Advanced Manufacturing of High-Purity Radiopharmaceutical Precursor Intermediates via Nanofiltration
Advanced Manufacturing of High-Purity Radiopharmaceutical Precursor Intermediates via Nanofiltration
The pharmaceutical industry is currently witnessing a paradigm shift in the synthesis of complex radiopharmaceutical precursors, driven by the urgent need for safer, more scalable, and environmentally sustainable manufacturing processes. Patent CN115611779A introduces a groundbreaking preparation method for a critical intermediate of a radiopharmaceutical precursor, addressing long-standing bottlenecks in purity and process safety that have historically plagued this sector. This innovation replaces traditional, hazardous isolation steps with a continuous nanofiltration strategy, ensuring that the unstable diazonium intermediates are never handled as dry solids, thereby mitigating significant operational risks. For global procurement leaders seeking a reliable radiopharmaceutical intermediate supplier, this technology represents a substantial leap forward in supply chain security and product consistency. By integrating advanced membrane separation technology directly into the reaction workflow, the process achieves exceptional purity levels exceeding 96% without the need for resource-intensive preparative chromatography. This report analyzes the technical merits and commercial implications of this novel route, providing R&D and supply chain executives with the insights needed to evaluate its adoption for large-scale production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of radiopharmaceutical intermediates involving diazonium chemistry has been fraught with significant safety hazards and purification challenges that limit commercial viability. Traditional protocols typically require the isolation of the diazonium salt intermediate via lyophilization, a process that creates a dry, highly unstable solid prone to decomposition or even explosive hazards during transfer and storage. Furthermore, existing methods described in prior art often rely on high concentrations of trifluoroacetic acid for deprotection, generating substantial volumes of acidic wastewater that impose heavy environmental compliance burdens and disposal costs. The crude products obtained from these conventional routes are frequently contaminated with large amounts of inorganic salts and reaction solvents, necessitating multiple rounds of preparative HPLC purification to achieve acceptable purity standards. This reliance on chromatography not only drastically increases production time and operational expenses but also limits the ability to scale up manufacturing to meet industrial demand. Consequently, the overall yield is often compromised, and the supply continuity for these critical pharmaceutical intermediates remains vulnerable to process inefficiencies and safety shutdowns.
The Novel Approach
In stark contrast to these legacy methods, the innovative process disclosed in patent CN115611779A utilizes a continuous aqueous workflow that fundamentally alters the risk profile and economic model of production. The core breakthrough lies in the strategic application of nanofiltration immediately following the diazotization reaction, allowing the unstable diazonium salt to remain in solution rather than being isolated as a hazardous solid. 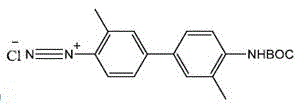 This approach effectively eliminates the safety risks associated with transferring dry diazonium compounds while simultaneously removing inorganic salts and small molecule impurities through membrane separation. By avoiding the freeze-drying of the intermediate Boc-EB and instead subjecting the reaction mixture to nanofiltration, the process removes excess water-soluble raw materials and by-products without the need for column chromatography. The subsequent deprotection step is conducted directly in the aqueous filtrate using mild acid conditions, which simplifies the workflow and minimizes the generation of hazardous waste. This streamlined methodology not only enhances operator safety but also significantly reduces the environmental footprint, making it an ideal candidate for cost reduction in radiopharmaceutical manufacturing while ensuring robust quality control.
This approach effectively eliminates the safety risks associated with transferring dry diazonium compounds while simultaneously removing inorganic salts and small molecule impurities through membrane separation. By avoiding the freeze-drying of the intermediate Boc-EB and instead subjecting the reaction mixture to nanofiltration, the process removes excess water-soluble raw materials and by-products without the need for column chromatography. The subsequent deprotection step is conducted directly in the aqueous filtrate using mild acid conditions, which simplifies the workflow and minimizes the generation of hazardous waste. This streamlined methodology not only enhances operator safety but also significantly reduces the environmental footprint, making it an ideal candidate for cost reduction in radiopharmaceutical manufacturing while ensuring robust quality control.
