Advanced Synthetic Route for 2-Fluoro-5-Bromoacetophenone: Enabling Scalable Production of Kinase Inhibitor Intermediates
Advanced Synthetic Route for 2-Fluoro-5-Bromoacetophenone: Enabling Scalable Production of Kinase Inhibitor Intermediates
The pharmaceutical industry continuously seeks robust and efficient synthetic pathways for critical heterocyclic intermediates, particularly those serving as scaffolds for kinase inhibitors. Patent CN107011134B, published in early 2021, introduces a transformative synthetic method for 2-fluoro-5-bromoacetophenone, a pivotal precursor in the construction of 5-bromo-3-methyl-1H-indazole derivatives. These indazole-based compounds have demonstrated profound biological activity, specifically as protein kinase Akt1 inhibitors with potential applications in treating malignancies, cardiovascular disorders, and inflammatory conditions. The disclosed technology addresses long-standing challenges in the supply chain of these high-value intermediates by replacing multi-step, hazardous processes with a streamlined, two-step lithiation-acetylation protocol. For R&D directors and procurement specialists alike, this innovation represents a significant leap forward in process chemistry, offering a pathway to reduce lead time for high-purity pharmaceutical intermediates while mitigating the risks associated with complex, low-yielding legacy syntheses.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
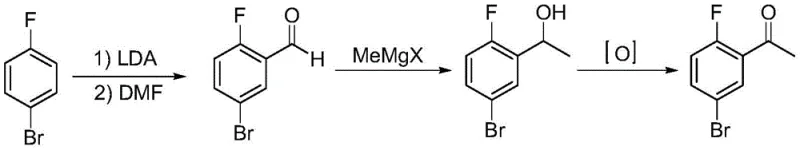
Historically, the synthesis of 2-fluoro-5-bromoacetophenone has been plagued by inefficiencies that hinder large-scale commercial viability. Traditional routes often rely on starting materials like 2-fluoro-5-bromobenzaldehyde or 2-fluoro-5-bromobenzoic acid, necessitating lengthy reaction sequences that involve multiple purification stages and harsh reagents. For instance, one common prior art route involves the conversion of 4-bromofluorobenzene to the corresponding aldehyde, followed by Grignard addition and oxidation, a process that is not only operationally complex but also suffers from moderate yields and difficult impurity profiles. Another established method utilizes 2-fluoro-5-bromobenzoic acid, requiring activation to an acyl chloride followed by reaction with organometallic species, which introduces significant safety hazards due to the handling of corrosive acyl chlorides and generates substantial waste streams. Furthermore, a previously reported two-step route utilizing ethyl difluoroacetate, while shorter in step count, relies on a raw material that is prohibitively expensive and exhibits high toxicity, rendering it unsuitable for sustainable industrial application. These conventional methodologies collectively impose a heavy burden on manufacturing costs and environmental compliance, creating bottlenecks in the reliable supply of this essential chemical building block.
The Novel Approach
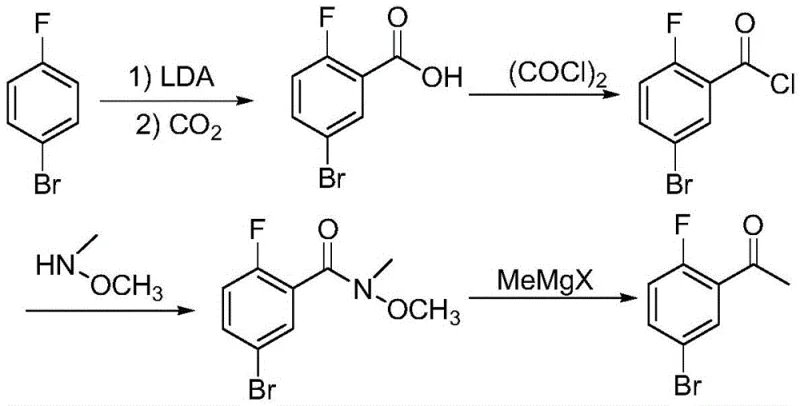
In stark contrast to these cumbersome legacy processes, the method disclosed in CN107011134B leverages the power of directed ortho-metalation to achieve a direct and highly efficient transformation. By utilizing 4-bromofluorobenzene as the sole starting material, the process bypasses the need for pre-functionalized aldehydes or acids entirely. The core innovation lies in the generation of a specific aryl lithium intermediate via reaction with lithium diisopropylamide (LDA) at controlled low temperatures, followed immediately by quenching with inexpensive and safe acetylation reagents such as acetic anhydride or ethyl acetate. This strategic shift eliminates the need for toxic ethyl difluoroacetate and avoids the generation of hazardous acyl chlorides, fundamentally simplifying the operational workflow. The result is a concise two-step sequence that delivers the target ketone with exceptional purity exceeding 99.0% and yields surpassing 80%. This novel approach not only drastically simplifies the synthetic logic but also aligns perfectly with green chemistry principles by reducing waste and utilizing safer reagents, thereby offering a compelling solution for cost reduction in API manufacturing.
