Revolutionizing 4-Amino-2,6-Dimethoxypyrimidine Production: A Green Commercial Scale-Up Strategy
The pharmaceutical industry is currently witnessing a pivotal shift towards greener synthetic methodologies, particularly in the production of critical heterocyclic intermediates. Patent CN109232441B, published in August 2021, introduces a groundbreaking preparation method for 4-amino-2,6-dimethoxypyrimidine, a key precursor for the long-acting sulfonamide antibiotic sulfadimethoxine. This technical disclosure addresses the longstanding environmental and safety hazards associated with traditional chlorination processes by proposing a novel condensation-methylation-cyclization pathway. For R&D directors and procurement strategists, this patent represents more than just a chemical variation; it signifies a robust opportunity to optimize supply chain resilience while adhering to increasingly stringent environmental regulations. The method utilizes o-methyl isourea salts and cyanoacetates as primary feedstocks, effectively bypassing the need for toxic phosphorus oxychloride. This fundamental change in synthetic logic not only enhances the safety profile of the manufacturing facility but also streamlines the downstream purification processes, offering a compelling value proposition for high-volume commercial production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 4-amino-2,6-dimethoxypyrimidine has relied heavily on two mature but problematic process routes, both of which pose significant operational risks and environmental burdens. The first conventional method utilizes barbituric acid as the starting material, employing phosphorus oxychloride as a chlorinating agent to introduce the necessary functional groups. This approach is fraught with danger, as the reaction generates 4-amino-2,6-dichloropyrimidine phosphoryl dichloride as a byproduct, a compound known to be highly explosive upon contact with moisture. Furthermore, the second mainstream method, which employs ethyl cyanoacetate and urea, also necessitates the use of phosphorus oxychloride in the chlorination step. Both traditional pathways result in the generation of substantial volumes of colored, phosphorus-containing wastewater, which requires complex and costly treatment protocols to meet environmental discharge standards. Additionally, these older methods often suffer from low selectivity, leading to the formation of difficult-to-separate isomer by-products that compromise the overall yield and purity of the final active pharmaceutical ingredient intermediate.
The Novel Approach
In stark contrast to the hazardous legacy methods, the novel approach detailed in the patent data revolutionizes the synthetic landscape by completely eliminating the chlorination step. This new strategy leverages a condensation reaction between o-methyl isourea salts and cyanoacetates, followed by a controlled methylation and a final self-cyclization event. By circumventing the use of phosphorus oxychloride, the process inherently removes the risk of generating explosive phosphoryl dichloride intermediates, thereby drastically improving plant safety. The reaction pathway is designed to optimize yield and selectivity through precise temperature control and solvent management, utilizing common reagents like dimethyl sulfate or dimethyl carbonate for methylation. This streamlined workflow not only simplifies the operational complexity but also significantly reduces the volume and toxicity of waste effluent, providing a green and feasible route for the industrialized production of 4-amino-2,6-dimethoxypyrimidine. The elimination of phosphorus waste streams translates directly into reduced environmental compliance costs and a more sustainable manufacturing footprint.
