfur atom of the difluoromethyl benzyl thiol. This transformation converts the relatively nucleophilic thiol into the highly electrophilic sulfenyl chloride (HCF2SCl). The presence of the electron-withdrawing difluoromethyl group stabilizes the sulfur center sufficiently to allow for the subsequent substitution, yet the molecule remains reactive enough to engage with nitrogen nucleophiles. The use of a slight excess of chlorine ensures complete conversion of the thiol, preventing the formation of disulfide byproducts which could complicate downstream purification.
In the second stage, the phthalimide anion, generated from the potassium salt, acts as a potent nucleophile. It attacks the sulfur atom of the in-situ generated HCF2SCl, displacing the chloride ion and forming the robust N-S bond characteristic of the final product. The reaction kinetics are carefully managed by temperature control; initiating the mixing at -10°C prevents the rapid decomposition of the unstable sulfenyl chloride, while warming to room temperature provides the necessary activation energy for the substitution to proceed efficiently. This mechanistic pathway avoids the radical mechanisms often seen in other difluoromethylthiolation strategies, thereby minimizing side reactions and ensuring a clean impurity profile that is essential for regulatory compliance in drug substance production.
How to Synthesize N-Difluoromethylthio Phthalimide Efficiently
Implementing this synthesis requires precise control over reaction parameters to ensure safety and maximize yield. The process begins with the preparation of the difluoromethyl thiol precursor, which can be achieved via difluorocarbene insertion into benzyl mercaptan using sources like F-22 gas under basic conditions. Once the thiol is secured, the critical chlorination step must be performed in a well-ventilated system capable of handling chlorine gas safely. The detailed standardized synthetic steps, including specific molar ratios, solvent volumes, and temperature ramps optimized for commercial scale-up, are outlined in the guide below.
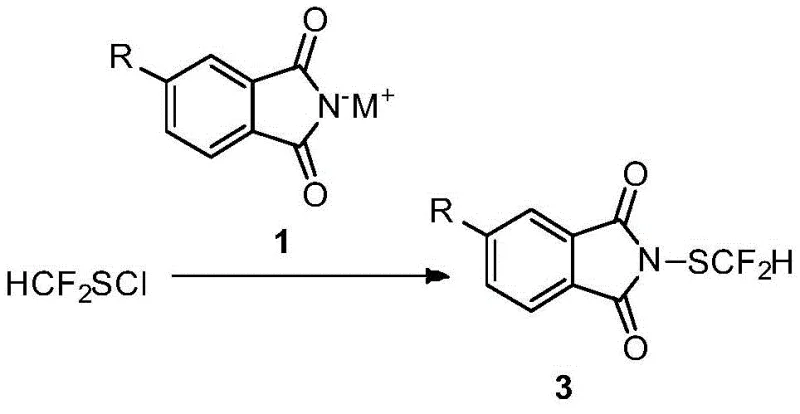
- Prepare difluoromethyl-substituted benzyl thiol by reacting benzyl mercaptan with a difluorocarbene precursor like F-22 under basic conditions.
- Generate difluoromethylthio chloride in situ by reacting the thiol intermediate with chlorine gas in an organic solvent at low temperature (-10°C).
- Add potassium phthalimide salt to the reaction mixture without isolation, warm to room temperature, and purify via flash chromatography to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from silver-mediated chemistry to this base-metal-free protocol represents a strategic opportunity to de-risk the supply of critical fluorinated intermediates. The reliance on stoichiometric silver reagents in previous methods created a vulnerability to fluctuations in precious metal prices and introduced complex waste disposal requirements. By shifting to a chlorine-based oxidation system using abundant organic feedstocks, manufacturers can achieve significant cost savings in raw material acquisition. Furthermore, the simplified purification process reduces the consumption of silica gel and solvents during workup, contributing to a lower overall cost of goods sold (COGS) and a smaller environmental footprint.
- Cost Reduction in Manufacturing: The elimination of expensive organosilver reagents is the primary driver of cost efficiency in this new process. Silver salts are not only costly to purchase but also require recovery or specialized disposal, adding hidden operational expenses. By replacing these with chlorine gas and potassium phthalimide, the variable cost per kilogram of the final intermediate is drastically reduced. Additionally, the high conversion rates and yields reported in the patent minimize the loss of valuable starting materials, ensuring that every gram of input contributes effectively to the final output, which is vital for maintaining healthy margins in competitive generic and specialty chemical markets.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including benzyl mercaptan and phthalimide salts, are commodity chemicals produced by multiple global suppliers. This diversification of the supply base mitigates the risk of single-source dependency that often plagues specialized reagent markets. Moreover, the robustness of the reaction conditions means that production is less susceptible to minor variations in utility supply or equipment performance. This reliability ensures consistent delivery schedules for downstream customers, allowing pharmaceutical companies to plan their clinical trial material production with greater confidence and reduced lead times for high-purity intermediates.
- Scalability and Environmental Compliance: From an engineering perspective, the mild temperatures and ambient pressure requirements of this reaction make it highly amenable to scale-up from kilogram to multi-ton batches. The avoidance of pyrophoric reagents or extreme cryogenic conditions simplifies the reactor design and safety systems needed for commercial production. Environmentally, the process generates fewer heavy metal contaminants, easing the burden on wastewater treatment facilities and aligning with increasingly stringent global regulations on industrial effluent. This alignment with green chemistry principles enhances the corporate sustainability profile of manufacturers adopting this technology.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this difluoromethylthiolation technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on safety, scope, and optimization strategies for potential licensees or manufacturing partners.
Q: Why is the in-situ generation of HCF2SCl preferred over isolation?
A: Difluoromethylthio chloride (HCF2SCl) has a low boiling point (25-35°C) and potential toxicity. The patent methodology generates it in situ and immediately consumes it in the subsequent step, significantly enhancing operational safety and preventing yield loss due to volatility.
Q: What are the primary cost advantages of this method compared to silver-mediated routes?
A: Conventional methods often rely on expensive organosilver reagents (e.g., SIPrAgSCF2H) in stoichiometric amounts. This novel route utilizes commodity chemicals like benzyl mercaptan and chlorine gas, eliminating the need for precious metal catalysts and drastically reducing raw material costs.
Q: Can this method accommodate electron-withdrawing groups on the phthalimide ring?
A: Yes, the patent demonstrates successful synthesis of derivatives such as N-difluoromethylthio-4-nitrophthalimide. The mild reaction conditions (-10°C to room temperature) ensure compatibility with sensitive functional groups, maintaining high purity and yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Difluoromethylthio Phthalimide Supplier
The technological advancements detailed in Patent CN107540598B underscore the dynamic nature of fluorine chemistry and its pivotal role in next-generation therapeutics. At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating such innovative laboratory methodologies into robust, commercial-scale manufacturing processes. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive materials that meet the most stringent purity specifications. Our rigorous QC labs are equipped to analyze complex fluorinated impurities, guaranteeing that every batch of N-difluoromethylthio phthalimide delivered adheres to the highest quality standards required for API synthesis.
We invite R&D and procurement leaders to collaborate with us to leverage this cost-effective synthetic route for their projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for both performance and economic efficiency in the rapidly evolving landscape of fluorinated pharmaceutical intermediates.
