Advanced Iodine-Catalyzed Synthesis of Pyrazolopyrimidine Derivatives for Commercial Scale-Up
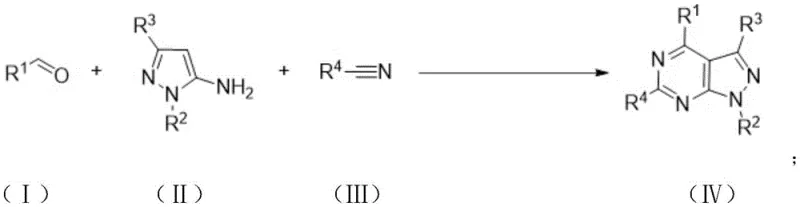
The chemical landscape for nitrogen-containing heterocycles is undergoing a significant transformation driven by the demand for greener, more efficient synthetic routes. Patent CN115304607B introduces a groundbreaking preparation method for pyrazolopyrimidine derivatives, utilizing an iodine-catalyzed three-component coupling reaction. This technology addresses critical bottlenecks in the synthesis of complex fused-ring systems, which are pivotal scaffolds in both medicinal chemistry and advanced material science. By leveraging molecular iodine as a cost-effective catalyst and atmospheric oxygen as the terminal oxidant, this process eliminates the reliance on precious transition metals and harsh stoichiometric oxidants. The resulting methodology not only streamlines the construction of the pyrazolopyrimidine core but also ensures high atom economy and operational safety, positioning it as a superior alternative for the manufacturing of high-value intermediates.
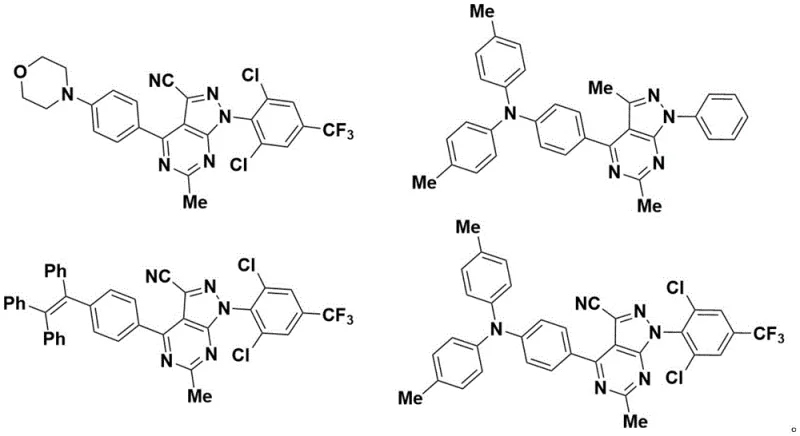
Furthermore, the versatility of this synthetic platform extends beyond simple structural analogs. The patent demonstrates exceptional substrate tolerance, accommodating a wide array of functional groups including halogens, trifluoromethyl groups, and bulky aromatic systems. This breadth of compatibility is essential for R&D teams aiming to diversify their chemical libraries for drug discovery or to fine-tune the photophysical properties of organic luminescent materials. The ability to synthesize these derivatives in a single pot reduces the cumulative environmental footprint and minimizes the loss of material associated with intermediate isolation steps. As the industry shifts towards sustainable manufacturing practices, the adoption of such iodine-mediated aerobic oxidation protocols represents a strategic advantage for producers seeking to optimize both cost and compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of pyrazolopyrimidine frameworks has been fraught with synthetic challenges that hinder large-scale production. Traditional methodologies often rely on multi-step sequences involving the pre-functionalization of starting materials, which inherently lowers the overall yield and increases the generation of chemical waste. Many classical routes require the use of strong acids or bases at elevated temperatures, leading to poor chemoselectivity and the formation of difficult-to-remove impurities. Moreover, conventional catalytic systems frequently employ expensive palladium or copper complexes that necessitate rigorous removal processes to meet the stringent residual metal limits imposed by regulatory bodies in the pharmaceutical sector. These factors collectively contribute to high production costs and extended lead times, creating significant supply chain vulnerabilities for manufacturers dependent on these critical intermediates.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a direct, one-pot condensation of aldehydes, 5-aminopyrazoles, and nitriles mediated by elemental iodine. This strategy bypasses the need for pre-activated substrates and precious metal catalysts, fundamentally simplifying the process flow. The reaction proceeds smoothly under aerobic conditions, where molecular oxygen serves as a clean oxidant, producing water as the only byproduct. This shift from stoichiometric oxidants to catalytic aerobic oxidation drastically improves the E-factor of the process. Additionally, the use of iodine, a readily available and inexpensive halogen, removes the economic barrier associated with noble metal catalysis. The operational simplicity of heating the mixture in a common solvent like acetonitrile allows for easy scale-up, making this method highly attractive for commercial adoption in the fine chemical industry.
Mechanistic Insights into Iodine-Catalyzed Cyclization
The mechanistic pathway of this transformation is a sophisticated interplay of nucleophilic addition and oxidative cyclization. Initially, the iodine catalyst activates the aldehyde component, facilitating a nucleophilic attack by the amino group of the 5-aminopyrazole. This step forms an imine intermediate, which is crucial for the subsequent ring closure. The presence of the nitrile component then participates in a Ritter-type amination process, where the nitrile nitrogen attacks the activated iminium species. This sequence is followed by an intramolecular cyclization that constructs the pyrimidine ring fused to the pyrazole core. The role of iodine is multifaceted; it acts not only as a Lewis acid to activate electrophiles but also mediates the electron transfer processes required for the final aromatization of the heterocyclic system under an oxygen atmosphere.
