Advanced Organocatalytic Synthesis of D-p-MPSE for Scalable Florfenicol Production
The pharmaceutical landscape for veterinary antibiotics is constantly evolving, driven by the critical need for high-purity intermediates that ensure drug efficacy and safety. A significant breakthrough in this domain is documented in patent CN112479946B, which discloses a sophisticated method for preparing D-p-methylsulfonyl phenylserine ethyl ester (D-p-MPSE) with exceptional stereoselectivity. This compound serves as a pivotal chiral building block for Florfenicol, a broad-spectrum antibiotic widely utilized in clinical and agricultural settings. The patent highlights a transition from traditional resolution methods to a more elegant asymmetric organocatalytic approach, utilizing a polystyrene-supported L-proline catalyst system. This technological shift addresses long-standing challenges in chiral synthesis, such as low yields and excessive waste generation, positioning this methodology as a cornerstone for modern reliable veterinary antibiotic intermediate supplier strategies aiming for greener and more efficient manufacturing protocols.
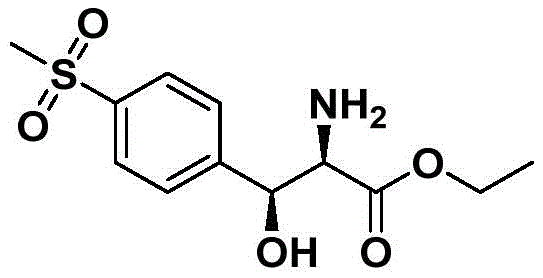
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of D-p-MPSE has relied heavily on classical resolution techniques or copper-mediated pathways, which present substantial operational bottlenecks. Traditional routes often necessitate the use of large quantities of resolving agents to separate racemic mixtures, a process that inherently caps the maximum theoretical yield at 50% and generates significant amounts of unwanted isomeric waste. Furthermore, existing methods frequently employ strong mineral acids like hydrochloric or sulfuric acid during the synthesis or workup phases, leading to severe corrosion of production equipment and increased maintenance costs. The recovery of solvents and chiral auxiliaries in these legacy processes is often inefficient, contributing to a higher environmental footprint and complicating the supply chain logistics for cost reduction in veterinary pharmaceutical manufacturing. These inefficiencies underscore the urgent need for a catalytic system that can bypass resolution entirely while maintaining rigorous quality standards.
The Novel Approach
The methodology outlined in the patent introduces a paradigm shift by employing an asymmetric Aldol reaction catalyzed by polystyrene-supported L-proline (PS-L-proline) in conjunction with a specific boron additive. This novel catalytic system operates under remarkably mild conditions, typically between 20-30°C, eliminating the need for extreme temperatures or pressures that strain facility resources. By utilizing a heterogeneous catalyst, the process allows for the physical separation of the catalyst via simple filtration post-reaction, facilitating immediate catalyst recovery and reuse without complex extraction procedures. The integration of di-n-butyl boron trifluoromethanesulfonate as an additive further refines the stereochemical outcome, effectively suppressing the formation of undesired cis-isomers and enhancing the enantiomeric excess. This approach not only streamlines the synthetic route but also aligns perfectly with the goals of a high-purity pharmaceutical intermediate producer seeking to minimize downstream purification burdens.
Mechanistic Insights into PS-L-Proline Catalyzed Asymmetric Aldol Reaction
The core of this technological advancement lies in the synergistic interaction between the chiral organocatalyst and the Lewis acid additive during the carbon-carbon bond-forming event. The PS-L-proline functions by forming a chiral enamine intermediate with the N-Boc-glycine ethyl ester, which then attacks the p-methylsulfonylbenzaldehyde electrophile. The polystyrene support plays a crucial role beyond mere immobilization; it creates a unique microenvironment that can influence the transition state geometry, potentially reducing steric hindrance compared to free proline in solution. The addition of the boron triflate species is hypothesized to coordinate with the carbonyl oxygen of the aldehyde or the enolate species, rigidifying the transition state and thereby exerting tighter control over the facial selectivity of the nucleophilic attack. This dual-activation strategy ensures that the reaction proceeds with high diastereoselectivity, favoring the formation of the desired threo-configuration essential for biological activity.
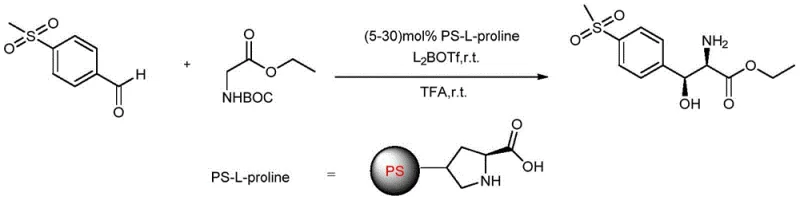
Controlling the impurity profile is paramount when synthesizing intermediates for potent antibiotics like Florfenicol, where even minor stereoisomers can compromise therapeutic value or introduce toxicity. The described catalytic system inherently minimizes the generation of the L-enantiomer and other diastereomeric byproducts through precise stereochemical steering. Unlike enzymatic resolutions which may suffer from substrate specificity issues or narrow operating windows, this small-molecule catalytic system demonstrates robust tolerance to reaction variables. The use of trifluoroacetic acid (TFA) in the final deprotection step is specifically chosen because it cleaves the Boc protecting group efficiently without racemizing the sensitive alpha-amino acid center, preserving the optical integrity established during the initial Aldol step. This meticulous attention to mechanistic detail ensures that the final commercial scale-up of complex veterinary intermediates yields a product with consistent and reliable optical purity, meeting the stringent specifications required by global regulatory bodies.