Mechanistic Insights into Nanofiltration-Assisted Diazotization and Coupling
The chemical elegance of this synthesis lies in the precise control of molecular weight cut-offs during the nanofiltration stages, which serves as a physical barrier to impurities while retaining the desired product. The process begins with the diazotization of N-Boc-2-toluidine using hydrochloric acid and sodium nitrite at controlled low temperatures between 0°C and 15°C to form the diazonium salt intermediate. Instead of quenching or isolating this species, the reaction mixture is passed through a nanofiltration membrane with a molecular weight cut-off of 100 to 300 Daltons. This specific range is critical as it allows small inorganic ions like chloride and nitrite to pass through into the permeate, while the larger organic diazonium cation is retained in the retentate. This in-situ desalting step ensures that the subsequent coupling reaction proceeds with minimal interference from inorganic salts, which can otherwise catalyze decomposition pathways or complicate downstream purification. The retention of the diazonium species in an aqueous environment stabilizes the reactive intermediate through solvation effects, preventing the thermal runaway scenarios often seen in dry states.
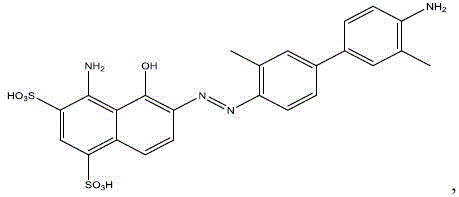
Following the initial filtration, the purified diazonium solution is coupled with 1-amino-8-naphthol-2,4-disulfonic acid monosodium salt to form the Boc-protected azo dye intermediate, known as Boc-EB. 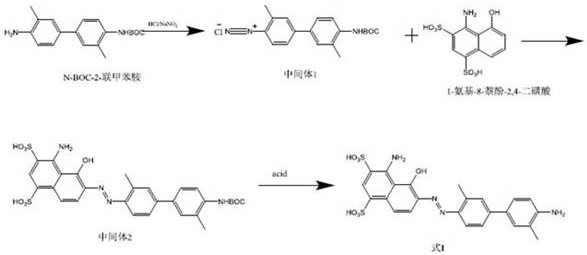 The reaction mixture undergoes a second nanofiltration cycle, which is instrumental in removing unreacted naphthol derivatives and other small molecule by-products generated during the coupling phase. A quenching agent, such as dithiothreitol (DTT), is introduced to neutralize any residual oxidative species, further safeguarding the integrity of the azo linkage. The final deprotection is achieved by adjusting the pH of the filtrate to below 2 using dilute hydrochloric acid, which cleaves the Boc group under mild conditions without requiring harsh organic solvents. This entire sequence maintains the product in a homogeneous aqueous phase until the final lyophilization step, ensuring that the high-purity radiopharmaceutical precursor is obtained with a purity exceeding 96% and a reproducible yield. The mechanistic efficiency of this route demonstrates how physical separation technologies can be seamlessly integrated with organic synthesis to overcome traditional purification bottlenecks.
The reaction mixture undergoes a second nanofiltration cycle, which is instrumental in removing unreacted naphthol derivatives and other small molecule by-products generated during the coupling phase. A quenching agent, such as dithiothreitol (DTT), is introduced to neutralize any residual oxidative species, further safeguarding the integrity of the azo linkage. The final deprotection is achieved by adjusting the pH of the filtrate to below 2 using dilute hydrochloric acid, which cleaves the Boc group under mild conditions without requiring harsh organic solvents. This entire sequence maintains the product in a homogeneous aqueous phase until the final lyophilization step, ensuring that the high-purity radiopharmaceutical precursor is obtained with a purity exceeding 96% and a reproducible yield. The mechanistic efficiency of this route demonstrates how physical separation technologies can be seamlessly integrated with organic synthesis to overcome traditional purification bottlenecks.
How to Synthesize Radiopharmaceutical Precursor Intermediate Efficiently
The implementation of this synthesis route requires precise control over reaction parameters and membrane specifications to ensure optimal performance and product quality. The process is designed to be operationally simple yet chemically rigorous, leveraging the selectivity of nanofiltration membranes to replace complex chromatographic separations. Detailed standard operating procedures regarding solvent ratios, temperature gradients, and membrane pressure settings are critical for maintaining the integrity of the diazonium intermediate throughout the workflow. Manufacturers must adhere to strict inert atmosphere conditions, typically using nitrogen or argon, to prevent oxidative degradation of the sensitive intermediates during the coupling and filtration stages. The following guide outlines the critical operational phases necessary to replicate the high yields and purity profiles reported in the patent data, serving as a foundational reference for process engineers aiming to adopt this technology.
- Perform diazotization on N-Boc-2-toluidine with hydrochloric acid and sodium nitrite at 0-15°C, followed immediately by nanofiltration to remove inorganic salts.
- Execute coupling reaction with 1-amino-8-naphthol-2,4-disulfonic acid monosodium salt in aqueous solution, utilizing nanofiltration again to isolate the Boc-EB intermediate.
