Advanced Oxidative Carbonylation Technology for High-Purity Alpha,Beta-Unsaturated Alkynamide Manufacturing
Introduction to Next-Generation Alkynamide Synthesis
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct complex amide scaffolds, particularly alpha,beta-unsaturated alkynamides, which serve as critical pharmacophores in bioactive molecules ranging from opioid receptor modulators to kinase inhibitors. Patent CN112142617A introduces a transformative approach to synthesizing these valuable intermediates through an oxidative carbonylation strategy that fundamentally alters the economic and operational landscape of their production. Unlike traditional methods that rely on pre-functionalized carboxylic acid derivatives, this innovation leverages readily available terminal alkynes and diamines as direct feedstocks, utilizing a sophisticated water-soluble palladium catalyst system to drive the reaction with exceptional precision. The technology addresses long-standing challenges in selectivity and catalyst recovery, offering a robust platform for the commercial scale-up of complex pharmaceutical intermediates. By integrating carbon monoxide and oxygen directly into the synthetic sequence, the process eliminates the need for stoichiometric activating agents, thereby reducing waste generation and simplifying downstream purification protocols significantly.
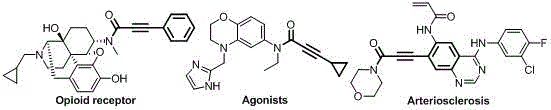
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the alkynamide motif has been fraught with inefficiencies that hinder large-scale manufacturing viability. The most straightforward conventional route involves the direct amidation of alkynoic acids with amines; however, this transformation typically demands harsh thermal conditions that can degrade sensitive functional groups and limit the scope of compatible substrates. Alternatively, chemists have relied on activated derivatives such as alkynyl chlorides or acid anhydrides, but these reagents require stoichiometric amounts of base additives and generate substantial quantities of salt waste during the neutralization steps. Furthermore, earlier attempts to utilize transition metal catalysis for oxidative carbonylation have met with limited success; for instance, nickel-based systems reported in literature often suffer from poor conversion rates and selectivity below 20 percent, rendering them impractical for industrial application. Even palladium iodide systems, while showing some promise, have been plagued by narrow substrate tolerance and an inability to recycle the expensive metal catalyst, leading to prohibitive costs and environmental burdens associated with heavy metal disposal.
The Novel Approach
The methodology disclosed in the patent represents a paradigm shift by employing a highly active palladium catalyst coordinated with a specifically designed water-soluble alpha-diimine ligand. This novel system enables the direct coupling of terminal alkynes and diamines under mild oxidative carbonylation conditions, typically operating between 60°C and 120°C. The reaction proceeds with remarkable chemo-selectivity, consistently achieving product selectivity greater than 99 percent across a diverse array of substrates. A defining feature of this approach is the use of a mixed gas atmosphere of carbon monoxide and oxygen, which serves as the carbonyl source, effectively replacing hazardous and wasteful acylating agents. The process is not only atom-economical but also operationally simple, requiring standard high-pressure reactor equipment that is commonplace in modern fine chemical facilities. This advancement allows for the streamlined production of high-purity alkynamides without the extensive purification steps necessitated by older methodologies.

Mechanistic Insights into Water-Soluble Palladium Catalysis
At the heart of this technological breakthrough lies the unique architecture of the water-soluble alpha-diimine ligand, which orchestrates the catalytic cycle with high fidelity. The ligand, synthesized from 2,6-diisopropylaniline via sulfonation and subsequent condensation with acenaphthenequinone, possesses strong sigma-donating and pi-accepting capabilities that stabilize the palladium center throughout the reaction. Crucially, the introduction of sulfonic acid groups onto the ligand framework imparts hydrophilicity, a feature that is exploited for catalyst recovery. During the catalytic cycle, the palladium species facilitates the activation of the terminal alkyne and the insertion of carbon monoxide, followed by nucleophilic attack by the amine. The electronic properties of the diimine backbone ensure that the oxidative addition and reductive elimination steps proceed rapidly, minimizing the formation of side products such as homocoupled diynes or over-carbonylated species. This precise control over the reaction pathway is what enables the observed selectivity exceeding 99 percent, ensuring that the final crude mixture is predominantly the desired alkynamide.
Impurity control is inherently built into the mechanism through the biphasic nature of the workup procedure. Because the catalyst resides in the aqueous phase due to the sulfonated ligand, simple phase separation after the reaction effectively removes the majority of palladium residues from the organic product layer. This minimizes the risk of metal contamination in the final API intermediate, a critical quality attribute for pharmaceutical manufacturing. Furthermore, the mild reaction conditions prevent thermal degradation of the sensitive alkyne moiety, which is often prone to polymerization or decomposition under the high temperatures required by non-catalytic methods. The stability of the catalyst system over multiple cycles, as evidenced by maintained activity after repeated use, suggests that the ligand effectively prevents the aggregation of palladium into inactive black metal precipitates, a common failure mode in homogeneous carbonylation reactions. This mechanistic robustness translates directly to process reliability and consistent product quality.
