Revolutionizing Tranexamic Acid Production: A Barium-Free Continuous Flow Strategy for Global Pharma Supply Chains
Revolutionizing Tranexamic Acid Production: A Barium-Free Continuous Flow Strategy for Global Pharma Supply Chains
The pharmaceutical industry is currently witnessing a paradigm shift in the synthesis of critical hemostatic agents, driven by the urgent need for safer, more efficient manufacturing protocols. Patent CN114014768A introduces a groundbreaking methodology for the preparation of tranexamic acid that fundamentally addresses the long-standing issue of barium salt impurities. This innovation leverages advanced continuous flow reactor technology to execute a four-step sequence involving amine substitution, catalytic hydrogenation, configuration conversion, and refining. By transitioning from traditional batch processing to a modular continuous flow system, this technical breakthrough not only ensures the complete absence of toxic heavy metal residues but also significantly enhances the overall process safety profile. For R&D directors and procurement specialists alike, this represents a vital evolution in API intermediate manufacturing, promising a supply of high-purity tranexamic acid that meets the most stringent global regulatory standards for injectable medications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of tranexamic acid has been plagued by significant safety and quality control challenges, primarily stemming from the reliance on barium hydroxide during the configuration conversion step. In traditional synthetic routes, barium salts are introduced to facilitate the isomerization from the cis-form to the therapeutically active trans-form, necessitating a subsequent neutralization with sulfuric acid to precipitate and remove the barium as barium sulfate. This process is inherently risky; incomplete filtration of the precipitate can leave behind microscopic insoluble particles in the final drug substance. For an injectable medication, such particulate matter poses severe clinical hazards, including pyrogen-like reactions, inflammatory responses, and potentially life-threatening vascular embolisms. Furthermore, the use of strong acids and heavy metals creates a corrosive environment that demands expensive titanium-lined reactors, driving up capital expenditure and complicating maintenance schedules for manufacturing facilities.
The Novel Approach
The methodology outlined in patent CN114014768A offers a transformative solution by completely eliminating the introduction of barium salts throughout the entire synthesis pathway. Instead of relying on hazardous heavy metal promoters, this novel approach utilizes a sophisticated continuous flow reactor system divided into three distinct functional units. This architectural change allows for precise control over reaction parameters such as residence time, temperature, and pressure, which are critical for optimizing yield and selectivity. By removing the barium precipitation and filtration steps, the process inherently simplifies the downstream purification workflow, reducing the generation of dangerous solid wastes. This shift not only aligns with green chemistry principles but also drastically lowers the operational complexity, making it an ideal candidate for reliable tranexamic acid supplier networks seeking to modernize their production capabilities and ensure uninterrupted supply continuity.
Mechanistic Insights into Continuous Flow Catalytic Hydrogenation
The core of this technological advancement lies in the precise orchestration of catalytic hydrogenation within a continuous flow environment, specifically designed to maximize the formation of the desired trans-isomer. In Unit B of the reactor system, aminomethylbenzoic acid undergoes hydrogenation under strictly controlled alkaline conditions, utilizing a specialized ruthenium or platinum catalyst supported on alumina or carbon. Unlike batch processes where mass transfer limitations often lead to poor selectivity, the continuous flow regime ensures uniform exposure of the substrate to the catalyst surface and hydrogen gas. This results in a dramatic improvement in the cis-to-trans isomer ratio, achieving trans-isomer proportions of up to 85% compared to the mere 15% typical of older methods. The ability to tune the reaction environment dynamically allows manufacturers to suppress the formation of unwanted byproducts, thereby delivering high-purity pharmaceutical intermediates that require less intensive chromatographic separation.
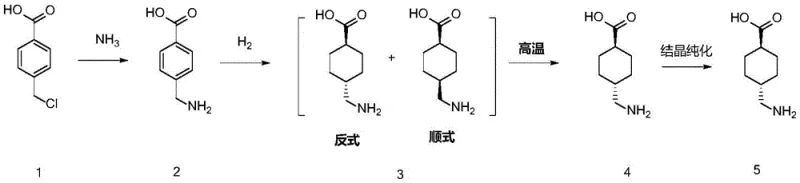
Furthermore, the mechanistic advantage extends to the configuration conversion step performed in Unit C, where the cis-trans mixture is subjected to elevated temperatures and pressures without any barium additives. The continuous flow setup facilitates superior heat transfer efficiency, which is crucial for managing the exothermic nature of high-temperature isomerization reactions. This thermal control prevents localized hot spots that could degrade the product or lead to safety incidents. By maintaining the reaction mixture in a liquid phase under high pressure, the system ensures that the equilibrium shifts favorably towards the thermodynamically stable trans-tranexamic acid. This mechanism effectively decouples the isomerization efficiency from the need for toxic promoters, proving that process intensification through flow chemistry can solve complex stereochemical challenges while adhering to strict safety and environmental compliance standards.
