Advanced Synthesis of 3-(Piperidin-4-yl)oxazolidin-2-one: A Safer Route for Commercial Scale-up
The pharmaceutical industry continuously seeks robust synthetic pathways for critical heterocyclic scaffolds, particularly those serving as key building blocks for cardiovascular and metabolic therapeutics. A recent technological breakthrough detailed in patent CN110483490B introduces a highly efficient and safe method for synthesizing 3-(piperidin-4-yl)oxazolidin-2-one and its salts, addressing long-standing challenges in process safety and cost-efficiency. This compound serves as a vital structural unit in the development of drugs treating thrombus, acute myocardial infarction, pulmonary embolism, and obesity, making its reliable production paramount for global supply chains. The disclosed innovation replaces hazardous traditional reagents with benign, commercially accessible starting materials, thereby streamlining the manufacturing process from laboratory bench to commercial reactor. By leveraging a strategic sequence of substitution, cyclization, and deprotection, this methodology not only enhances operator safety but also ensures high purity profiles essential for downstream API synthesis. For procurement leaders and R&D directors, understanding the nuances of this patent is crucial for optimizing sourcing strategies and mitigating supply risks associated with complex intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-(piperidin-4-yl)oxazolidin-2-one has been plagued by significant safety hazards and economic inefficiencies inherent in prior art methodologies. As illustrated in the reaction schemes below, traditional routes often rely on dangerous reagents such as sodium azide, which poses explosion risks, and pyrophoric palladium on charcoal combined with flammable hydrogen gas, necessitating expensive high-pressure equipment and rigorous safety protocols. Furthermore, certain legacy pathways utilize sodium cyanoborohydride under strong acidic conditions, creating a severe risk of releasing virulent hydrocyanic acid, a potent toxin that complicates waste treatment and endangers personnel. Other methods involve the use of dichloroethane, a suspected carcinogen, or require multi-step sequences with low overall yields that drive up the cost of goods sold. These factors collectively result in higher production costs, increased regulatory scrutiny, and fragile supply chains vulnerable to shutdowns due to safety incidents or equipment failures.
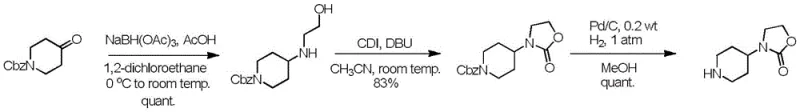
The Novel Approach
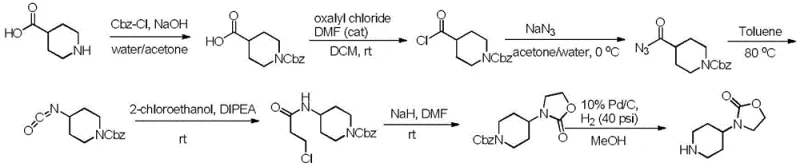
In stark contrast to these perilous conventional methods, the novel approach outlined in the patent utilizes 4-amino-1-tert-butoxycarbonylpiperidine and chloroformic acid-2-chloroethyl ester as primary raw materials, both of which are stable, non-toxic, and readily available on the global market. This new strategy circumvents the need for transition metal catalysts, high-pressure hydrogenation, and explosive azide chemistry, fundamentally altering the risk profile of the synthesis. The process operates under mild conditions, typically ranging from -20°C to 50°C, which reduces energy consumption and minimizes the thermal load on reactors, thereby enhancing operational stability. By employing a straightforward substitution followed by an intramolecular ring-closing reaction, the method achieves high yields with minimal byproduct formation, simplifying downstream purification. This shift towards benign chemistry not only lowers the barrier to entry for manufacturers but also aligns with modern green chemistry principles, offering a sustainable alternative that supports long-term supply chain resilience and cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Nucleophilic Substitution and Cyclization
The core of this innovative synthesis lies in a carefully orchestrated sequence of nucleophilic substitution and base-mediated cyclization, which constructs the oxazolidinone ring with high fidelity. Initially, the primary amine of 4-amino-1-tert-butoxycarbonylpiperidine acts as a nucleophile, attacking the carbonyl carbon of 2-chloroethyl chloroformate in the presence of a mild base such as potassium carbonate or triethylamine. This step forms a carbamate linkage while retaining the chloroethyl side chain, setting the stage for ring closure. The reaction is typically conducted in solvents like dichloromethane or tetrahydrofuran at controlled low temperatures to manage exotherms and prevent side reactions, ensuring the formation of the tert-butyl-4-(((2-chloroethoxy)acyl)amino)piperidine-1-carbonate intermediate with excellent selectivity. The preservation of the Boc protecting group during this stage is critical, as it prevents unwanted polymerization or side reactions at the piperidine nitrogen, thereby maintaining the structural integrity required for the subsequent cyclization step.
Following the substitution, the mechanism proceeds to an intramolecular cyclization driven by a stronger base, such as sodium hydride or potassium tert-butoxide, which deprotonates the carbamate nitrogen. This generates a reactive nucleophilic species that attacks the terminal carbon of the chloroethyl chain, displacing the chloride ion and closing the five-membered oxazolidinone ring. This ring-closing step is highly efficient and occurs under mild thermal conditions, avoiding the harsh environments that often lead to impurity generation in other synthetic routes. Finally, the tert-butyloxycarbonyl group is removed under acidic conditions, typically using hydrogen chloride in an organic solvent, to liberate the final 3-(piperidin-4-yl)oxazolidin-2-one salt. This deprotection is clean and quantitative, yielding the target molecule with high purity and minimizing the need for extensive chromatographic purification, which is a significant advantage for scaling up production to meet commercial demand.