How to Synthesize Alpha,Beta-Unsaturated Alkynamides Efficiently
Implementing this oxidative carbonylation protocol requires careful attention to the preparation of the specialized ligand and the management of gas pressures to ensure safety and reproducibility. The synthesis begins with the preparation of the water-soluble ligand, followed by the assembly of the reaction mixture in a pressure-rated vessel. Operators must ensure that the ratio of carbon monoxide to oxygen is strictly controlled within the specified range to maintain the oxidative cycle without risking explosive limits. The following guide outlines the standardized steps derived from the patent examples to achieve optimal yields and purity.
- Prepare the water-soluble alpha-diimine ligand by sulfonating 2,6-diisopropylaniline followed by condensation with acenaphthenequinone.
- Charge an autoclave with terminal alkyne, diamine, palladium salt, the water-soluble ligand, and iodide additive in a solvent like THF.
- Pressurize with CO and O2 mixed gas to 3.0-5.0 MPa and heat to 60-120°C for 6-20 hours, then separate the catalyst via aqueous extraction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this oxidative carbonylation technology offers tangible strategic benefits that extend beyond mere technical performance. The primary advantage lies in the drastic simplification of the raw material supply chain; by utilizing commodity terminal alkynes and simple diamines, manufacturers can bypass the volatile pricing and supply constraints associated with specialized alkynoic acid derivatives or acid chlorides. This shift to abundant feedstocks significantly de-risks the production schedule and enhances the overall resilience of the supply chain against market fluctuations. Moreover, the ability to recycle the catalyst system multiple times without significant loss of activity means that the consumption of precious palladium metal is minimized, leading to substantial cost savings in raw material expenditure over the lifecycle of the product. The elimination of stoichiometric base additives and the reduction of salt waste also lower the costs associated with waste treatment and environmental compliance, contributing to a more sustainable and economically favorable manufacturing profile.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive pre-activated carboxylic acid derivatives and stoichiometric coupling reagents, which traditionally account for a significant portion of the cost of goods. By using direct oxidative carbonylation with gaseous CO, the atom economy is maximized, and the reliance on costly activators is removed entirely. Additionally, the recyclability of the palladium catalyst reduces the effective cost of the metal per kilogram of product, as the same catalyst batch can be utilized for multiple production runs. This cumulative effect results in a significantly lower production cost structure compared to conventional amidation routes, allowing for more competitive pricing in the global market.
- Enhanced Supply Chain Reliability: Sourcing terminal alkynes and simple amines is far more reliable than procuring specialized alkynyl chlorides or anhydrides, which often have limited suppliers and longer lead times. The robustness of the reaction conditions, which tolerate a wide range of functional groups, means that grade variations in raw materials are less likely to cause batch failures. This flexibility ensures a steady flow of intermediates to downstream customers, reducing the risk of production stoppages. Furthermore, the simplified workflow reduces the number of unit operations required, shortening the overall manufacturing cycle time and enabling faster response to demand spikes.
- Scalability and Environmental Compliance: The reaction operates at moderate temperatures and pressures that are well within the capabilities of standard industrial reactors, facilitating seamless scale-up from pilot to commercial production volumes. The biphasic workup strategy simplifies isolation and reduces the volume of organic solvents required for purification, aligning with green chemistry principles. The reduction in hazardous waste generation, particularly salt byproducts and heavy metal residues, eases the burden on wastewater treatment facilities and ensures compliance with increasingly stringent environmental regulations. This makes the process not only scalable but also future-proof against evolving regulatory landscapes.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this oxidative carbonylation technology. These answers are derived directly from the experimental data and specifications outlined in the patent documentation to provide clarity on process capabilities and limitations. Understanding these details is essential for evaluating the feasibility of integrating this method into existing manufacturing workflows.
Q: How is the palladium catalyst recycled in this oxidative carbonylation process?
A: The catalyst utilizes a water-soluble alpha-diimine ligand containing sulfonic acid groups. After the reaction, adding water creates a biphasic system where the catalyst remains in the aqueous phase, allowing for easy separation and reuse without significant loss of activity.
Q: What are the typical reaction conditions for synthesizing these alkynamides?
A: The reaction typically proceeds at temperatures between 60°C and 120°C under a mixed gas pressure of 3.0 to 5.0 MPa (CO/O2 ratio of 1.5-4). The process requires 6 to 20 hours to reach completion with high selectivity.
Q: Does this method support a wide range of terminal alkyne substrates?
A: Yes, the method demonstrates broad substrate tolerance, successfully converting various terminal alkynes including phenylacetylene derivatives with electron-donating or withdrawing groups, as well as aliphatic alkynes like 1-hexyne and cyclohexylacetylene.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha,Beta-Unsaturated Alkynamide Supplier
As the demand for high-quality pharmaceutical intermediates continues to grow, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures access to cutting-edge synthetic technologies such as this oxidative carbonylation process. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory concept to industrial reality. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of alpha,beta-unsaturated alkynamide meets the highest international standards, providing you with the confidence needed to advance your drug development programs. We understand the critical nature of supply continuity and quality consistency in the pharmaceutical sector and have structured our operations to deliver on these promises reliably.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic benefits of switching to this catalytic method for your specific target molecule. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability to be your trusted partner in delivering complex chemical solutions efficiently and sustainably.
