Advanced Safe Synthesis of N-Methyl-5-Hexene-1-Amine for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust and safe pathways for constructing complex amine scaffolds, particularly those serving as critical building blocks for macrocyclic drugs such as hepatitis C inhibitors. A pivotal advancement in this domain is detailed in patent CN108003037B, which discloses a revolutionary synthesis process for N-methyl-5-hexene-1-amine. This intermediate is highly valued for its dual functionality, featuring a reactive secondary amine and a terminal alkene group capable of diverse downstream transformations including alkylation, acylation, and cyclization. Unlike legacy methods that rely on hazardous reagents, this novel approach utilizes a direct nucleophilic substitution between 6-bromo-1-hexene and an excess of methylamine solution. By operating under mild thermal conditions of 40-50°C and employing specific stabilizers, the process achieves high purity levels exceeding 95% while fundamentally altering the safety profile of the manufacturing workflow. For R&D directors and procurement specialists, this represents a significant leap forward in securing a reliable supply chain for high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of unsaturated amines like N-methyl-5-hexene-1-amine has been fraught with significant safety and operational challenges, primarily due to the reliance on strong bases such as sodium hydride (NaH). As illustrated in prior art literature (e.g., Organic and Biomolecular Chemistry, 2006), the conventional route typically involves reacting the alkyl halide with methylamine in the presence of NaH in polar aprotic solvents like DMF. The fundamental flaw in this methodology is the generation of hydrogen gas (H2) as a stoichiometric byproduct of the deprotonation step. Hydrogen possesses a notoriously wide explosive limit range, creating a severe potential safety hazard, especially when scaling up to industrial volumes where gas accumulation can lead to catastrophic events. Furthermore, the use of NaH requires stringent anhydrous conditions and careful handling of pyrophoric solids, which complicates the operational protocol and increases the cost of goods sold due to specialized safety infrastructure requirements.
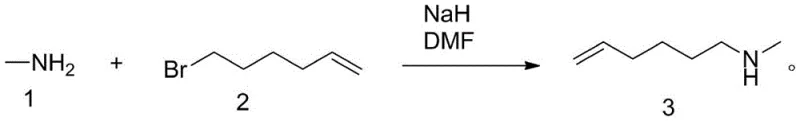
The Novel Approach
In stark contrast to the hazardous legacy protocols, the process disclosed in patent CN108003037B introduces a streamlined, green chemistry approach that entirely circumvents the use of reactive metal hydrides. The core innovation lies in utilizing a large molar excess of methylamine solution—available in various solvents such as methanol, ethanol, water, or acetonitrile—as both the nucleophile and the base medium. By heating the mixture of 6-bromo-1-hexene and methylamine solution to a moderate 40-50°C, the reaction proceeds efficiently via an SN2 mechanism without generating any gaseous byproducts. This elimination of hydrogen gas generation removes the explosion risk entirely, allowing for safer operation in standard stainless steel reactors. Additionally, the process incorporates a stabilizer, such as p-methoxyphenol, to prevent the polymerization of the terminal alkene group during the heating phase, ensuring the structural integrity of the final product. The post-reaction workup is equally elegant, involving simple neutralization and vacuum distillation to recover excess methylamine, thereby simplifying the purification train and enhancing overall process economics.
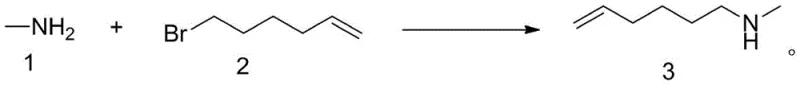
Mechanistic Insights into Nucleophilic Substitution and Stabilization
The chemical transformation at the heart of this process is a classic bimolecular nucleophilic substitution (SN2), yet its execution is optimized for industrial viability through careful control of reaction parameters. The primary substrate, 6-bromo-1-hexene, presents a primary alkyl halide moiety which is highly susceptible to backside attack by the lone pair of electrons on the nitrogen atom of methylamine. The use of a significant molar excess of methylamine (ranging from 2 to 15 equivalents in the patent examples) serves a dual kinetic and thermodynamic purpose: kinetically, it increases the frequency of effective collisions between the nucleophile and the electrophile, driving the reaction rate; thermodynamically, it pushes the equilibrium towards the formation of the secondary amine product while suppressing the formation of tertiary amine byproducts or quaternary ammonium salts. The choice of solvent plays a critical role as well; polar protic solvents like methanol or ethanol can solvate the transition state effectively, while the presence of water in methylamine water solutions offers an environmentally benign alternative without compromising the reaction efficiency significantly.
Beyond the primary substitution mechanism, the preservation of the terminal alkene functionality is paramount, as this group is essential for subsequent downstream coupling reactions in drug synthesis. Alkenes are prone to radical-induced polymerization, especially under thermal stress. To mitigate this, the patent mandates the addition of radical scavengers or stabilizers such as p-methoxyphenol, 1,2-diphenol, or 2,6-dimethyl-4-tert-butylphenol. These additives function by intercepting free radicals that might initiate chain polymerization of the vinyl group, thereby maintaining the monomeric state of the product. Following the reaction, the addition of sodium hydroxide serves to neutralize any acidic byproducts (such as HBr formed from trace hydrolysis or side reactions) and ensures the amine remains in its free base form, which is volatile enough to be purified via reduced pressure distillation. This meticulous attention to stabilization and pH control results in a colorless liquid product with purity consistently above 95%, suitable for direct use in sensitive pharmaceutical applications.
