Scalable One-Pot Synthesis of Beta-Amino Ketones for Global Pharmaceutical Supply Chains
Introduction to Advanced Beta-Amino Ketone Synthesis
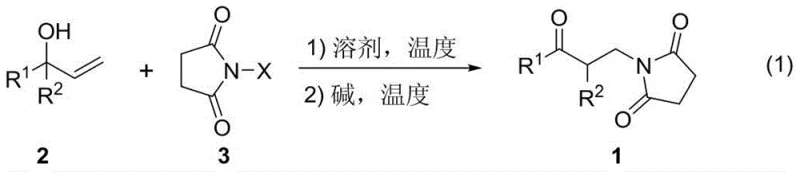
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access bioactive scaffolds, and the technology disclosed in patent CN108147989B represents a significant leap forward in the synthesis of beta-amino ketone derivatives. This innovative methodology utilizes readily available allyl alcohol compounds and N-halosuccinimides as starting materials to construct the valuable beta-amino ketone backbone through a streamlined one-pot two-step process. By leveraging the dual functionality of N-halosuccinimides, which act as both electrophilic halogen sources and nucleophilic nitrogen donors, this approach achieves exceptional atom economy while minimizing waste generation. The reaction proceeds under mild conditions, typically ranging from 25°C to 140°C, and delivers products with outstanding purity and yields reaching up to 99 percent. For R&D directors and process chemists, this patent offers a robust alternative to classical methods, promising to simplify synthetic routes for a wide array of drug candidates and functional materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of beta-amino ketone motifs has relied heavily on established protocols such as the Mannich reaction or the aza-Michael addition of amines to alpha,beta-unsaturated ketones. While these classical transformations are foundational in organic chemistry, they frequently suffer from significant operational drawbacks when applied to complex molecule synthesis. Traditional Mannich reactions often necessitate stringent control of pH and temperature, and they can be plagued by issues related to regioselectivity and the formation of polymeric byproducts. Furthermore, methods involving aziridines or vinyl magnesium bromide additions, as cited in prior art, often require cryogenic conditions, strictly anhydrous environments, or the use of expensive and toxic transition metal catalysts. These factors collectively contribute to prolonged reaction times, complicated downstream processing, and increased environmental burdens due to the generation of hazardous waste streams, thereby inflating the overall cost of goods for pharmaceutical intermediates.
The Novel Approach
In stark contrast to these legacy techniques, the novel approach detailed in CN108147989B introduces a paradigm shift by employing a direct functionalization of allyl alcohols. This method bypasses the need for pre-functionalized enones or sensitive organometallic reagents, instead utilizing stable and commodity-grade N-halosuccinimides. The reaction design is elegantly simple: the initial step involves the interaction of the allyl alcohol with the N-halosuccinimide in an organic solvent, followed by the addition of a mild inorganic base to trigger the amination cascade. This one-pot strategy eliminates the need for isolating unstable intermediates, significantly reducing solvent consumption and labor costs. The broad substrate scope allows for the incorporation of various aryl and alkyl groups, enabling the rapid generation of diverse chemical libraries for drug discovery without the logistical headaches associated with handling air-sensitive reagents or specialized catalytic systems.
Mechanistic Insights into N-Halosuccinimide Mediated Amination
The core innovation of this technology lies in the unique mechanistic role played by the N-halosuccinimide reagent, which serves a dual purpose that enhances the overall atom economy of the transformation. In the initial phase of the reaction, the N-halosuccinimide acts as an electrophilic halogen source, likely facilitating the activation of the allyl alcohol double bond or generating a reactive halohydrin-like intermediate. Subsequently, upon the addition of the base, the succinimide moiety, now deprotonated or activated, functions as a nucleophilic nitrogen source to attack the electrophilic carbon center. This intramolecular or intermolecular rearrangement effectively installs the amino group at the beta-position relative to the newly formed ketone functionality. This mechanism avoids the use of external amine sources that might require protection and deprotection strategies, thereby streamlining the synthetic sequence and reducing the total number of steps required to reach the target molecule.
From an impurity control perspective, this mechanism offers distinct advantages for manufacturing high-purity pharmaceutical intermediates. Because the reaction does not rely on transition metal catalysts, there is no risk of heavy metal contamination, which is a critical quality attribute for API manufacturing. The byproducts of the reaction are primarily succinimide derivatives and inorganic salts, which are generally easier to remove via standard aqueous workups or crystallization compared to complex organometallic residues. The mild reaction conditions, often proceeding efficiently at temperatures as low as 25°C or moderately elevated temperatures like 80°C, minimize thermal degradation of sensitive functional groups. This thermal stability ensures that the final product profile remains clean, with minimal formation of decomposition byproducts, thus simplifying the purification process and ensuring consistent batch-to-batch quality essential for regulatory compliance.