Understanding the impurity profile is vital for maintaining high product quality. The high selectivity of this iodine-catalyzed system minimizes the formation of side products such as over-oxidized species or polymerization byproducts often seen in radical-based oxidations. The mild reaction conditions preserve sensitive functional groups on the aromatic rings, allowing for the synthesis of complex derivatives without the need for protecting group strategies. This chemoselectivity ensures that the crude reaction mixture is cleaner, thereby simplifying downstream purification. For R&D directors, this means faster iteration cycles in structure-activity relationship (SAR) studies, as the synthetic route reliably delivers the target scaffold with minimal structural degradation or unwanted modification of peripheral substituents.
How to Synthesize Pyrazolopyrimidine Derivatives Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing these valuable heterocycles with high efficiency. The process begins with the precise stoichiometric mixing of the aldehyde, aminopyrazole, and nitrile components in a reaction vessel equipped for heating and stirring. The addition of the iodine catalyst is critical, with molar ratios optimized to balance reaction rate and cost. The reaction is typically conducted in acetonitrile, which serves a dual purpose as both the solvent and a potential reactant, further enhancing atom economy. Heating the mixture to approximately 120°C under an oxygen or air atmosphere drives the cyclization to completion. Detailed standardized synthesis steps follow below to ensure reproducibility and safety during implementation.
- Mix aldehyde compound, 5-aminopyrazole compound, nitrile compound, and iodine catalyst in a suitable organic solvent such as acetonitrile.
- Heat the reaction mixture to 110-140°C under an oxygen or air atmosphere for 10 to 20 hours to facilitate cyclization.
- Cool the product, extract with ethyl acetate, wash with sodium thiosulfate, and purify via column chromatography or recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this iodine-catalyzed methodology offers profound strategic benefits centered around cost stability and operational resilience. The elimination of precious metal catalysts removes exposure to the volatile pricing markets of palladium and platinum, ensuring more predictable raw material costs. Furthermore, the one-pot nature of the reaction significantly reduces the number of unit operations required, translating to lower energy consumption and reduced labor hours per batch. The use of commodity chemicals like iodine and acetonitrile ensures a secure supply base, mitigating the risk of shortages that often plague specialized reagents. This process intensification allows manufacturers to produce higher volumes within existing infrastructure, effectively increasing capacity without significant capital expenditure.
- Cost Reduction in Manufacturing: The replacement of expensive transition metal catalysts with inexpensive molecular iodine results in substantial raw material cost savings. Additionally, the simplified workup procedure, which avoids complex metal scavenging steps, reduces the consumption of auxiliary materials and solvents. The high atom economy of the three-component coupling ensures that a greater proportion of the input mass is converted into the final product, minimizing waste disposal costs. These factors combine to drastically lower the overall cost of goods sold (COGS) for pyrazolopyrimidine derivatives, enhancing profit margins for downstream applications.
- Enhanced Supply Chain Reliability: By relying on widely available bulk chemicals rather than specialized proprietary catalysts, the supply chain becomes more robust and less susceptible to disruptions. The ability to use air or oxygen as the oxidant eliminates the need for storing and handling hazardous stoichiometric oxidants, improving site safety and reducing regulatory compliance burdens. The scalability of the process from gram to kilogram scales ensures that supply can be ramped up quickly to meet market demand without the need for lengthy process re-validation. This reliability is crucial for maintaining continuous production schedules in the fast-paced pharmaceutical and electronics sectors.
- Scalability and Environmental Compliance: The green chemistry principles embedded in this method, such as the use of low-toxicity reagents and the generation of minimal waste, align perfectly with modern environmental regulations. The absence of heavy metal residues simplifies the environmental impact assessment and wastewater treatment processes. The process is inherently safer due to the mild reaction conditions and the avoidance of pyrophoric reagents. This environmental and safety profile facilitates easier permitting for new production lines and supports corporate sustainability goals, making the supply chain more resilient to future regulatory changes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the feasibility and advantages of the method for potential partners and licensees.
Q: What are the primary advantages of the iodine-catalyzed method over traditional synthesis?
A: The iodine-catalyzed method offers high atom economy, operates under mild aerobic conditions without expensive transition metals, and utilizes a simple one-pot procedure that significantly reduces waste and operational complexity compared to multi-step traditional routes.
Q: Can this synthesis method be scaled for industrial production of OLED materials?
A: Yes, the process uses low-toxicity reagents and common solvents like acetonitrile, making it environmentally friendly and highly suitable for scale-up. The robust substrate scope allows for the production of various photoluminescent derivatives required for organic light-emitting devices.
Q: What is the typical yield and purity profile for these derivatives?
A: Experimental data indicates yields ranging from moderate to excellent (up to 88% in specific examples), with high selectivity. The products are easily purified via standard chromatography or recrystallization, ensuring high purity specifications suitable for pharmaceutical and electronic applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrazolopyrimidine Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the iodine-catalyzed synthesis route for pyrazolopyrimidine derivatives. As a leading CDMO partner, we possess the technical expertise to translate this patented laboratory methodology into robust, commercial-scale manufacturing processes. Our facilities are equipped to handle diverse synthetic pathways, ranging from small-batch R&D quantities of 100 kgs to full-scale commercial production of 100 MT annually. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, ensuring that every batch conforms to the highest standards required for pharmaceutical intermediates and advanced electronic materials.
We invite you to collaborate with us to optimize your supply chain for these critical building blocks. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our scale and expertise, we can help you realize the full economic benefits of this green synthesis technology. Please contact us to request specific COA data and route feasibility assessments for your target molecules, and let us demonstrate how our engineering capabilities can accelerate your project timelines.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
