Advanced Synthetic Route for Glyoxylspermidine: Scaling High-Purity Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust synthetic pathways for critical immunomodulatory and anticancer intermediates, and the technology disclosed in patent CN1014604B represents a significant leap forward in this domain. This patent details a novel method for preparing glyoxylspermidine, a pivotal intermediate in the synthesis of Spergualin analogs, which are renowned for their potent antitumor and immunosuppressive activities. Unlike traditional fermentation-based extraction methods that suffer from low yields and complex purification burdens, this chemical synthesis approach offers a deterministic and scalable alternative. By leveraging selective oxidative cleavage of specific carbon-carbon bonds within tailored polyamine precursors, the process bypasses the need for tedious aldehyde protection strategies. This technological breakthrough not only enhances the structural integrity of the final product but also aligns perfectly with the demands of a reliable pharmaceutical intermediates supplier seeking to optimize production efficiency. The ability to generate high-purity glyoxylspermidine through a controlled chemical sequence ensures consistent quality, which is paramount for downstream drug development.
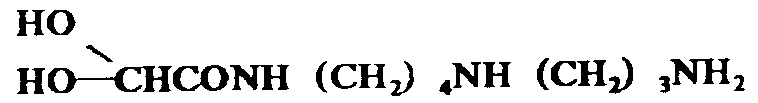
Furthermore, the versatility of this synthetic route allows for the derivation of various 15-deoxy Spergualin related compounds, expanding the chemical space available for medicinal chemistry teams. The strategic design of the precursor molecules enables precise control over the final substitution patterns, thereby facilitating the exploration of structure-activity relationships without the constraints of biological variability. For global supply chain stakeholders, this means a transition from unpredictable biological sources to a stable, chemically defined manufacturing process. The implications for cost reduction in API manufacturing are substantial, as the streamlined workflow reduces both material consumption and processing time. As we delve deeper into the technical specifics, it becomes evident that this patent provides a foundational platform for the commercial scale-up of complex polyamines, addressing long-standing bottlenecks in the production of this valuable therapeutic class.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of glyoxylspermidine has been heavily reliant on the hydrolysis of Spergualin obtained from microbial fermentation, a process fraught with inherent inefficiencies and operational complexities. The isolation of Spergualin from bacterial nutrient solutions involves arduous purification steps to separate the target molecule from a myriad of metabolic byproducts, often resulting in suboptimal recovery rates and inconsistent batch quality. Moreover, alternative total synthesis routes utilizing 3-amino-1-propanol as a starting material have encountered significant hurdles, particularly regarding the availability of glyoxylic acid hydrate and the necessity of introducing and subsequently removing aldehyde protecting groups. These protection-deprotection sequences add multiple synthetic steps, each contributing to yield loss, increased solvent usage, and higher overall production costs. The cumbersome nature of protecting group chemistry, involving acetals, hydrazones, or oximes, introduces additional safety hazards and waste disposal challenges, making these conventional methods less attractive for modern green chemistry initiatives. Consequently, the industry has faced persistent challenges in securing a cost reduction in electronic chemical manufacturing and pharma sectors alike due to these intrinsic process limitations.
The Novel Approach
In stark contrast, the novel approach outlined in the patent revolutionizes the synthesis by employing a direct oxidative cleavage strategy that elegantly circumvents the need for aldehyde protection entirely. By designing precursors with specific vicinal diol or amino-alcohol motifs, the method utilizes periodic acid reagents to selectively cleave carbon-carbon bonds, instantly generating the desired aldehyde functionality in the final step. This paradigm shift drastically simplifies the synthetic tree, reducing the number of unit operations and minimizing the exposure of sensitive intermediates to harsh conditions. The use of readily available starting materials such as tartaric acid derivatives and polyamines ensures a robust supply chain, mitigating the risks associated with scarce reagents. This streamlined methodology not only accelerates the timeline for reducing lead time for high-purity intermediates but also enhances the overall atom economy of the process. For procurement managers, this translates to a more predictable cost structure and a reduced dependency on complex, multi-step sequences that are prone to failure. The novel approach stands as a testament to process intensification, offering a clear pathway toward sustainable and economically viable manufacturing of high-value pharmaceutical building blocks.
Mechanistic Insights into Periodic Acid-Mediated Oxidative Cleavage
The core chemical transformation driving this innovative synthesis is the periodic acid-mediated oxidative cleavage of carbon-carbon bonds, a reaction renowned for its specificity towards vicinal diols and analogous functional groups. In this mechanism, the periodic acid reagent, such as sodium metaperiodate, coordinates with the adjacent hydroxyl groups on the precursor molecule to form a cyclic periodate ester intermediate. This cyclic species is inherently unstable and undergoes fragmentation to cleave the carbon-carbon bond connecting the hydroxyl-bearing carbons, resulting in the formation of two carbonyl groups. In the context of glyoxylspermidine synthesis, this reaction is meticulously tuned to cleave the bond adjacent to the amide linkage while preserving the integrity of the polyamine chain. The selectivity of this oxidation is crucial, as it prevents over-oxidation of the amine functionalities or degradation of the peptide bond, ensuring high fidelity in the product structure. Reaction conditions, including pH control and solvent composition, are optimized to facilitate this transformation in aqueous or mixed solvent systems, enhancing the environmental profile of the process. Understanding this mechanistic nuance is vital for R&D directors focusing on purity and impurity profiles, as it highlights the precision engineering embedded in the synthetic design.
