Scaling High-Purity Polyimide Monomers: A Technical Breakthrough in Copper-Catalyzed Synthesis
The landscape of high-performance polymer manufacturing is undergoing a significant transformation driven by the need for more efficient and cost-effective monomer synthesis. Patent CN113603662B introduces a groundbreaking method for preparing 4-(1-ketone-2-propargyl phenyl propane) phthalic anhydride, a critical precursor for thermosetting polyimides known for their exceptional heat resistance and mechanical strength. This technical disclosure represents a pivotal shift from traditional palladium-catalyzed systems to a more robust copper-catalyzed acylation protocol. For R&D Directors and Procurement Managers in the electronic materials sector, this development offers a pathway to drastically simplify supply chains while enhancing the purity profiles of final polymer products. The innovation lies not just in the chemical transformation but in the strategic selection of catalysts and solvents that enable high conversion rates under relatively mild conditions. By leveraging this proprietary knowledge, manufacturers can overcome historical bottlenecks associated with low-yielding acylation reactions, thereby securing a more reliable [Display & Optoelectronic Materials] supplier status in the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-(1-keto-2-propargyl) phthalic anhydride has been plagued by inefficiencies inherent to palladium-based catalytic systems. Literature reports, including prior art such as Chinese patent 201180024067.3, detail processes utilizing palladium acetate in toluene with triethylamine as a base, which consistently suffer from suboptimal performance metrics. The most glaring deficiency in these conventional methods is the remarkably low yield, often stagnating around 39.64%, which renders the process economically unviable for large-scale [cost reduction in electronic chemical manufacturing]. Furthermore, the reliance on palladium introduces significant supply chain vulnerabilities due to the high cost and geopolitical volatility associated with precious metal catalysts. The removal of residual palladium from the final product also necessitates additional purification steps, increasing both the operational complexity and the environmental footprint of the manufacturing process. These factors combined create a substantial barrier to entry for producers seeking to offer high-purity [OLED material] or polyimide precursors at competitive price points, forcing many to rely on inefficient batch processes that struggle to meet modern quality standards.
The Novel Approach
In stark contrast to the limitations of legacy technologies, the method disclosed in CN113603662B employs a sophisticated copper-catalyzed system that fundamentally redefines the efficiency of this acylation reaction. By utilizing catalysts such as copper triphenylphosphine bromide in conjunction with specific organic bases like tetramethyl ethylenediamine, the new protocol achieves reaction conversions that can reach approximately 90%. This dramatic improvement in yield is complemented by the use of more accessible and cost-effective solvents such as tetrahydrofuran, which facilitates better solubility and reaction kinetics compared to traditional toluene systems. The strategic optimization of molar ratios, specifically maintaining a balance between the acid chloride, phenylacetylene, and catalyst, ensures that the reaction proceeds with minimal formation of by-products. This approach not only enhances the economic feasibility of the synthesis but also aligns with the growing industry demand for sustainable and scalable chemical processes. For supply chain heads, this transition represents a move towards a more resilient [commercial scale-up of complex polymer additives] strategy, reducing dependency on critical raw materials while maximizing output efficiency.
Mechanistic Insights into Copper-Catalyzed Acylation
The core of this technological advancement lies in the intricate mechanistic pathway facilitated by the copper catalyst, which promotes the acylation of phenylacetylene with 1,2,4-trimellitic anhydride acyl chloride through a highly efficient cycle. Unlike palladium systems that may suffer from catalyst deactivation or slow oxidative addition steps, the copper species in this protocol appears to maintain high activity throughout the reaction duration, even at moderate temperatures ranging from 20°C to 90°C. The presence of the organic base plays a crucial role in scavenging the hydrochloric acid by-product, thereby driving the equilibrium towards the formation of the desired ketone product. This mechanistic efficiency is further evidenced by the ability of the system to tolerate slight variations in reagent ratios without significant loss in performance, provided the stoichiometry remains within the optimized window of 1:(1-1.1) for the main reactants. Such robustness is essential for industrial applications where precise control over every variable is challenging, ensuring consistent quality across different production batches.
Furthermore, the impurity control mechanism inherent in this copper-catalyzed route is a key factor contributing to the exceptional purity of the final product. Analytical data from the patent indicates a liquid phase content of 99.77%, suggesting that the reaction conditions effectively suppress the formation of side products that typically contaminate polyimide monomers. The specific choice of tetramethyl ethylenediamine as the base likely contributes to this selectivity by coordinating with the copper center and modulating its electronic properties to favor the desired acylation pathway over competing reactions. This high level of purity is critical for downstream applications in the electronics industry, where trace impurities can adversely affect the thermal stability and dielectric properties of the resulting polyimide films. By minimizing the need for extensive post-reaction purification, this method streamlines the production workflow and reduces the overall cost of goods sold.
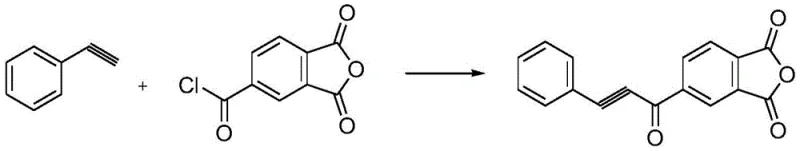
How to Synthesize 4-(1-Ketone-2-Propargyl Phenyl Propane) Phthalic Anhydride Efficiently
Implementing this synthesis route requires careful attention to the sequential addition of reagents and temperature control to maximize the benefits of the copper catalytic system. The process begins with the preparation of a homogeneous reaction mixture containing the acid chloride, solvent, and base, followed by the controlled introduction of the catalyst to initiate the acylation. Detailed standard operating procedures regarding specific addition rates, stirring speeds, and cooling protocols are essential to replicate the high yields reported in the patent literature. The following guide outlines the critical operational parameters necessary to achieve successful scale-up while maintaining the stringent quality standards required for electronic grade materials.