Mechanistic Insights into Directed Ortho-Lithiation and Acylation
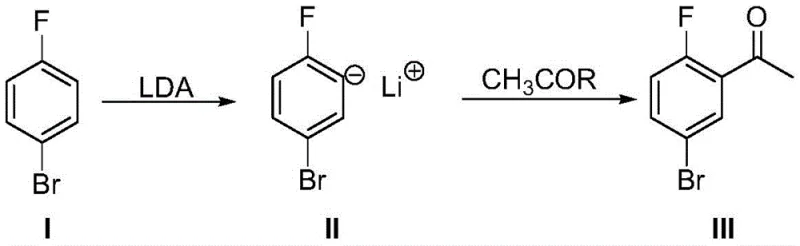
The success of this synthetic strategy hinges on the precise control of regioselectivity during the lithiation step. When 4-bromofluorobenzene is treated with a strong base like LDA in an inert solvent such as tetrahydrofuran or ether at temperatures ranging from -90°C to -40°C, the fluorine atom acts as a powerful directing group. This phenomenon, known as Directed Ortho Metalation (DoM), ensures that the lithiation occurs specifically at the position ortho to the fluorine atom, generating the desired aryl lithium species (Intermediate II) rather than attacking other positions on the aromatic ring or undergoing halogen-metal exchange at the bromine site. The choice of solvent and temperature is critical here; ether solvents stabilize the lithium cation, while the low temperature suppresses side reactions such as nucleophilic aromatic substitution or decomposition of the sensitive organolithium intermediate. This high degree of regiocontrol is what allows the process to proceed with such high fidelity, minimizing the formation of isomeric byproducts that would otherwise complicate downstream purification and reduce overall yield.
Following the formation of the aryl lithium intermediate, the subsequent acylation step is equally critical for maintaining product quality. The nucleophilic aryl lithium species attacks the carbonyl carbon of the acetylation reagent—preferably acetic anhydride or an ester like ethyl acetate—to form a tetrahedral intermediate which collapses to release the ketone product upon acidic workup. The patent specifies that the acetylation reagent should be added in a molar ratio of 1:1 to 10:1 relative to the starting material, ensuring complete consumption of the reactive organolithium species. The quenching process, involving the addition of a protonic acid such as acetic acid or dilute hydrochloric acid, is designed to safely neutralize excess base and decompose any remaining organometallic species without degrading the sensitive ketone product. This mechanistic understanding underscores why the process achieves such high purity; by avoiding harsh oxidation states and utilizing mild acetylation conditions, the formation of colored impurities and polymeric byproducts is effectively suppressed, resulting in a crude product that requires minimal refinement to meet stringent pharmaceutical specifications.
How to Synthesize 2-Fluoro-5-Bromoacetophenone Efficiently
The implementation of this lithiation-acetylation protocol requires careful attention to thermal management and reagent addition rates to ensure safety and reproducibility. The process begins with the preparation of the aryl lithium intermediate under an inert atmosphere, typically nitrogen, to prevent moisture ingress which would destroy the reactive species. Once the intermediate is formed and cooled to the appropriate range of -70°C to -100°C, it is transferred to the acetylation vessel containing the pre-cooled ester or anhydride. Detailed standard operating procedures regarding specific addition rates, stirring speeds, and workup protocols are essential for transferring this chemistry from the laboratory bench to pilot plant reactors. For a comprehensive guide on the exact experimental parameters, including solvent volumes and specific temperature ramps observed in the patent examples, please refer to the standardized synthesis steps outlined below.
- React 4-bromofluorobenzene with lithium diisopropylamide (LDA) in an inert solvent at low temperature (-90 to -40°C) to generate the aryl lithium intermediate.
- React the obtained aryl lithium intermediate with an acetylation reagent (such as acetic anhydride or ethyl acetate) at low temperature (-120 to -60°C).