How to Synthesize N-Methyl-5-Hexene-1-Amine Efficiently
Implementing this synthesis route in a pilot or production plant requires adherence to specific operational parameters to maximize yield and safety. The process is designed to be operationally simple, avoiding the need for cryogenic conditions or inert atmosphere gloveboxes typically associated with organometallic chemistry. The key to success lies in the precise management of the methylamine excess and the temperature profile during the reaction and distillation phases. Operators must ensure that the recovery of excess methylamine is conducted under negative pressure to prevent leakage and to facilitate recycling, aligning with green chemistry principles. The following guide outlines the standardized steps derived from the patent examples to ensure reproducible high-quality output.
- Prepare a reaction vessel with a methylamine solution (e.g., in methanol) ensuring a molar excess relative to the alkyl halide substrate.
- Add 6-bromo-1-hexene to the mixture and heat to 40-50°C for 2-3 hours to facilitate the substitution reaction.
- Cool the mixture, add a stabilizer and sodium hydroxide, then recover excess methylamine and distill under reduced pressure to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis method offers profound strategic advantages beyond mere technical feasibility. The elimination of hazardous reagents like sodium hydride translates directly into a reduction in regulatory compliance burdens and insurance costs associated with handling pyrophoric materials. Furthermore, the ability to recycle the excess methylamine solvent system creates a closed-loop material flow that drastically reduces raw material consumption per kilogram of product. This efficiency gain is critical in a market where margin compression is a constant pressure, allowing manufacturers to offer more competitive pricing without sacrificing quality. The simplified workup, which eschews complex organic extractions in favor of direct distillation, also shortens the batch cycle time, thereby increasing the throughput capacity of existing manufacturing assets and reducing the lead time for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the substitution of expensive, hazardous reagents with commodity chemicals. Sodium hydride is not only costly but requires special disposal protocols for its quenching residues; replacing it with aqueous or alcoholic methylamine solutions significantly lowers the direct material cost. Moreover, the process avoids the use of large volumes of extraction solvents like dichloromethane or ethyl acetate, which are subject to volatile pricing and strict environmental regulations regarding VOC emissions. By relying on vacuum distillation for purification, the process minimizes solvent waste generation, leading to substantial cost savings in waste treatment and disposal. The recyclability of the methylamine solution further amplifies these savings, as the unreacted starting material is not lost but rather reintroduced into the process, optimizing the atom economy of the synthesis.
- Enhanced Supply Chain Reliability: From a sourcing perspective, the raw materials for this synthesis—6-bromo-1-hexene and methylamine—are widely available commodity chemicals produced by multiple global suppliers. This diversification of the supply base mitigates the risk of single-source bottlenecks that often plague specialized reagent markets. The robustness of the reaction conditions (40-50°C) means that the process can be executed in standard glass-lined or stainless steel reactors found in most multipurpose chemical plants, removing the need for specialized equipment that might have long lead times for fabrication. Consequently, manufacturers can scale up production rapidly to meet surging demand for hepatitis C inhibitors or other downstream APIs, ensuring a continuous and reliable supply of this critical intermediate to their customers.
- Scalability and Environmental Compliance: The green chemistry credentials of this process are a major asset for companies aiming to meet increasingly stringent ESG (Environmental, Social, and Governance) targets. By eliminating the generation of explosive hydrogen gas, the process inherently reduces the risk of major industrial accidents, aligning with 'Responsible Care' initiatives. The absence of heavy metal catalysts or toxic organometallic reagents simplifies the impurity profile, making it easier to meet the rigorous residual solvent and heavy metal limits imposed by pharmacopeial standards (e.g., ICH Q3D). The simplified downstream processing reduces the energy footprint associated with solvent recovery and waste incineration. This environmental efficiency not only safeguards the company's social license to operate but also future-proofs the supply chain against tightening environmental regulations that could otherwise render older, dirtier processes obsolete.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of N-methyl-5-hexene-1-amine. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a transparent view of the process capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this intermediate into their own synthetic routes for complex drug molecules.
Q: Why is the new synthesis method safer than the traditional NaH route?
A: The traditional method utilizes sodium hydride (NaH) which generates explosive hydrogen gas as a byproduct. The novel process described in patent CN108003037B uses a direct nucleophilic substitution with excess methylamine, completely eliminating the generation of hazardous hydrogen gas.
Q: What is the expected purity and yield of this process?
A: According to the patent data, the process consistently achieves a product purity of greater than 95% and yields ranging from 60% to 67%, depending on the specific solvent and stabilizer conditions employed.
Q: Can the excess methylamine be recycled?
A: Yes, the process involves a negative pressure recovery step where the excessive methylamine solution is distilled off and can be recycled for subsequent batches, significantly reducing raw material costs and waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Methyl-5-Hexene-1-Amine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceutical intermediates requires more than just a patent; it demands engineering excellence and unwavering commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. We have rigorously validated the synthesis of N-methyl-5-hexene-1-amine described in CN108003037B within our own facilities, confirming its ability to deliver stringent purity specifications (>95%) consistently. Our rigorous QC labs employ advanced analytical techniques to monitor every batch, guaranteeing that the impurity profile remains well within the limits required for GMP-grade API synthesis.
We invite you to collaborate with us to optimize your supply chain for this critical building block. Whether you require a Customized Cost-Saving Analysis for your specific volume requirements or need to evaluate the compatibility of this intermediate with your downstream chemistry, our technical procurement team is ready to assist. We encourage you to contact us to request specific COA data and route feasibility assessments tailored to your project needs. Let us help you secure a sustainable, safe, and cost-effective supply of high-purity N-methyl-5-hexene-1-amine for your next-generation therapeutics.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
