Advanced On-DNA Diamine Cyclization for High-Throughput Drug Discovery Libraries
Advanced On-DNA Diamine Cyclization for High-Throughput Drug Discovery Libraries
The pharmaceutical industry is currently witnessing a paradigm shift in lead compound identification, driven largely by the exponential growth of DNA-encoded library (DEL) technologies. Within this high-stakes environment, Patent CN113089104B emerges as a critical technical resource, detailing a robust synthetic method for the ring-closing reaction of On-DNA diamine compounds. This innovation addresses the perennial challenge of maintaining DNA integrity while performing complex organic transformations, specifically utilizing cyanogen bromide to construct nitrogen heterocyclic frameworks directly on the DNA tag. For R&D directors and procurement specialists alike, this patent represents a significant leap forward in expanding the chemical diversity of screening libraries without compromising the stability of the encoding molecule. The methodology described offers a streamlined pathway to generate high-purity On-DNA nitrogen heterocyclic compounds, which are essential scaffolds in modern medicinal chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of nitrogen heterocycles on DNA scaffolds has been plagued by severe compatibility issues between organic synthesis reagents and the fragile phosphodiester backbone of DNA. Conventional cyclization methods often rely on harsh organic solvents, strong acids, or high temperatures that can lead to DNA depurination, strand breakage, or loss of the encoding barcode. Furthermore, many standard protocols require transition metal catalysts that are difficult to remove completely, leading to contamination that interferes with downstream biological screening assays. The inability to efficiently close rings on diamine precursors in aqueous environments has historically limited the structural diversity of DELs, forcing chemists to rely on simpler, less pharmacologically relevant building blocks. This bottleneck restricts the chemical space available for high-throughput screening, potentially causing promising drug candidates to be overlooked due to library limitations.
The Novel Approach
In stark contrast to these legacy challenges, the novel approach outlined in the patent utilizes a mild, two-step aqueous buffer system that preserves DNA integrity while achieving high conversion rates. By employing cyanogen bromide as the cyclization agent in a controlled phosphate and borate buffer environment, the method successfully constructs benzimidazole and related nitrogen heterocyclic cores directly on the DNA tag. 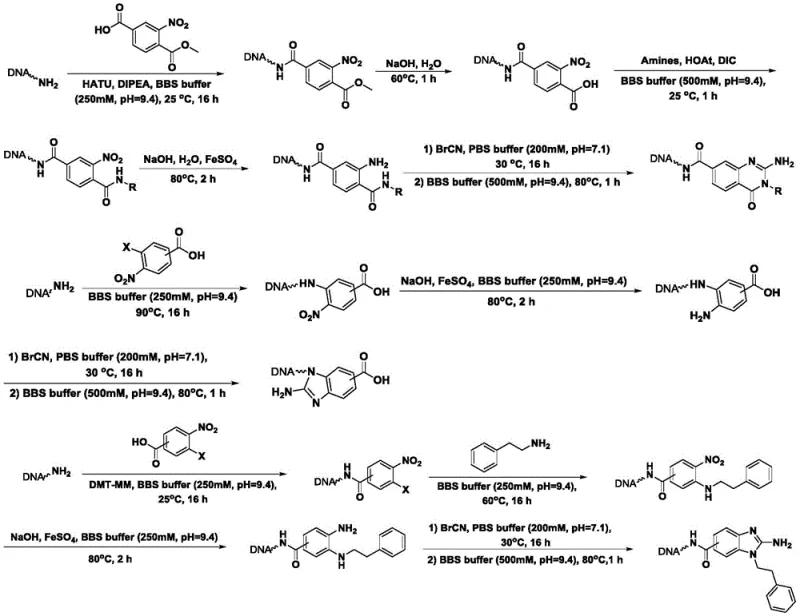 This strategy eliminates the need for toxic organic solvents and expensive transition metal catalysts, thereby simplifying the purification process and reducing the risk of sample loss. The protocol demonstrates remarkable versatility, accommodating a wide range of substituents on the aromatic ring, which allows for the rapid generation of diverse library members. This breakthrough effectively decouples the harshness of organic synthesis from the delicacy of molecular biology, enabling the creation of richer, more drug-like compound libraries.
