Scalable Synthesis of Elexacaftor Intermediates: A Technical Breakthrough for Global Supply Chains
The rapid advancement of cystic fibrosis treatments has placed immense pressure on the global supply chain for critical active pharmaceutical ingredients (APIs) and their precursors. Specifically, the demand for Elexacaftor (VX-445), a next-generation CFTR protein corrector, has necessitated robust and scalable synthetic routes for its key building blocks. Patent CN111943848B introduces a transformative preparation method for 3,3,3-trifluoro-2,2-dimethylpropane-1-ol, a vital intermediate in this therapeutic landscape. This technology addresses long-standing bottlenecks in fluorinated chemical synthesis by replacing hazardous, hard-to-scale protocols with a streamlined, three-step sequence that prioritizes safety and atom economy. For procurement leaders and R&D directors alike, understanding the nuances of this patent is essential for securing a reliable pharmaceutical intermediates supplier capable of meeting the rigorous quality standards required for modern combination therapies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of gem-dimethyl trifluoro structures has been plagued by significant safety and operational challenges that hinder commercial viability. Prior art, such as the methodology disclosed in patent WO2008/147544A1, relies heavily on pyrophoric reagents like n-butyllithium and trimethylaluminum. These substances require stringent inert atmosphere conditions and specialized handling equipment, creating substantial safety hazards and inflating operational costs due to the need for extensive safety protocols. Furthermore, alternative routes described in patent CN109180459A utilize sodium cyanide, a highly toxic compound that poses severe environmental and worker safety risks, alongside requiring harsh reaction temperatures around 150 degrees Celsius. Another approach found in patent JP2016-104702A employs sulfur tetrafluoride and hydrofluoric acid, which are corrosive gases demanding special stainless steel equipment and resulting in poor yields of approximately 20 percent. These legacy methods create fragile supply chains vulnerable to regulatory scrutiny and production delays.

The Novel Approach
In stark contrast, the novel approach outlined in CN111943848B circumvents these dangers by utilizing readily available 3,3,3-trifluoropropionic acid as the starting material. This strategy eliminates the need for cryogenic conditions, pyrophoric organometallics, and toxic cyanides, fundamentally shifting the risk profile of the manufacturing process. By employing standard condensing reagents and mild alkylation conditions, the new method achieves high purity and yield without the burden of specialized containment systems. This shift not only enhances workplace safety but also drastically simplifies the engineering requirements for scale-up, making it an ideal candidate for cost reduction in pharmaceutical intermediates manufacturing. The ability to use conventional reaction equipment means that production capacity can be expanded rapidly to meet market demand without the lead time associated with installing exotic corrosion-resistant infrastructure.
Mechanistic Insights into the Three-Step Cascade Synthesis
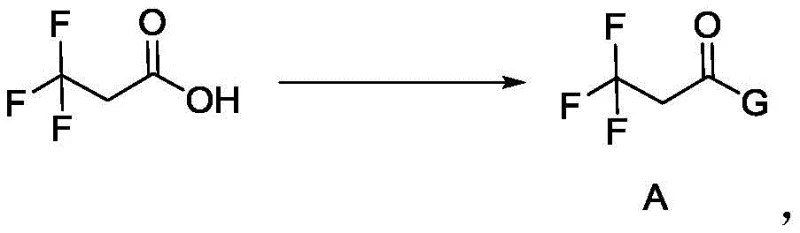
The core innovation lies in a logical three-step cascade that builds molecular complexity while maintaining functional group tolerance. The first stage involves a protection strategy where 3,3,3-trifluoropropionic acid reacts with a protecting agent—such as an alcohol, phenol, or amine—in the presence of a condensing reagent like DCC, EDCI, or HATU. This esterification or amidation step generates Intermediate A, effectively masking the carboxylic acid functionality to prevent side reactions during subsequent alkylation. The choice of protecting group is versatile, ranging from cyclohexyl to substituted phenyl groups, allowing process chemists to optimize for crystallinity and purification ease. This flexibility is crucial for ensuring high-purity pharmaceutical intermediates, as it enables the removal of impurities before the critical carbon-carbon bond-forming step.
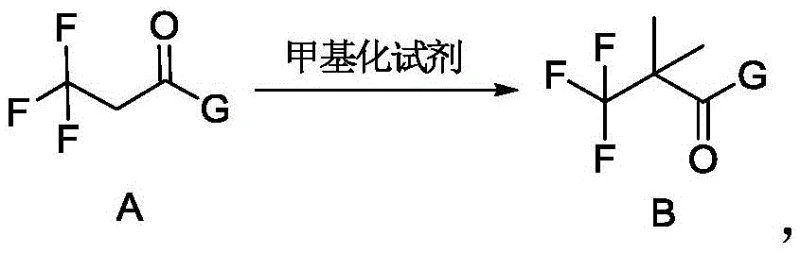
Following protection, the second stage executes a precise alpha-alkylation to install the gem-dimethyl motif. Intermediate A is treated with a methylating agent, such as methyl iodide or dimethyl sulfate, in the presence of a base like potassium carbonate or cesium carbonate. This step leverages the acidity of the alpha-protons adjacent to the electron-withdrawing trifluoromethyl and carbonyl groups. The reaction proceeds under reflux conditions in solvents like acetone or acetonitrile, delivering Intermediate B with excellent conversion rates. The mechanistic efficiency here is paramount; by avoiding strong, non-selective bases, the process minimizes elimination side products and ensures that the quaternary carbon center is formed cleanly. This precision directly translates to reduced downstream purification burdens and higher overall material throughput.