Furthermore, the tolerance of this oxidative system towards various substituents allows for significant flexibility in precursor design, enabling the synthesis of a broad library of Spergualin analogs. The reaction proceeds efficiently at temperatures ranging from 0°C to the boiling point of the solvent, providing operational flexibility for scale-up. The use of water as a primary solvent component not only reduces the reliance on volatile organic compounds but also simplifies the workup procedure, as inorganic byproducts can be easily removed through filtration or ion exchange. This mechanistic robustness ensures that the process remains stable even when transitioning from laboratory bench scales to pilot plant operations. For technical teams, the predictability of the oxidative cleavage step minimizes the risk of unexpected side reactions, thereby safeguarding the quality of the final active pharmaceutical ingredient. The ability to execute this transformation without protecting the nascent aldehyde group is a masterstroke of synthetic planning, eliminating entire classes of impurities that typically arise from incomplete deprotection steps in conventional routes.
How to Synthesize Glyoxylspermidine Efficiently
The practical implementation of this synthesis involves a sequence of well-defined chemical operations that prioritize safety, efficiency, and yield optimization. The process begins with the preparation of the key precursor, typically achieved through the condensation of activated tartaric acid derivatives with protected polyamines, followed by necessary functional group modifications such as cyano reduction. Once the precursor is secured, the critical oxidative cleavage step is performed under controlled conditions to generate the target aldehyde. Detailed standard operating procedures for each stage, including reagent stoichiometry, temperature profiles, and purification protocols, are essential for reproducibility. The following guide outlines the standardized synthesis steps derived from the patent examples to ensure consistent production outcomes.
- Prepare the precursor compound (Formula I) by condensing protected amino acids or tartaric acid derivatives with spermidine or putrescine analogs.
- Perform selective oxidative cleavage using periodic acid reagents (e.g., sodium metaperiodate) in an aqueous or mixed solvent system.
- Purify the resulting glyoxylspermidine using ion-exchange chromatography and lyophilization to obtain the high-purity dihydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers profound advantages that resonate deeply with procurement managers and supply chain heads focused on operational excellence and cost efficiency. The elimination of aldehyde protecting groups fundamentally alters the cost structure of the manufacturing process by removing several synthetic steps, each of which incurs material, labor, and overhead costs. This simplification leads to substantial cost savings, not merely through reduced reagent consumption but also by shortening the overall production cycle time. The reliance on commodity chemicals like tartaric acid and putrescine derivatives ensures a stable and diversified supply base, reducing the vulnerability to raw material shortages that often plague specialty chemical markets. Furthermore, the aqueous nature of the key oxidation step aligns with increasingly stringent environmental regulations, potentially lowering waste disposal fees and enhancing the sustainability profile of the supply chain. These factors collectively contribute to a more resilient and cost-effective sourcing strategy for high-value intermediates.
- Cost Reduction in Manufacturing: The streamlined synthetic pathway significantly lowers the cost of goods sold by minimizing the number of unit operations and eliminating expensive protecting group reagents. By avoiding the need for specialized aldehyde protection and subsequent deprotection steps, the process reduces solvent usage and waste generation, leading to direct financial benefits. The high efficiency of the oxidative cleavage step ensures better resource utilization, allowing manufacturers to achieve competitive pricing without compromising on quality standards. This economic advantage is critical for maintaining margins in the highly competitive pharmaceutical intermediates market.
- Enhanced Supply Chain Reliability: Utilizing widely available starting materials such as tartaric acid and simple polyamines mitigates the risk of supply disruptions associated with exotic or fermentation-derived precursors. The chemical synthesis route is less susceptible to biological variability, ensuring consistent batch-to-batch quality and reliable delivery schedules. This stability is essential for downstream drug manufacturers who require uninterrupted supply to meet clinical and commercial demands. The robustness of the process supports long-term supply agreements and fosters stronger partnerships between suppliers and pharmaceutical clients.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing standard chemical engineering principles and equipment that are readily available in multipurpose manufacturing facilities. The preference for aqueous reaction media reduces the environmental footprint, facilitating compliance with green chemistry initiatives and regulatory requirements. This scalability ensures that production volumes can be rapidly increased to meet market demand without the need for significant capital investment in specialized infrastructure. The combination of operational flexibility and environmental stewardship makes this technology a preferred choice for sustainable manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and supply of glyoxylspermidine, providing clarity on the process capabilities and quality assurances. These insights are derived directly from the patent specifications and are intended to assist decision-makers in evaluating the feasibility of integrating this intermediate into their development pipelines. Understanding these nuances is key to leveraging the full potential of this advanced synthetic technology.
Q: What is the primary advantage of this glyoxylspermidine synthesis method?
A: The primary advantage is the elimination of aldehyde protecting groups. Conventional methods require cumbersome protection and deprotection steps, whereas this novel route utilizes selective oxidative cleavage to generate the aldehyde functionality directly, significantly simplifying the process.
Q: What are the key starting materials for this process?
A: The process utilizes widely available and cost-effective raw materials such as tartaric acid, diethyl tartrate, putrescine, and cyanoethyl putrescine. These commodities ensure a stable supply chain compared to fermentation-derived precursors.
Q: Is this method suitable for large-scale manufacturing?
A: Yes, the method is designed for industrial scalability. It avoids difficult purification steps associated with fermentation broths and uses standard chemical reagents like periodic acid, making it highly adaptable for commercial scale-up of complex polyamines.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Glyoxylspermidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of life-saving therapies, and we are committed to delivering excellence in every batch. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with precision and reliability. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every gram of glyoxylspermidine meets the highest industry standards. Our expertise in handling complex polyamine chemistry allows us to navigate the intricacies of this synthesis with confidence, providing you with a secure and dependable source for your project needs.
We invite you to collaborate with us to optimize your supply chain and accelerate your drug development timelines. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project requirements, demonstrating how our advanced manufacturing capabilities can drive value for your organization. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate why we are the partner of choice for your pharmaceutical intermediate needs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