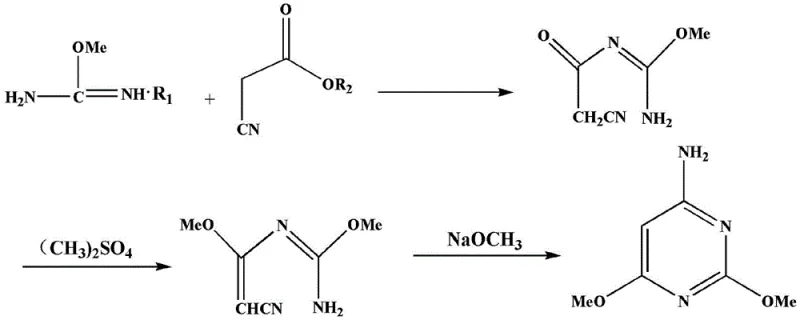
Mechanistic Insights into Condensation and Cyclization Dynamics
The core of this technological advancement lies in the precise manipulation of reaction intermediates to achieve high-fidelity ring closure without the need for harsh chlorinating agents. The mechanism initiates with the condensation of o-methyl isourea salt and cyanoacetate in an alcoholic solvent, typically methanol or ethanol, under reflux conditions ranging from 60°C to 90°C. This step forms the critical intermediate N'-ethylcyanocarbonyl-O-methylisourea, which serves as the scaffold for subsequent functionalization. The reaction kinetics are carefully managed to ensure complete conversion while minimizing side reactions, with the solvent being distilled off under reduced pressure to isolate the intermediate as a pale yellow powder. This isolation step is crucial for maintaining the stoichiometry required for the subsequent methylation phase, where phase transfer catalysts such as tetrabutylammonium bromide facilitate the efficient transfer of ionic species into the organic phase. The meticulous control over these intermediate stages ensures that the molecular architecture is primed for the final cyclization event, maximizing the structural integrity of the pyrimidine ring.
Following the methylation step, which introduces the second methoxy group using agents like dimethyl sulfate, the synthesis proceeds to the final cyclization using a sodium methoxide methanol solution. This base-catalyzed cyclization is the defining moment where the linear precursor folds into the stable 4-amino-2,6-dimethoxypyrimidine structure. The patent data highlights the importance of pH adjustment during the workup phase, where alkali such as sodium carbonate is added to neutralize the reaction mixture to a pH of 7-9. This specific pH control is vital for precipitating the product in its purest form, effectively separating it from any remaining soluble impurities or unreacted starting materials. The structural evolution from the open-chain intermediates to the final cyclic product is depicted clearly in the provided chemical diagrams, showcasing the transformation of the nitrile and amidine functionalities into the aromatic pyrimidine system. This mechanistic clarity allows process chemists to fine-tune reaction parameters for optimal impurity profiles, ensuring that the final material meets the stringent purity specifications required for pharmaceutical applications.

How to Synthesize 4-Amino-2,6-Dimethoxypyrimidine Efficiently
Implementing this synthesis route requires a disciplined approach to reaction engineering, focusing on the sequential execution of condensation, methylation, and cyclization steps under controlled thermal conditions. The process begins with the dissolution of o-methyl isourea salts in a suitable alcohol solvent, followed by the addition of cyanoacetate and heating to initiate the condensation. Once the initial intermediate is formed and isolated via distillation, it is subjected to methylation in the presence of a solid base and a phase transfer catalyst, a step that demands precise temperature regulation between 50°C and 80°C to prevent decomposition. The final cyclization is achieved by treating the methylated intermediate with sodium methoxide, followed by a careful workup involving solvent removal and pH adjustment to induce crystallization. For a detailed breakdown of the specific molar ratios, solvent volumes, and thermal profiles required for each stage, please refer to the standardized synthesis guide below.
- Condense o-methyl isourea salt with cyanoacetate in alcohol solvent under reflux to form N'-ethylcyanocarbonyl-O-methylisourea.
- Perform methylation using dimethyl sulfate or dimethyl carbonate with a phase transfer catalyst to yield the vinyl intermediate.
- Execute thermal cyclization using sodium methoxide in methanol, followed by pH adjustment and crystallization to isolate the final pyrimidine.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this POCl3-free synthesis route offers substantial advantages in terms of cost structure and supply chain stability. By eliminating the need for phosphorus oxychloride, manufacturers can avoid the high costs associated with handling, storing, and disposing of hazardous chlorinating agents and their toxic byproducts. This reduction in hazardous material usage directly correlates to lower operational expenditures related to safety compliance, waste treatment, and environmental remediation. Furthermore, the simplified process flow, which removes the complex purification steps often required to separate chlorinated isomers, leads to a more efficient utilization of reactor time and labor resources. For supply chain heads, this translates into a more predictable production schedule with reduced risk of unplanned downtime caused by safety incidents or regulatory inspections related to hazardous waste management.