- Preparation of Reaction Mixture: Charge 1,2,4-trimellitic anhydride acyl chloride, organic solvent (tetrahydrofuran), and organic base (tetramethyl ethylenediamine) into a reactor, then add the copper catalyst under mechanical stirring and heat to 20-90°C.
- Acylation Reaction: Dissolve phenylacetylene in the organic solvent and slowly add it dropwise to the heated mixture, maintaining the temperature to ensure complete conversion of the acid chloride.
- Workup and Purification: Upon completion, cool the reaction liquid to 10°C, filter the precipitate, wash the filter cake with tetrahydrofuran and water, and dry in a vacuum oven to obtain the high-purity white solid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this copper-catalyzed methodology offers profound strategic advantages that extend beyond simple chemical yield improvements. The elimination of palladium from the catalyst system immediately addresses one of the most significant cost drivers in fine chemical synthesis, as copper salts are substantially more abundant and affordable than precious metal alternatives. This shift allows for a significant reduction in raw material costs without compromising the quality of the final product, enabling manufacturers to offer more competitive pricing structures to their downstream clients. Additionally, the simplified workup procedure, which involves straightforward filtration and washing steps, reduces the operational time and labor required for each batch, further enhancing the overall economic efficiency of the manufacturing process.
- Cost Reduction in Manufacturing: The transition from palladium to copper catalysts fundamentally alters the cost structure of producing 4-(1-ketone-2-propargyl phenyl propane) phthalic anhydride by removing the need for expensive precious metal recovery systems. Since the catalyst is cheaper and the reaction yield is significantly higher, the cost per kilogram of the final product is drastically lowered, allowing for better margin management in volatile markets. Moreover, the use of common solvents like tetrahydrofuran instead of specialized aromatic solvents reduces procurement complexity and storage costs. This economic efficiency is compounded by the high conversion rate, which minimizes waste generation and maximizes the utilization of starting materials, leading to substantial cost savings in the overall production lifecycle.
- Enhanced Supply Chain Reliability: Relying on copper-based catalysts mitigates the supply chain risks associated with the fluctuating availability and pricing of palladium, which is often subject to geopolitical tensions and mining constraints. The raw materials required for this process, including phenylacetylene and trimellitic anhydride acyl chloride, are readily available from multiple global suppliers, ensuring a continuous and stable flow of inputs for production. This diversification of the supply base enhances the resilience of the manufacturing operation against disruptions, guaranteeing consistent delivery schedules for customers in the fast-paced electronics industry. Furthermore, the robustness of the reaction conditions means that production can be maintained even if minor variations in raw material quality occur, reducing the risk of batch failures.
- Scalability and Environmental Compliance: The simplicity of the reaction protocol, which avoids extreme temperatures and pressures, makes it highly suitable for scaling from laboratory benchtop to multi-ton commercial production facilities. The reduced generation of hazardous waste, particularly heavy metal residues, simplifies the environmental compliance burden and lowers the costs associated with waste treatment and disposal. This alignment with green chemistry principles not only improves the corporate sustainability profile but also facilitates easier regulatory approvals in jurisdictions with strict environmental standards. The ability to scale efficiently ensures that supply can meet growing demand for high-performance polyimides in emerging applications such as flexible displays and advanced packaging.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis method, providing clarity for stakeholders evaluating its potential for integration into their supply chains. These insights are derived directly from the experimental data and technical disclosures within the patent, ensuring accuracy and relevance for decision-makers. Understanding these details is crucial for assessing the feasibility of adopting this technology for large-scale manufacturing of high-purity intermediates.
Q: How does the copper-catalyzed method improve yield compared to palladium systems?
A: The novel copper-catalyzed protocol described in CN113603662B achieves reaction conversions reaching approximately 90%, significantly outperforming conventional palladium acetate methods which historically report yields as low as 39.64%. This improvement is attributed to the specific ligand environment of the copper catalyst which facilitates more efficient acylation kinetics.
Q: What is the purity profile of the synthesized polyimide monomer?
A: Analytical data from the patent indicates a liquid phase content of 99.77%, demonstrating exceptional control over impurity formation. This high purity is critical for downstream polyimide applications where trace contaminants can degrade thermal and mechanical properties.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method utilizes easily obtained raw materials and simple operation steps such as filtration and vacuum drying. The elimination of expensive transition metals and the robustness of the reaction conditions make it highly amenable to commercial scale-up of complex electronic chemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-(1-Ketone-2-Propargyl Phenyl Propane) Phthalic Anhydride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating innovative patent technologies into reliable commercial realities for our global partners. Our team of expert chemists and process engineers possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the copper-catalyzed route are fully realized in industrial settings. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ state-of-the-art analytical techniques to verify every batch against the highest industry standards. Our capability to manage complex synthesis routes allows us to provide a stable supply of high-performance monomers essential for the next generation of electronic materials.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through the adoption of this advanced synthesis technology. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete technical evidence. By partnering with us, you gain access to a reliable source of high-quality intermediates that can drive innovation and efficiency in your product development pipeline.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