This strategy eliminates the need for toxic organic solvents and expensive transition metal catalysts, thereby simplifying the purification process and reducing the risk of sample loss. The protocol demonstrates remarkable versatility, accommodating a wide range of substituents on the aromatic ring, which allows for the rapid generation of diverse library members. This breakthrough effectively decouples the harshness of organic synthesis from the delicacy of molecular biology, enabling the creation of richer, more drug-like compound libraries.
Mechanistic Insights into Cyanogen Bromide-Mediated Cyclization
The core of this technological advancement lies in the precise mechanistic control of the nucleophilic attack and subsequent cyclization under physiological-like conditions. The reaction initiates with the nucleophilic attack of one of the amino groups on the On-DNA diamine substrate onto the electrophilic carbon of the cyanogen bromide. This step forms a reactive cyanamide intermediate, which is stabilized by the specific pH of the phosphate buffer (pH 7.1). The choice of pH is critical; it ensures that the amine is sufficiently nucleophilic to react while preventing the hydrolysis of the cyanogen bromide or the degradation of the DNA. Following the formation of the intermediate, the reaction conditions are shifted to a basic borate buffer (pH 9.4) at elevated temperatures (80°C). This change facilitates the intramolecular nucleophilic attack of the second amino group onto the nitrile carbon, driving the ring closure to form the stable five-membered nitrogen heterocycle. 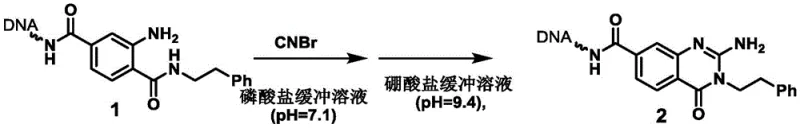 The careful modulation of pH and temperature between the two steps is the key to suppressing side reactions and ensuring high selectivity for the desired cyclic product over linear byproducts.
The careful modulation of pH and temperature between the two steps is the key to suppressing side reactions and ensuring high selectivity for the desired cyclic product over linear byproducts.
From an impurity control perspective, this mechanism offers distinct advantages over traditional thermal cyclizations. The use of excess cyanogen bromide (20-150 molar equivalents) drives the first step to completion, minimizing the presence of unreacted starting material. Furthermore, the aqueous nature of the reaction allows for simple ethanol precipitation to remove excess reagents and salts between steps, effectively purifying the intermediate before the final cyclization. This "clean-up" step is crucial for preventing the carryover of reactive species that could modify the DNA or other functional groups on the scaffold. The result is a product profile with a single dominant peak in LCMS analysis, indicating high chemical purity and structural fidelity. For quality control teams, this translates to reliable data where biological activity can be confidently attributed to the intended chemical structure rather than artifacts of synthesis.
How to Synthesize On-DNA Nitrogen Heterocycles Efficiently
The practical implementation of this synthesis route is designed for scalability and ease of operation, making it highly suitable for automated library production. The process begins by dissolving the On-DNA diamine compound in a phosphate buffer solution to achieve a specific molar concentration, typically around 1mM. Cyanogen bromide is then added in significant excess to ensure complete conversion of the starting material. The reaction mixture is incubated at a mild temperature of 30°C for approximately 16 hours, allowing the formation of the cyanamide intermediate without stressing the DNA linkage. Following this incubation, the product is precipitated using ethanol and sodium chloride, centrifuged, and freeze-dried to isolate the solid intermediate. This solid is then redissolved in a borate buffer solution and heated to 80°C for a short duration of 1 hour to effect the final ring closure. Detailed standardized synthesis steps are provided below for technical reference.
- Dissolve the On-DNA diamine substrate in a phosphate buffer solution (pH 7.1) and add 20-150 molar equivalents of cyanogen bromide, reacting at 30°C for 16 hours to form the intermediate.
- Precipitate the intermediate solid using ethanol and sodium chloride, then centrifuge and freeze-dry to isolate the crude product before the second step.
- Redissolve the solid in a borate buffer solution (pH 9.4) and heat at 80°C for 1 hour to complete the ring-closing cyclization, followed by final purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this aqueous cyclization technology offers substantial strategic benefits beyond mere technical feasibility. The elimination of organic solvents and transition metal catalysts drastically simplifies the waste management profile of the synthesis, aligning with increasingly stringent environmental regulations and corporate sustainability goals. By shifting to a water-based system, facilities can reduce the costs associated with solvent disposal and hazardous waste treatment, leading to significant operational expenditure savings. Moreover, the high conversion rates reported (up to 89%) mean that less starting material is wasted, optimizing the utilization of expensive DNA-tagged building blocks. This efficiency is critical when scaling up library production, where the cost of goods sold (COGS) is heavily influenced by the yield of each synthetic step.