How to Synthesize Tranexamic Acid Efficiently
Implementing this continuous flow strategy requires a systematic approach to reactor design and parameter optimization to fully realize the benefits of barium-free synthesis. The process begins with the dissolution of 4-chloromethylbenzoic acid in a suitable organic solvent mixed with aqueous ammonia, which is then pumped into the first reactor unit for amine substitution. Following this, the intermediate stream is directed to the hydrogenation unit where pH and catalyst loading are meticulously managed to ensure high conversion rates. Finally, the crude mixture enters the high-temperature conversion unit to finalize the stereochemistry before undergoing standard crystallization and refining procedures. The detailed standardized synthesis steps see the guide below for specific operational parameters and safety protocols required for commercial implementation.
- Perform amine substitution on 4-chloromethylbenzoic acid in Unit A of a continuous flow reactor using aqueous ammonia at 60-150°C to generate aminomethylbenzoic acid.
- Execute catalytic hydrogenation in Unit B under alkaline conditions using a Ruthenium or Platinum catalyst to produce a tranexamic acid cis/trans mixture with high trans-isomer selectivity.
- Complete configuration conversion in Unit C at elevated temperatures (180-260°C) and pressure without barium salts, followed by refining to obtain the pure final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this barium-free continuous flow technology translates into tangible strategic advantages that go beyond mere technical specifications. The elimination of barium salts removes an entire category of hazardous waste disposal costs, which are often substantial in heavy metal-mediated syntheses. Additionally, the shift away from strong sulfuric acid environments significantly reduces the corrosion rate of production equipment, extending the lifespan of reactors and minimizing unplanned downtime for maintenance. This robustness ensures a more predictable production schedule, which is critical for maintaining the continuity of supply for essential medicines like tranexamic acid. By simplifying the purification train and removing complex filtration steps for barium sulfate, the overall manufacturing cycle time is compressed, allowing for faster turnover and improved responsiveness to market demand fluctuations.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the fundamental simplification of the workflow and the removal of expensive reagents. By avoiding the use of barium hydroxide and the subsequent acid neutralization steps, the consumption of raw materials is optimized, and the volume of waste effluent requiring treatment is drastically reduced. The alkaline hydrogenation conditions further contribute to cost reduction in API manufacturing by negating the need for costly titanium-lined vessels, allowing the use of standard high-grade stainless steel equipment. These cumulative efficiencies result in a leaner cost structure that enhances competitiveness without compromising on the quality or safety of the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the inherent stability and scalability of the continuous flow platform. Unlike batch processes that are susceptible to variability between runs, flow chemistry offers consistent product quality, reducing the risk of batch failures and rejected shipments. The process relies on readily available starting materials like 4-chloromethylbenzoic acid and common catalysts, mitigating the risk of raw material shortages. This reliability is paramount for reducing lead time for high-purity pharmaceutical intermediates, ensuring that downstream formulation partners receive their materials on schedule and can maintain their own production timelines without interruption.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this technology offers a clear pathway for the commercial scale-up of complex amino acids. The continuous nature of the reactor system allows for linear scaling by increasing run time or numbering up reactor units, rather than the geometric challenges associated with enlarging batch tanks. The significant reduction in hazardous waste discharge aligns with increasingly strict global environmental regulations, future-proofing the manufacturing site against tightening compliance standards. This sustainability angle not only protects the company from regulatory fines but also enhances the brand reputation among eco-conscious stakeholders and partners in the global pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of how this technology outperforms legacy methods. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this process into existing manufacturing lines or for procurement teams assessing the long-term value proposition of suppliers adopting this standard.
Q: Why is the elimination of barium salts critical in tranexamic acid synthesis?
A: Traditional methods utilize barium hydroxide for configuration conversion, which introduces toxic heavy metal impurities. Since tranexamic acid is often administered via injection, even trace insoluble barium particles can cause severe safety risks such as vascular embolism or granuloma. This new process completely eliminates barium usage, ensuring superior safety profiles for parenteral formulations.
Q: How does continuous flow chemistry improve the cis/trans isomer ratio?
A: By utilizing a specialized continuous flow reactor system with precise control over temperature and pressure, the process optimizes the catalytic hydrogenation step. Specifically, operating under alkaline conditions with ruthenium-based catalysts allows for a trans-isomer proportion of up to 85%, significantly higher than the 15% typically seen in conventional batch processes, thereby reducing the burden on downstream purification.
Q: What are the equipment advantages of using alkaline conditions in hydrogenation?
A: Conventional routes often rely on strong sulfuric acid, which causes severe corrosion to reactor vessels and necessitates expensive titanium equipment. This novel approach employs alkaline conditions (using sodium or potassium hydroxide) during hydrogenation, which drastically reduces equipment corrosion, lowers maintenance costs, and mitigates safety risks associated with handling strong acids at high pressures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tranexamic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced manufacturing technologies like continuous flow chemistry is key to securing the future of pharmaceutical supply chains. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the barium-free tranexamic acid process can be seamlessly translated from the laboratory to full-scale industrial output. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch against the highest international standards, guaranteeing that our clients receive materials that are safe, effective, and fully compliant with regulatory requirements for injectable drugs.
We invite global pharmaceutical partners to collaborate with us to leverage these technical advancements for their product portfolios. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume needs and quality targets. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will optimize your supply chain efficiency and reduce the total cost of ownership for your critical API intermediates.
