Scaling Isopropylpyrazolone Production: A Low-Pressure Hydrogenation Breakthrough for Global API Supply Chains
The pharmaceutical industry continuously seeks robust manufacturing pathways that balance high purity with economic viability, particularly for established analgesic intermediates. Patent CN101328147A introduces a transformative low-pressure hydrogenation process for the synthesis of Isopropylpyrazolone, a critical precursor to the widely used antipyretic drug Isopropylantipyrine. This technology addresses longstanding inefficiencies in traditional manufacturing by replacing expensive noble metal catalysts with cost-effective Raney Nickel while simultaneously lowering reaction pressures to safe, manageable levels. By utilizing acetone as a dual-function solvent and reactant, the process achieves conversion rates exceeding 99% and product purity levels above 99%, setting a new benchmark for quality in pharmaceutical intermediate production. For global procurement teams and R&D directors, this patent represents a strategic opportunity to optimize supply chains through a method that is not only chemically superior but also environmentally sustainable and economically scalable.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Isopropylpyrazolone has relied on three primary methodologies, each fraught with significant operational and economic drawbacks that hinder efficient large-scale manufacturing. The so-called Roman process utilizes Palladium on Carbon (Pd/C) or Pd/Al2O3 catalysts; while effective, the reliance on precious metals drives up raw material costs substantially and introduces risks of catalyst poisoning that can halt production lines. Alternatively, the German process employs zinc powder reduction in acetic acid, a method notorious for generating massive quantities of hazardous wastewater and inorganic salt waste, creating severe environmental compliance burdens and disposal costs for manufacturers. Furthermore, the Czech process, which utilizes nickel catalysts, requires extremely high reaction pressures ranging from 1.6 to 3.0 MPa, necessitating expensive high-pressure vessel investments and posing elevated safety risks to plant personnel. These legacy methods collectively struggle to meet modern demands for green chemistry, cost containment, and operational safety, creating a pressing need for technological innovation in this sector.
The Novel Approach
The innovative process detailed in the patent data overcomes these historical barriers by integrating Raney Nickel catalysis with a low-pressure hydrogenation regime, fundamentally reshaping the production landscape for this key intermediate. By operating at a mild pressure range of 0.20 to 0.35 MPa, the new method eliminates the need for specialized high-pressure equipment, allowing facilities to utilize standard glass-lined or stainless steel reactors that are already common in fine chemical plants. The substitution of methanol or acetic acid with acetone serves a dual purpose: it acts as the solvent to enhance reactant concentration and simultaneously participates as a reactant in the condensation step, thereby streamlining the stoichiometry and improving reaction kinetics. This approach not only matches the high yield (≥90%) and purity (≥99%) of the expensive palladium routes but also avoids the heavy metal contamination issues associated with zinc reduction, delivering a cleaner, safer, and more cost-efficient manufacturing solution.
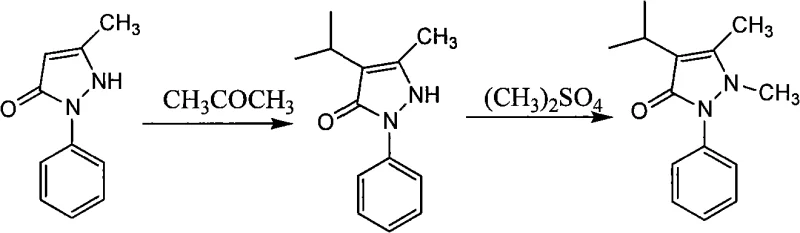
Mechanistic Insights into Raney Nickel-Catalyzed Low-Pressure Hydrogenation
The core of this technological advancement lies in the synergistic interaction between the Raney Nickel catalyst and the unique solvent system, which facilitates a highly selective hydrogenation pathway. Raney Nickel, a solid catalyst formed from a nickel-aluminum alloy, provides a high surface area of active nickel sites that effectively adsorb hydrogen and the unsaturated bonds of the pyrazolone-acetone condensation product. Unlike homogeneous catalysts or soluble metal salts, the heterogeneous nature of Raney Nickel allows for easy separation via simple filtration post-reaction, preventing metal leaching into the final product and ensuring the stringent purity specifications required for pharmaceutical intermediates are met without complex purification steps. The catalyst's robustness under the specified conditions of 75 to 85°C ensures consistent activity throughout the batch cycle, maintaining a steady reaction rate that drives the conversion of pyrazolone to near completion.
Impurity control is rigorously managed through the precise regulation of reaction parameters and the inherent selectivity of the catalytic system. The use of acetone as the reaction medium significantly increases the local concentration of reactants around the catalyst surface, which favors the desired addition reaction over potential side reactions such as polymerization or over-reduction. Monitoring the reaction endpoint via High-Performance Liquid Chromatography (HPLC) allows operators to terminate hydrogen intake precisely when pyrazolone conversion reaches ≥99%, preventing the formation of degradation byproducts that could compromise the quality of the downstream Isopropylantipyrine. This tight control over the reaction profile ensures that the resulting Isopropylpyrazolone possesses a liquid phase content of ≥99%, minimizing the burden on downstream crystallization and drying units and guaranteeing a consistent quality profile for the final API.