How to Synthesize 3-(Piperidin-4-yl)oxazolidin-2-one Efficiently
To implement this synthesis effectively, manufacturers must adhere to precise stoichiometric ratios and temperature controls to maximize yield and safety. The process begins with the preparation of the carbamate intermediate, followed by the critical cyclization step which requires careful addition of the base to control exothermicity. Detailed standard operating procedures regarding solvent selection, quenching protocols, and isolation techniques are essential for reproducibility. For a comprehensive guide on the specific experimental conditions, reagent grades, and workup procedures validated in the patent examples, please refer to the standardized synthesis steps provided below.
- Perform a substitution reaction between 4-amino-1-tert-butoxycarbonylpiperidine and 2-chloroethyl chloroformate in the presence of a base to form the carbamate intermediate.
- Execute an intramolecular cyclization using a strong base in a suitable solvent to close the oxazolidinone ring, yielding the Boc-protected product.
- Remove the tert-butyloxycarbonyl protecting group under acidic conditions to obtain the final 3-(piperidin-4-yl)oxazolidin-2-one salt, followed by liberation to the free base.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthesis route offers transformative benefits for procurement managers and supply chain heads tasked with securing reliable sources of high-value intermediates. By eliminating the dependency on scarce or regulated reagents like sodium azide and palladium catalysts, the method drastically simplifies the sourcing landscape, allowing for greater flexibility in vendor selection and raw material procurement. The absence of heavy metals in the process removes the need for costly and time-consuming metal scavenging steps, which often bottleneck production timelines and inflate processing costs. Furthermore, the use of common organic solvents and standard reaction vessels means that existing manufacturing infrastructure can be utilized without significant capital expenditure on specialized high-pressure or explosion-proof equipment. This compatibility with standard facilities accelerates technology transfer and reduces the time-to-market for downstream drug products, providing a competitive edge in fast-paced therapeutic areas.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of inexpensive, commodity-grade starting materials that are produced in vast quantities globally, ensuring price stability and availability. By removing expensive transition metal catalysts and avoiding the complex waste disposal costs associated with toxic cyanides and azides, the overall cost of goods is significantly lowered. Additionally, the high yields reported in the patent examples indicate minimal material loss, further enhancing the economic efficiency of the process. The simplified post-treatment procedures reduce labor hours and solvent consumption, contributing to substantial operational savings that can be passed down the supply chain or reinvested in R&D initiatives.
- Enhanced Supply Chain Reliability: Supply chain continuity is greatly improved by the robustness of this synthetic route, which avoids reagents subject to strict regulatory controls or geopolitical supply constraints. The operational safety profile reduces the risk of unplanned plant shutdowns due to safety incidents, ensuring consistent delivery schedules for customers. Moreover, the scalability of the reaction conditions allows for seamless transition from pilot scale to multi-ton production, mitigating the risks associated with scale-up failures. This reliability is crucial for pharmaceutical companies managing just-in-time inventory systems and seeking to minimize buffer stocks of critical intermediates.
- Scalability and Environmental Compliance: The environmental footprint of this manufacturing process is markedly smaller than that of traditional methods, aligning with increasingly stringent global environmental regulations. The avoidance of carcinogenic solvents and toxic byproducts simplifies effluent treatment and reduces the burden on waste management systems. The process operates at near-ambient pressures and moderate temperatures, lowering energy consumption and greenhouse gas emissions associated with heating and cooling. These sustainability credentials not only facilitate regulatory approval but also enhance the corporate social responsibility profile of the manufacturer, appealing to environmentally conscious stakeholders and partners in the global pharmaceutical market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of 3-(piperidin-4-yl)oxazolidin-2-one. These insights are derived directly from the technical specifications and comparative data presented in the patent literature, aiming to clarify the operational benefits and safety improvements of this novel method. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their specific production needs.
Q: What are the primary safety advantages of this new synthesis route compared to prior art?
A: Unlike conventional methods that utilize explosive sodium azide, pyrophoric palladium on charcoal, or toxic sodium cyanoborohydride which releases hydrocyanic acid, this novel route employs stable, commercially available reagents like 4-amino-1-Boc-piperidine and 2-chloroethyl chloroformate, significantly reducing operational hazards and equipment investment requirements.
Q: How does this method impact the overall production cost for pharmaceutical intermediates?
A: The process eliminates the need for expensive transition metal catalysts and high-pressure hydrogenation equipment. By utilizing cheap starting materials and avoiding complex purification steps associated with heavy metal removal, the total manufacturing cost is substantially reduced while maintaining high reaction yields suitable for industrial production.
Q: Is this synthesis method scalable for commercial API manufacturing?
A: Yes, the method is specifically designed for industrial scalability. It features simple reaction conditions, mild temperature ranges, and robust operability without the risk of reaction runaways common in older methods, ensuring consistent supply continuity and easier regulatory compliance for large-scale pharmaceutical production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-(Piperidin-4-yl)oxazolidin-2-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of life-saving medications. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ state-of-the-art analytical instrumentation to verify identity and assay. Our dedication to process excellence means that we can reliably supply 3-(piperidin-4-yl)oxazolidin-2-one and related derivatives, supporting your pipeline with the consistency and quality required for regulatory filings and commercial launch.
We invite you to collaborate with us to optimize your supply chain and achieve your cost targets without compromising on quality. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. By partnering with us, you gain access to deep technical expertise and a flexible manufacturing network capable of adapting to your evolving needs. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced synthesis capabilities can drive value for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