- Conduct deprotection using dilute acid in the aqueous filtrate, followed by direct lyophilization to obtain the final high-purity radiopharmaceutical precursor intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this nanofiltration-based synthesis route offers transformative advantages that extend far beyond simple technical improvements. The elimination of preparative HPLC purification steps represents a massive reduction in operational expenditure, as chromatography is notoriously expensive due to the cost of stationary phases, solvents, and the extensive labor required for fraction collection and analysis. By replacing this with a continuous membrane filtration process, the manufacturing timeline is drastically compressed, allowing for faster batch turnover and improved responsiveness to market demand fluctuations. Furthermore, the shift towards an aqueous-based system significantly reduces the consumption of volatile organic compounds (VOCs), lowering both the cost of solvent procurement and the expenses associated with hazardous waste disposal and environmental compliance. These efficiencies collectively contribute to a more resilient and cost-effective supply chain for commercial scale-up of complex pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The removal of chromatographic purification and the reduction in organic solvent usage directly lower the variable costs associated with each production batch. The process avoids the use of high-cost reagents like concentrated trifluoroacetic acid in favor of dilute hydrochloric acid, which is significantly cheaper and easier to handle on an industrial scale. Additionally, the ability to recycle aqueous streams and the reduced need for energy-intensive freeze-drying of intermediates further enhance the economic viability of the route. These factors combine to create a manufacturing process that is inherently leaner and less susceptible to raw material price volatility, ensuring stable pricing for long-term supply agreements.
- Enhanced Supply Chain Reliability: Safety is a critical component of supply chain reliability, and by eliminating the isolation of unstable dry diazonium salts, the risk of production halts due to safety incidents is substantially mitigated. The continuous nature of the process reduces the number of unit operations and transfer steps, minimizing the potential for human error or cross-contamination between batches. This streamlined workflow ensures consistent output quality and reduces the lead time required to validate and release batches for downstream radiolabeling. For partners seeking reducing lead time for high-purity radiopharmaceutical intermediates, this technology provides a robust framework for maintaining uninterrupted supply even during periods of high demand.
- Scalability and Environmental Compliance: The use of water as the primary solvent aligns perfectly with modern green chemistry initiatives and regulatory pressures to reduce the environmental impact of pharmaceutical manufacturing. The process generates significantly less hazardous waste compared to traditional methods, simplifying the permitting process for new manufacturing facilities and reducing the liability associated with waste treatment. The nanofiltration equipment used in this process is readily scalable from pilot to commercial production volumes without the need for complex re-engineering of the chemical pathway. This scalability ensures that the supply chain can grow in tandem with the clinical and commercial success of the final radiopharmaceutical product, providing a future-proof solution for long-term partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this novel synthesis method. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing factual clarity for decision-makers evaluating this technology. Understanding the nuances of membrane selection, safety protocols, and purity outcomes is essential for integrating this route into existing manufacturing portfolios. The responses below highlight the practical advantages and operational considerations that distinguish this method from conventional approaches.
Q: How does nanofiltration improve the safety of diazonium salt handling?
A: By maintaining the diazonium intermediate in an aqueous solution and processing it directly through nanofiltration, the need to isolate the unstable dry solid is eliminated, significantly reducing explosion risks during transfer.
Q: Does this method eliminate the need for preparative HPLC purification?
A: Yes, the dual nanofiltration steps effectively remove small molecule impurities and inorganic salts, allowing the final product to reach over 96% purity via direct lyophilization without costly chromatography.
Q: What are the environmental benefits of this aqueous-based synthesis route?
A: The process minimizes organic solvent usage by relying primarily on water, avoids high concentrations of trifluoroacetic acid, and reduces waste generation, aligning with green chemistry principles for sustainable manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Radiopharmaceutical Precursor Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising synthetic routes like the one described in CN115611779A can be successfully implemented at an industrial level. We maintain stringent purity specifications and operate rigorous QC labs equipped to handle the analytical demands of radiopharmaceutical intermediates, guaranteeing that every batch meets the exacting standards required for clinical applications. Our commitment to quality and safety makes us the ideal partner for organizations looking to secure a stable supply of critical precursors without compromising on regulatory compliance or product integrity.
We invite you to engage with our technical procurement team to discuss how this advanced nanofiltration technology can be tailored to your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the potential economic benefits and efficiency gains applicable to your production volume. We encourage you to contact us to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on empirical evidence and expert evaluation. Let us help you optimize your supply chain with cutting-edge chemistry that delivers both performance and value.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