- Quench the reaction mixture with a protonic acid or aqueous solution, separate the organic phase, and purify via distillation to obtain the final product with >99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers tangible strategic benefits that extend far beyond simple chemical yield. By shifting away from multi-step sequences that rely on scarce or hazardous precursors, manufacturers can significantly de-risk their supply chains and improve the predictability of delivery schedules. The elimination of toxic reagents like ethyl difluoroacetate not only lowers the cost of goods sold by reducing raw material expenses but also diminishes the regulatory burden associated with handling and disposing of hazardous chemicals. Furthermore, the simplicity of the two-step process enhances the overall throughput of production facilities, allowing for faster turnaround times and greater flexibility in responding to market demand fluctuations. This operational agility is crucial in the fast-paced pharmaceutical sector, where delays in intermediate supply can stall entire drug development programs.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the substitution of expensive, specialized reagents with commodity chemicals. By utilizing 4-bromofluorobenzene and common acetylating agents like ethyl acetate or acetic anhydride, the raw material cost profile is drastically improved compared to routes requiring ethyl difluoroacetate or activated acid chlorides. Additionally, the reduction in reaction steps from four or more down to just two significantly lowers utility consumption, labor hours, and equipment occupancy time. The high yield and purity achieved directly translate to less material loss during purification, further enhancing the economic efficiency of the process and delivering substantial cost savings for the final API manufacturer.
- Enhanced Supply Chain Reliability: Reliability is bolstered by the use of widely available starting materials that are not subject to the same supply constraints as niche fluorinated esters. 4-Bromofluorobenzene is a bulk chemical produced by multiple global suppliers, ensuring a stable and competitive sourcing environment. The robustness of the reaction conditions, which tolerate standard industrial solvents like THF and ethers, means that the process can be easily replicated across different manufacturing sites without requiring specialized infrastructure. This geographic flexibility reduces the risk of supply disruption due to regional logistical issues or single-source dependency, providing procurement teams with greater leverage and security in their vendor negotiations.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this route is inherently superior. The avoidance of acyl chlorides eliminates the generation of corrosive hydrochloric acid gas and the need for complex scrubbing systems, simplifying waste treatment protocols. The use of milder acetylation reagents reduces the potential for runaway exotherms, making the process safer to scale up to multi-ton quantities. Moreover, the high selectivity of the reaction minimizes the formation of difficult-to-remove impurities, reducing the volume of solvent required for recrystallization or chromatography. These factors collectively contribute to a smaller environmental footprint and easier compliance with increasingly stringent global environmental regulations, facilitating smoother regulatory approvals for the final drug substance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of 2-fluoro-5-bromoacetophenone. These insights are derived directly from the technical specifications and comparative data presented in the patent literature, aiming to clarify the operational benefits and chemical rationale behind this advanced manufacturing method. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this intermediate into their existing production workflows or drug discovery pipelines.
Q: What are the key advantages of the new lithiation-acetylation route over traditional methods?
A: The new route significantly reduces reaction steps compared to traditional aldehyde or acid pathways, eliminates the need for expensive and toxic ethyl difluoroacetate, and utilizes readily available acetylation reagents, resulting in lower raw material costs and simplified operations suitable for industrial scale-up.
Q: What is the expected purity and yield of 2-fluoro-5-bromoacetophenone using this method?
A: According to the patent data, this synthetic method consistently achieves a product purity of greater than 99.0% and a synthesis yield exceeding 80%, ensuring high-quality material for downstream pharmaceutical applications.
Q: Why is 2-fluoro-5-bromoacetophenone critical for oncology drug development?
A: It serves as a pivotal building block for synthesizing 5-bromo-3-methyl-1H-indazole, a core structure found in potent protein kinase Akt1 inhibitors used in the treatment of cancers, cardiovascular diseases, and inflammation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Fluoro-5-Bromoacetophenone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising efficiencies of the lithiation-acetylation route can be fully realized at an industrial level. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical instrumentation. Whether you are developing next-generation kinase inhibitors or optimizing an existing supply chain, our facility is equipped to handle the precise temperature controls and inert atmosphere requirements necessary for this sophisticated chemistry.
We invite you to engage with our technical procurement team to discuss how this optimized synthetic route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic impact of switching to this more efficient methodology. We encourage potential partners to contact us directly to obtain specific COA data for our batch productions and to receive detailed route feasibility assessments tailored to your volume needs. Let us collaborate to secure a stable, cost-effective, and high-quality supply of this critical pharmaceutical intermediate for your future success.