How to Synthesize Isopropylpyrazolone Efficiently
Implementing this synthesis route requires careful attention to the charging sequence and pressure management to maximize safety and yield. The process begins with the simultaneous introduction of pyrazolone, the Raney Nickel catalyst, and acetone into the reactor, establishing the optimal reactant-to-catalyst ratio defined in the patent examples. Following the initial charge, the system undergoes a rigorous purging sequence with nitrogen and hydrogen to remove atmospheric oxygen, a critical safety step before pressurization. Once the inert atmosphere is secured, the mixture is heated to the target range of 75 to 85°C, and hydrogen is introduced to maintain the system pressure between 0.20 and 0.35 MPa. Detailed standardized synthetic steps for this procedure are provided in the guide below.
- Charge the reactor with Pyrazolone, Raney Nickel catalyst, and Acetone acting as both solvent and reactant.
- Purge the system with Nitrogen and Hydrogen, then pressurize to 0.20-0.35 MPa and heat to 75-85°C.
- Monitor reaction progress via HPLC until Pyrazolone conversion reaches ≥99%, then filter, cool, and crystallize the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this low-pressure hydrogenation technology translates directly into tangible operational efficiencies and risk mitigation strategies. The shift away from precious metal catalysts like palladium removes a major variable cost driver from the bill of materials, as Raney Nickel is significantly more abundant and affordable while delivering equivalent catalytic performance. Additionally, the elimination of zinc powder and acetic acid solvents removes the logistical and financial burden associated with the treatment and disposal of heavy metal-laden wastewater, aligning production with increasingly strict global environmental regulations. The ability to run this reaction at near-atmospheric pressures further reduces capital expenditure requirements, as existing standard reactor infrastructure can be utilized without the need for costly upgrades to high-pressure containment systems.
- Cost Reduction in Manufacturing: The replacement of expensive palladium catalysts with Raney Nickel results in substantial raw material savings, as the latter is a non-precious metal with a much lower market price point. Furthermore, the dual role of acetone as both solvent and reactant simplifies the material input list and reduces the volume of solvents required, leading to lower procurement costs and reduced solvent recovery expenses. The high conversion rate minimizes the loss of valuable starting materials, ensuring that the theoretical yield is closely approached in practical production scenarios, which optimizes the overall cost per kilogram of the produced intermediate.
- Enhanced Supply Chain Reliability: Utilizing widely available commodities such as acetone and Raney Nickel mitigates the supply risk associated with scarce precious metals or specialized reagents that may face geopolitical or logistical bottlenecks. The robustness of the catalyst and the mild reaction conditions contribute to higher batch success rates and reduced downtime caused by equipment failure or safety incidents. This reliability ensures a consistent flow of high-purity intermediates to downstream API manufacturers, preventing production delays and securing the continuity of the broader pharmaceutical supply chain.
- Scalability and Environmental Compliance: The process is inherently scalable from laboratory benchtop to multi-ton commercial production due to the use of standard agitation and heating systems compatible with common reactor types. From an environmental perspective, the absence of heavy metal waste streams and the recyclability of the acetone solvent significantly reduce the ecological footprint of the manufacturing process. This alignment with green chemistry principles facilitates easier regulatory approval and permits for new production lines, accelerating the time-to-market for facilities adopting this technology.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this hydrogenation technology, derived directly from the patent specifications and comparative data. Understanding these nuances is essential for technical teams evaluating the feasibility of transitioning from legacy processes to this modernized approach. The answers provided reflect the specific advantages in safety, cost, and quality demonstrated in the experimental examples.
Q: Why is Raney Nickel preferred over Palladium catalysts for Isopropylpyrazolone synthesis?
A: Raney Nickel offers comparable catalytic activity to precious metal catalysts like Pd/C but at a significantly lower cost. Furthermore, it avoids the risk of catalyst poisoning often associated with noble metals and eliminates the need for expensive metal recovery processes, thereby optimizing the overall production economics.
Q: How does the low-pressure process improve safety compared to traditional methods?
A: Traditional Czech processes often require high pressures ranging from 1.6 to 3.0 MPa, necessitating specialized high-pressure equipment and posing greater safety risks. The novel process operates at a mild 0.20-0.35 MPa, allowing the use of standard glass-lined or stainless steel reactors, which drastically reduces capital expenditure and operational hazards.
Q: What are the environmental benefits of using Acetone as a solvent in this reaction?
A: Acetone serves a dual role as both the reactant and the solvent, which increases reactant concentration and reaction efficiency. Unlike the German zinc powder reduction method which generates large volumes of wastewater and inorganic salts, this hydrogenation route is cleaner, and the solvent can be easily recycled, minimizing environmental impact.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isopropylpyrazolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of deploying advanced synthetic routes like the Raney Nickel low-pressure hydrogenation process to meet the evolving demands of the global pharmaceutical market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Isopropylpyrazolone meets the highest international standards, providing our partners with a reliable foundation for their API synthesis.
We invite forward-thinking organizations to collaborate with us to leverage these process innovations for their supply chain optimization. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments that demonstrate how our manufacturing capabilities can drive value and efficiency in your production of analgesic intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