How to Synthesize Beta-Amino Ketone Derivatives Efficiently
To implement this synthesis in a laboratory or pilot plant setting, the procedure follows a straightforward protocol that balances reaction kinetics with operational simplicity. The process begins by dissolving the allyl alcohol substrate in a suitable polar aprotic solvent such as acetonitrile, which has been shown to provide optimal results in terms of solubility and reaction rate. The N-halosuccinimide, typically N-bromosuccinimide (NBS) or N-chlorosuccinimide (NCS), is added in a slight molar excess to drive the conversion to completion. Following the initial reaction period, a base such as sodium carbonate or potassium carbonate is introduced to the mixture, and the temperature is adjusted to facilitate the cyclization or rearrangement step. The detailed standardized synthesis steps for this process are outlined in the guide below.
- React allyl alcohol compound and N-halosuccinimide in an organic solvent at 25-140°C for 0.5-48 hours.
- Add a base such as sodium carbonate or potassium carbonate to the reaction mixture.
- Continue heating at 25-140°C to promote the amination reaction, then purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthesis route presents compelling economic and logistical benefits that directly impact the bottom line. The primary advantage stems from the utilization of commodity chemicals; allyl alcohols and N-halosuccinimides are produced on a massive industrial scale, ensuring a stable and competitive supply market that is less susceptible to the volatility seen with specialized catalysts or exotic reagents. This reliance on bulk chemicals significantly mitigates supply chain risks, ensuring continuity of supply even during global disruptions. Furthermore, the elimination of expensive transition metal catalysts removes a major cost driver from the bill of materials, while also obviating the need for costly metal scavenging resins or complex filtration steps during downstream processing.
- Cost Reduction in Manufacturing: The economic efficiency of this process is driven by its high atom economy and simplified operational workflow. By combining the halogenation and amination steps into a single vessel, manufacturers save substantially on solvent usage, energy consumption for heating and cooling cycles, and reactor occupancy time. The absence of precious metal catalysts means that capital is not tied up in expensive reagents that often have limited shelf lives or require special storage conditions. Additionally, the high yields reported, often exceeding 98 percent, mean that raw material waste is minimized, directly translating to a lower cost per kilogram of the final active pharmaceutical ingredient intermediate.
- Enhanced Supply Chain Reliability: The robustness of this chemical transformation enhances supply chain resilience by reducing dependency on single-source suppliers of niche reagents. Since the key inputs are widely available from multiple global chemical producers, procurement teams can easily diversify their vendor base to negotiate better pricing and secure backup supply lines. The mild reaction conditions also imply that the process can be safely scaled up in standard glass-lined or stainless steel reactors without requiring specialized high-pressure or cryogenic equipment, further expanding the pool of qualified contract manufacturing organizations (CMOs) capable of producing these intermediates.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method aligns well with green chemistry principles, which is increasingly important for maintaining social license to operate. The reaction generates benign byproducts and avoids the use of toxic heavy metals, simplifying wastewater treatment and hazardous waste disposal protocols. This reduced environmental footprint not only lowers compliance costs but also accelerates the regulatory approval process for new drug applications. The scalability is further supported by the exothermic nature of the reaction being manageable under standard cooling capacities, allowing for safe production scales ranging from pilot kilograms to multi-ton commercial batches.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this beta-amino ketone synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of this synthesis method over traditional Mannich reactions?
A: Unlike traditional Mannich reactions which often require harsh conditions and complex post-processing, this method utilizes a one-pot two-step process with mild conditions (25-140°C) and achieves yields up to 99%.
Q: Can N-chlorosuccinimide be used instead of N-bromosuccinimide in this reaction?
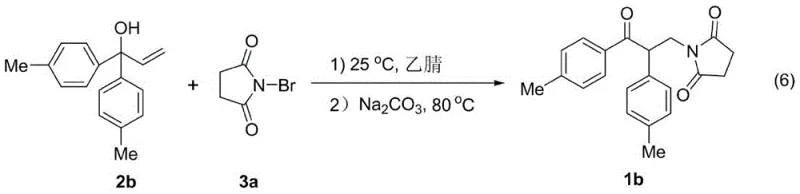
A: Yes, the patent demonstrates that N-chlorosuccinimide is a viable alternative, although N-bromosuccinimide generally provides higher yields (e.g., 99% vs 85% in specific examples).
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process uses easily obtainable raw materials like allyl alcohols and common bases, avoids expensive transition metal catalysts, and operates under mild conditions, making it highly scalable.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Amino Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of efficient synthetic methodologies like the one described in CN108147989B for accelerating drug development timelines. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped to handle the specific solvent systems and reaction conditions required for this chemistry, and our rigorous QC labs enforce stringent purity specifications to meet the highest international pharmacopeial standards. We are committed to delivering high-purity beta-amino ketone intermediates that enable our partners to focus on what they do best: innovating life-saving therapies.
We invite you to engage with our technical team to explore how this advanced synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this methodology for your supply chain. Please contact our technical procurement team today to discuss your requirements, request specific COA data for our catalog compounds, or initiate a conversation about route feasibility assessments for your proprietary molecules.