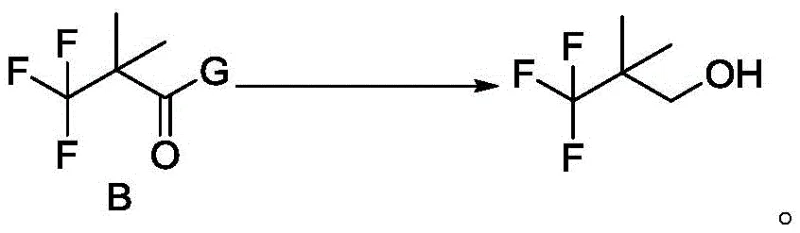
The final transformation involves the reduction of the protected Intermediate B to the target alcohol. Using reducing agents such as sodium borohydride, lithium aluminum hydride, or Red-Al, the ester or amide linkage is cleaved to reveal the primary alcohol functionality of 3,3,3-trifluoro-2,2-dimethylpropane-1-ol. This step is conducted under mild thermal conditions, often between 0 to 10 degrees Celsius initially, warming to room temperature to complete the reaction. The workup procedure is straightforward, typically involving aqueous quenching and organic extraction, which facilitates easy isolation of the product via distillation. The robustness of this reduction step ensures that the sensitive trifluoromethyl group remains intact while the desired alcohol is produced in high purity, ready for coupling into the final Elexacaftor API structure.
How to Synthesize 3,3,3-Trifluoro-2,2-Dimethylpropane-1-Ol Efficiently
Implementing this synthesis route requires careful attention to reagent stoichiometry and temperature control to maximize the reported molar yields of over 90 percent in each step. The process begins with the activation of the acid followed by sequential functionalization, offering a clear path from commodity chemicals to high-value intermediates. For technical teams looking to replicate or license this technology, the standardized nature of the reagents means that validation can be performed quickly in existing pilot plants. The detailed standardized synthesis steps see the guide below for specific operational parameters.
- Step 1: Protection - React 3,3,3-trifluoropropionic acid with a protecting agent (alcohol, phenol, or amine) using a condensing reagent like DCC or EDCI to form Intermediate A.
- Step 2: Methylation - Treat Intermediate A with a methylating agent (e.g., methyl iodide) and a base (e.g., potassium carbonate) to introduce gem-dimethyl groups, yielding Intermediate B.
- Step 3: Reduction - Reduce Intermediate B using a reducing agent such as sodium borohydride or Red-Al to obtain the final 3,3,3-trifluoro-2,2-dimethylpropane-1-ol.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patent offers compelling advantages that directly address the pain points of modern pharmaceutical sourcing. By eliminating the reliance on scarce or dangerous reagents, the supply chain becomes more resilient and less prone to disruption from regulatory changes or raw material shortages. The use of common solvents and bases means that procurement teams can source materials from multiple vendors, fostering competition and driving down input costs. Furthermore, the high yields reported in the examples suggest a significant reduction in waste generation, which aligns with increasingly strict environmental compliance standards and reduces disposal costs.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents like n-butyllithium and sulfur tetrafluoride removes the need for specialized storage and handling infrastructure. This simplification allows for the use of standard glass-lined or stainless steel reactors, significantly lowering capital expenditure and operational overhead. Additionally, the high atom economy and minimal byproduct formation reduce the load on waste treatment facilities, contributing to substantial cost savings in overall production.
- Enhanced Supply Chain Reliability: Because the starting material, 3,3,3-trifluoropropionic acid, is commercially available and the reagents are commodity chemicals, the risk of supply interruption is minimized. Unlike processes dependent on custom-synthesized precursors or tightly controlled substances, this route ensures a steady flow of materials. This reliability is critical for maintaining continuous production schedules and meeting the just-in-time delivery expectations of global pharmaceutical clients.
- Scalability and Environmental Compliance: The absence of highly corrosive gases and pyrophoric liquids makes this process inherently safer to scale from kilogram to tonne quantities. The mild reaction conditions reduce energy consumption for heating and cooling, supporting sustainability goals. Moreover, the simplified workup procedures minimize solvent usage and waste volume, facilitating easier compliance with environmental regulations and reducing the carbon footprint of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making.
Q: Why is this new synthesis route safer than prior art methods?
A: Unlike previous methods requiring hazardous reagents like n-butyllithium, trimethylaluminum, or sodium cyanide, this patent utilizes standard condensing agents and mild reducing conditions, significantly lowering explosion and toxicity risks.
Q: What are the typical yields for this three-step process?
A: Experimental data in the patent demonstrates consistently high molar yields across all steps, with individual step yields ranging from 92% to 98%, ensuring efficient material throughput.
Q: Can this process be scaled for industrial production?
A: Yes, the process avoids special pressure vessels and corrosive gases like sulfur tetrafluoride, allowing it to be conducted in standard stainless steel reactors suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,3,3-Trifluoro-2,2-Dimethylpropane-1-Ol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving medications like Elexacaftor. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3,3,3-trifluoro-2,2-dimethylpropane-1-ol meets the exacting standards required for API synthesis. Our commitment to technical excellence means we can navigate the complexities of fluorinated chemistry with precision and reliability.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your long-term strategic goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