- Cost Reduction in Manufacturing: The elimination of phosphorus oxychloride and the associated chlorination step removes the necessity for expensive corrosion-resistant equipment and specialized waste treatment facilities required for phosphorus-containing effluents. This fundamental process simplification leads to significant capital expenditure savings and reduced ongoing operational costs for waste disposal. Additionally, the higher selectivity of the new route minimizes the loss of raw materials to by-product formation, thereby improving the overall material efficiency and reducing the cost of goods sold. The use of readily available and cost-effective reagents like o-methyl isourea salts and cyanoacetates further enhances the economic viability of the process, making it a highly attractive option for large-scale commercial manufacturing.
- Enhanced Supply Chain Reliability: By relying on stable, non-hazardous raw materials, the supply chain becomes more resilient to regulatory changes and transportation restrictions often imposed on toxic chemicals like phosphorus oxychloride. The simplified workflow reduces the number of critical process steps, thereby lowering the probability of batch failures and ensuring a more consistent supply of high-purity intermediates. This reliability is crucial for pharmaceutical customers who require uninterrupted access to key building blocks for their drug production pipelines. The ability to source raw materials that are less regulated and more abundant in the global market further secures the supply chain against potential disruptions, ensuring long-term continuity of supply for downstream API manufacturers.
- Scalability and Environmental Compliance: The green nature of this synthesis pathway aligns perfectly with global trends towards sustainable chemistry, making it easier to obtain environmental permits for facility expansion. The absence of explosive intermediates and toxic phosphorus waste simplifies the safety validation process for scaling up from pilot to commercial production volumes. This ease of scale-up allows manufacturers to respond quickly to increased market demand without the lengthy lead times associated with upgrading hazardous waste treatment infrastructure. Moreover, the reduced environmental footprint enhances the corporate social responsibility profile of the manufacturer, appealing to multinational pharmaceutical companies that prioritize sustainable sourcing in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on safety, purity, and scalability. Understanding these aspects is essential for technical teams evaluating the feasibility of adopting this greener manufacturing process. The responses highlight the specific advantages over traditional methods, focusing on the elimination of hazardous reagents and the improvement in overall process efficiency.
Q: How does this new method improve safety compared to traditional barbituric acid routes?
A: Traditional methods rely on phosphorus oxychloride (POCl3), which generates explosive phosphoryl dichloride byproducts upon contact with water. This novel protocol eliminates chlorination entirely, removing the risk of explosive intermediates and significantly reducing hazardous waste handling requirements.
Q: What are the primary impurities controlled in this synthesis?
A: By avoiding the chlorination step, the formation of chlorinated isomers and phosphorus-containing organic impurities is prevented. The process utilizes specific pH adjustments during the workup phase to ensure high purity of the final 4-amino-2,6-dimethoxypyrimidine crystal lattice.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process simplifies the workflow by removing complex purification steps associated with phosphorus waste. The use of common solvents like methanol and acetone, along with stable solid bases, facilitates straightforward scale-up from pilot plants to multi-ton commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Amino-2,6-Dimethoxypyrimidine Supplier
The technical potential of this POCl3-free synthesis route is immense, offering a pathway to safer, cleaner, and more cost-effective production of this critical pharmaceutical intermediate. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that validate every batch against the highest industry standards. We understand the critical nature of supply chain continuity for our partners and have optimized our operations to deliver high-purity 4-amino-2,6-dimethoxypyrimidine with consistent reliability.
We invite procurement leaders and technical directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific production requirements. Our technical procurement team is ready to provide specific COA data and route feasibility assessments to demonstrate how this green synthesis method can enhance your supply chain efficiency. By partnering with us, you gain access to a manufacturing capability that balances economic performance with environmental responsibility, securing your position in a competitive global market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
