Optimizing Gabapentin Intermediate Production: A Technical Breakthrough for Commercial Scale-Up
The pharmaceutical landscape for anticonvulsant medications continues to evolve, with Gabapentin remaining a cornerstone therapy for epilepsy and neuropathic pain. Central to the efficient manufacturing of this high-volume Active Pharmaceutical Ingredient (API) is the availability of its critical precursor, 1-cyano-cyclohexyl acetonitrile. Recent advancements detailed in patent CN116253665A introduce a transformative preparation method that addresses long-standing inefficiencies in intermediate synthesis. This innovation leverages a sophisticated one-pot tandem reaction strategy, seamlessly integrating condensation and cyanation steps within a biphasic catalytic system. By fundamentally reengineering the synthetic pathway, this technology offers a robust solution for producing high-purity intermediates essential for downstream API manufacturing. The structural integrity of the final Gabapentin molecule relies heavily on the quality of this nitrile intermediate, making process optimization a critical priority for R&D teams aiming to enhance overall drug substance quality. 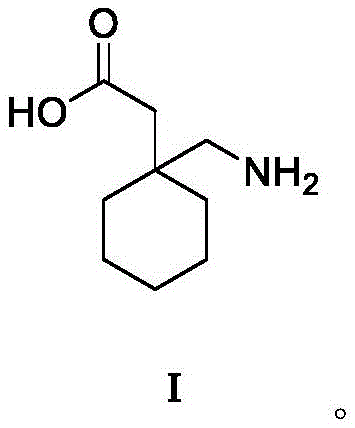
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1-cyano-cyclohexyl acetonitrile has relied heavily on multi-step processes or enzymatic routes that introduce significant operational complexity and cost burdens. Traditional chemical methods often require the isolation and purification of the Knoevenagel condensation product before proceeding to the cyanation step, leading to substantial material loss and increased solvent consumption. Furthermore, enzymatic approaches, while selective, are frequently constrained by the high cost of biocatalysts, sensitivity to reaction conditions, and difficulties in scaling up due to enzyme stability issues. These legacy processes often result in inconsistent impurity profiles, particularly regarding residual cyclohexanone, which can complicate subsequent purification stages and compromise the quality of the final API. The cumulative effect of these inefficiencies is a supply chain vulnerable to cost fluctuations and production delays, necessitating a more resilient manufacturing paradigm.
The Novel Approach
The methodology disclosed in CN116253665A represents a paradigm shift by employing a direct one-pot synthesis that eliminates intermediate isolation entirely. This approach utilizes a dual-phase system where cyclohexanone and alkyl cyanoacetate react in an organic phase buffered by weak alkali salts and organic acids, creating an optimal environment for the initial condensation. Without workup, the system is directly treated with an aqueous phase containing strong alkali, cyanide, and a phase transfer catalyst to drive the cyanation to completion. This seamless integration not only streamlines the workflow but also significantly enhances the overall yield, consistently achieving levels above 90%. The strategic use of phase transfer catalysis ensures efficient mass transfer between the immiscible layers, facilitating rapid reaction kinetics while maintaining strict control over side reactions. 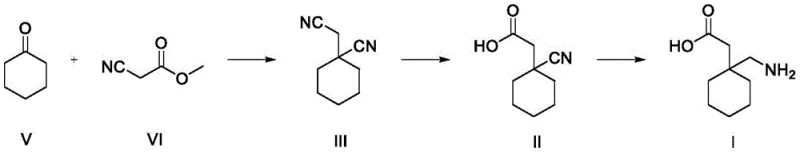
Mechanistic Insights into Biphasic Phase Transfer Catalysis
The core of this technological breakthrough lies in the precise orchestration of the biphasic reaction environment. In the organic phase, the presence of weak alkali salts such as ammonium acetate alongside organic acids like citric acid creates a buffered medium that promotes the formation of the alkylidene cyanoacetate intermediate without triggering premature decomposition or polymerization. This careful modulation of pH and reactivity is crucial for minimizing the formation of oligomeric byproducts that typically plague high-temperature condensation reactions. Once the organic phase is primed, the introduction of the aqueous phase introduces the cyanide nucleophile under the guidance of quaternary ammonium phase transfer catalysts. These catalysts shuttle the cyanide anions into the organic layer, where they attack the activated double bond with high regioselectivity. This mechanism ensures that the cyanation occurs efficiently at the desired position, preventing the formation of structural isomers that are difficult to separate.
Impurity control is another critical aspect where this mechanism excels, particularly concerning the reduction of residual starting materials. The rigorous reaction conditions, combined with the specific choice of solvents like toluene which are immiscible with water, allow for effective partitioning of inorganic salts and polar impurities into the aqueous waste stream. The patent data indicates that this method can reduce cyclohexanone content to less than 0.02% and limit other single impurities to below 0.03%, achieving a purity profile of 99.9% or higher. Such stringent control is achieved without the need for extensive chromatographic purification, relying instead on optimized crystallization protocols using alcohol-water mixtures. For R&D directors, this level of purity assurance translates to a more predictable and stable downstream process for the final API synthesis, reducing the risk of batch failures and regulatory queries related to impurity thresholds.