- Cost Reduction in Manufacturing: The transition to an aqueous buffer system removes the dependency on high-grade anhydrous organic solvents, which are often costly and require specialized storage and handling infrastructure. Additionally, the absence of transition metal catalysts eliminates the need for expensive scavenging resins or complex chromatography steps to remove metal residues, which are mandatory for pharmaceutical grade intermediates. This streamlining of the downstream processing workflow results in a leaner manufacturing process with fewer unit operations. Consequently, the overall production cost per compound is significantly reduced, allowing for the economic synthesis of larger and more diverse libraries within the same budget constraints.
- Enhanced Supply Chain Reliability: The reagents required for this method, such as cyanogen bromide and common buffer salts, are commodity chemicals with stable and robust global supply chains. Unlike specialized organometallic catalysts that may suffer from supply shortages or long lead times, these materials are readily available from multiple vendors. This redundancy in sourcing mitigates the risk of production delays caused by raw material scarcity. Furthermore, the mild reaction conditions reduce the risk of batch failure due to equipment malfunction or temperature excursions, ensuring a consistent and reliable output of high-quality intermediates. This reliability is paramount for maintaining the continuity of drug discovery pipelines that depend on timely library delivery.
- Scalability and Environmental Compliance: The protocol is inherently scalable, having been demonstrated in multi-well plate formats which are the industry standard for high-throughput library synthesis. The use of water as the primary solvent makes the process safer to operate at scale, removing fire hazards associated with volatile organic compounds. From an environmental compliance standpoint, the aqueous waste streams are easier to treat and neutralize compared to halogenated solvent waste. This facilitates faster regulatory approvals for new manufacturing processes and reduces the environmental footprint of the facility. The ability to scale this green chemistry approach ensures that supply can meet the growing demand for DEL services without compromising on safety or sustainability standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this On-DNA cyclization technology. These insights are derived directly from the experimental data and specifications detailed in the patent documentation, providing a clear understanding of the method's capabilities and limitations. Understanding these nuances is essential for R&D teams planning to integrate this chemistry into their existing library design workflows. The answers reflect the robustness of the aqueous buffer system and its compatibility with a wide array of chemical functionalities.
Q: What are the critical reaction conditions for On-DNA diamine cyclization?
A: The process requires a two-step buffer system: an initial reaction in phosphate buffer (pH 7.1) at 30°C with excess cyanogen bromide, followed by a cyclization step in borate buffer (pH 9.4) at 80°C.
Q: Why is aqueous compatibility important for DNA-encoded library synthesis?
A: DNA tags are sensitive to organic solvents and extreme pH; using aqueous buffers ensures the integrity of the genetic barcode while allowing diverse chemical transformations.
Q: What conversion rates can be achieved with this cyanogen bromide method?
A: Experimental data indicates conversion rates up to 89% for specific substrates, with broad applicability across 29 different diamine structures tested in the patent examples.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable On-DNA Diamine Supplier
The successful execution of complex On-DNA synthesis requires not only advanced intellectual property but also deep practical expertise in process optimization and quality control. NINGBO INNO PHARMCHEM stands at the forefront of this field, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the unique requirements of DNA-compatible chemistry, ensuring that every batch meets stringent purity specifications. We understand that the integrity of the DNA tag is as critical as the chemical structure of the small molecule, and our rigorous QC labs employ advanced analytical techniques to verify both. By partnering with us, you gain access to a supply chain that is optimized for both speed and precision, capable of delivering the high-purity On-DNA nitrogen heterocyclic compounds necessary for next-generation drug discovery.
We invite procurement leaders and R&D directors to engage with our technical team to explore how this patented cyclization method can be integrated into your specific project needs. Whether you are looking to expand an existing library or initiate a new screening campaign, our experts can provide a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your target scaffolds. Let us help you accelerate your lead identification process with reliable, high-quality intermediates that drive innovation.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