How to Synthesize D-p-MPSE Efficiently
Implementing this synthesis requires careful attention to reagent stoichiometry and reaction monitoring to maximize the benefits of the catalytic cycle. The process begins with the suspension of the solid-supported catalyst in a suitable solvent, followed by the sequential addition of the aldehyde and the glycine derivative. Maintaining the reaction temperature within the narrow 20-30°C window is critical to balance reaction kinetics with stereocontrol, as deviations could lead to background non-catalyzed reactions that erode enantiomeric excess. Following the completion of the Aldol condensation, the heterogeneous nature of the catalyst allows for a straightforward workup where the solid beads are removed by filtration, leaving a clear solution ready for deprotection. For a comprehensive understanding of the specific operational parameters, including exact molar ratios and purification techniques, please refer to the detailed standardized synthesis steps provided below.
- Combine p-methylsulfonylbenzaldehyde and N-Boc-glycine ethyl ester in dichloromethane with PS-L-proline catalyst and di-n-butyl boron trifluoromethanesulfonate additive.
- Maintain the asymmetric Aldol reaction at 20-30°C for 30-120 hours to ensure optimal stereochemical control.
- Filter to recover the catalyst, treat the filtrate with trifluoroacetic acid to remove the Boc group, and purify via recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this organocatalytic route offers transformative benefits that extend far beyond simple yield improvements. The ability to recycle the polystyrene-supported catalyst represents a direct avenue for cost reduction in fine chemical manufacturing, as it drastically lowers the recurring expense of chiral catalysts which are traditionally among the most expensive line items in a bill of materials. Furthermore, the elimination of strong corrosive acids during the main synthetic step significantly extends the lifespan of reactor vessels and piping, reducing capital expenditure on equipment replacement and maintenance downtime. The simplified workup procedure, characterized by filtration rather than complex extractions or chromatographic separations, accelerates the overall batch cycle time, thereby enhancing throughput capacity without the need for additional infrastructure investment. These factors collectively contribute to a more resilient and cost-effective supply chain capable of meeting fluctuating market demands.
- Cost Reduction in Manufacturing: The implementation of a recyclable heterogeneous catalyst system fundamentally alters the cost structure of production by minimizing catalyst consumption. Since the PS-L-proline can be recovered via simple filtration and reused in subsequent batches, the effective cost per kilogram of the active catalyst is dramatically reduced compared to homogeneous systems where the catalyst is lost in the aqueous waste stream. Additionally, the avoidance of expensive resolution reagents and the reduction in solvent usage due to streamlined purification steps further compound these savings. This economic efficiency allows manufacturers to offer more competitive pricing structures while maintaining healthy margins, a critical advantage in the price-sensitive veterinary pharmaceutical market.
- Enhanced Supply Chain Reliability: The robustness of this synthetic method ensures a stable and continuous supply of critical intermediates, mitigating the risks associated with batch failures or inconsistent quality. The mild reaction conditions reduce the dependency on specialized high-pressure or cryogenic equipment, making the process adaptable to a wider range of manufacturing facilities globally. This flexibility is vital for reducing lead time for high-purity intermediates, as it allows for rapid scaling across different sites if demand surges. Moreover, the use of readily available starting materials like p-methylsulfonylbenzaldehyde ensures that raw material sourcing remains stable, preventing bottlenecks that could disrupt the delivery of finished veterinary drugs to end-users.
- Scalability and Environmental Compliance: As regulatory pressure mounts regarding chemical waste and environmental impact, this green chemistry approach positions manufacturers favorably for future compliance. The reduction in hazardous waste generation, particularly from avoided resolution salts and corrosive acid neutralization streams, simplifies waste treatment protocols and lowers disposal costs. The process is inherently scalable, moving seamlessly from laboratory gram-scale experiments to multi-ton commercial production without significant re-optimization, ensuring that supply can grow in lockstep with the expanding global demand for animal health products. This alignment with sustainability goals also enhances the brand reputation of suppliers committed to responsible manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of D-p-MPSE, derived directly from the patented technology and industry best practices. Understanding these nuances is essential for R&D teams evaluating process transfer and procurement officers assessing supplier capabilities. The answers provided reflect the specific advantages of the PS-L-proline catalytic system over legacy methods, focusing on practical implementation details and quality assurance metrics that define modern pharmaceutical intermediate production standards.
Q: What is the primary advantage of using PS-L-proline over free L-proline?
A: The polystyrene-supported catalyst allows for simple filtration and reuse, significantly reducing catalyst consumption and simplifying downstream processing compared to homogeneous systems.
Q: How does the boron additive improve the reaction outcome?
A: Di-n-butyl boron trifluoromethanesulfonate acts as a Lewis acid additive that enhances the enolization process, working synergistically with the chiral catalyst to boost both yield and optical purity.
Q: Is this method suitable for large-scale veterinary drug manufacturing?
A: Yes, the mild reaction conditions (20-30°C) and avoidance of strong corrosive acids in the main step make this process highly scalable and equipment-friendly for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable D-p-MPSE Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the efficacy of veterinary therapeutics like Florfenicol. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated organocatalytic routes described in recent patents can be translated into robust industrial realities. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced chiral HPLC capabilities to guarantee that every batch of D-p-MPSE meets the exacting optical purity requirements necessary for downstream antibiotic synthesis. Our commitment to technical excellence ensures that our clients receive a product that is not only chemically pure but also consistently reliable in its performance during subsequent formulation steps.
We invite global partners to engage with our technical procurement team to discuss how our manufacturing capabilities can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized processes can reduce your overall cost of goods sold. We encourage potential collaborators to contact us for specific COA data and route feasibility assessments tailored to your specific volume requirements. Let us demonstrate how our expertise in chiral synthesis can become a strategic asset to your organization, driving efficiency and quality in your veterinary drug portfolio.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