How to Synthesize 1-Cyano-Cyclohexyl Acetonitrile Efficiently
Implementing this synthesis route requires careful attention to the preparation of the two distinct reaction phases and their subsequent combination under controlled thermal conditions. The process begins with the formulation of the aqueous catalytic system, followed by the independent generation of the organic condensation mixture, ensuring that each phase reaches its optimal state before mixing. Detailed operational parameters, including specific molar ratios of catalysts to substrates and precise temperature ramps, are critical for replicating the high yields reported in the patent literature. Operators must adhere to strict safety protocols when handling cyanide sources, utilizing the closed-system advantages of this one-pot design to minimize exposure risks.
- Prepare an aqueous phase by dissolving strong alkali, cyanide, and a phase transfer catalyst in water to establish the nucleophilic environment.
- React cyclohexanone with alkyl cyanoacetate in an organic solvent containing weak alkali salt and organic acid to form the initial condensation product without isolation.
- Combine the organic and aqueous phases for continuous reaction, followed by separation, solvent removal, and recrystallization to achieve >99.9% purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthesis method offers profound advantages that directly address the pain points of procurement managers and supply chain leaders. By consolidating two reaction steps into a single vessel operation, the process drastically reduces the requirement for intermediate storage, handling equipment, and associated labor costs. The elimination of isolation steps between condensation and cyanation means fewer unit operations, which translates to lower capital expenditure for manufacturing facilities and reduced energy consumption per kilogram of product. Furthermore, the use of commodity chemicals such as sodium hydroxide, sodium cyanide, and toluene ensures that raw material sourcing remains stable and cost-effective, shielding the supply chain from the volatility often associated with specialized reagents or proprietary enzymes.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the intensification of the reaction sequence and the high atom economy of the one-pot design. By removing the need for expensive biocatalysts and reducing solvent exchange operations, the overall cost of goods sold is significantly lowered without compromising quality. The high yield efficiency means that less raw material is wasted, maximizing the output from every batch and improving the return on investment for production runs. Additionally, the simplified workup procedure reduces the volume of waste generated, leading to substantial savings in waste disposal and environmental compliance costs.
- Enhanced Supply Chain Reliability: The robustness of this chemical route enhances supply chain continuity by reducing dependency on complex biological supply chains that can be susceptible to disruption. The reliance on stable, shelf-stable chemical reagents ensures that production can be scheduled with greater certainty and flexibility, allowing manufacturers to respond more agilely to market demand fluctuations. The scalability of the process from laboratory to industrial scale is straightforward, as it utilizes standard reactor configurations and does not require specialized fermentation infrastructure. This ease of scale-up facilitates faster technology transfer and quicker ramp-up times for new production lines, ensuring consistent availability of the intermediate for downstream API manufacturers.
- Scalability and Environmental Compliance: Environmental sustainability is increasingly a key metric for supply chain decisions, and this process aligns well with green chemistry principles through waste minimization. The ability to recycle aqueous phases and the reduced solvent usage contribute to a lower environmental footprint, helping companies meet their corporate sustainability goals. The high purity of the crude product reduces the need for energy-intensive recrystallization cycles, further lowering the carbon intensity of the manufacturing process. Compliance with environmental regulations is simplified due to the reduced generation of hazardous waste streams, making this method a preferred choice for facilities operating under strict environmental oversight.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology. These insights are derived directly from the experimental data and process descriptions provided in the patent documentation, offering clarity on performance metrics and operational feasibility. Understanding these details is essential for stakeholders evaluating the potential integration of this method into their existing manufacturing portfolios.
Q: How does this one-pot method improve impurity profiles compared to traditional enzymatic routes?
A: The novel chemical approach utilizes a controlled biphasic system that minimizes side reactions, achieving cyclohexanone residuals below 0.02% and single impurities under 0.03%, significantly outperforming standard enzymatic hydrolysis which often struggles with residual substrate removal.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the method eliminates complex isolation steps between the condensation and cyanation stages, using common solvents like toluene and water, which simplifies equipment requirements and enhances operational safety for metric-ton scale production.
Q: What are the key cost drivers reduced in this synthesis pathway?
A: By avoiding expensive biocatalysts and reducing solvent exchange operations, the process lowers raw material consumption and waste treatment costs, while the high yield (>90%) maximizes throughput efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-cyano-cyclohexyl acetonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global pharmaceutical supply chain. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative processes like the one described in CN116253665A can be successfully translated into industrial reality. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify every batch. Our capability to handle complex chemistries, including hazardous cyanation reactions under safe and controlled conditions, positions us as a strategic partner for companies seeking to optimize their Gabapentin supply chain.
We invite you to collaborate with us to explore how this optimized synthesis route can benefit your specific production needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and quality standards. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete technical evidence. By partnering with us, you gain access to a supply chain that prioritizes efficiency, quality, and reliability, ensuring your downstream operations run smoothly without interruption.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
