Advanced Solvent-Free Catalytic Route for High-Purity 2-Aminothiazole Manufacturing
Introduction to Next-Generation 2-Aminothiazole Synthesis
The pharmaceutical and fine chemical industries are constantly seeking more efficient, sustainable, and cost-effective pathways for producing critical heterocyclic intermediates. A significant breakthrough in this domain is detailed in patent CN114853692B, which discloses a novel preparation method for 2-aminothiazole, a versatile building block widely utilized in the synthesis of sulfonamide antibiotics, antifungal agents, and even as a specialized additive in photoresist formulations for the electronics sector. This technology represents a paradigm shift from traditional solvent-dependent processes to a sophisticated solvent-free one-pot methodology. By leveraging a unique boric acid-borate composite catalyst system, this invention successfully overcomes the longstanding challenges of low yield, poor purity, and undesirable product coloration that have plagued conventional manufacturing techniques. For global procurement leaders and R&D directors, understanding this technological leap is essential for securing a reliable pharma intermediate supplier capable of delivering high-specification materials at a competitive operational baseline.
The strategic importance of 2-aminothiazole cannot be overstated, given its pervasive role in medicinal chemistry and advanced material science. However, the historical reliance on aqueous or alcoholic solvent systems has introduced significant inefficiencies, including extensive wastewater treatment requirements and energy-intensive solvent recovery steps. The innovation described in CN114853692B addresses these pain points directly by enabling a heterogeneous reaction between thiourea and chloroacetaldehyde acetal under mild thermal conditions. This approach not only enhances the environmental profile of the manufacturing process but also drastically simplifies the downstream processing workflow. As we delve deeper into the technical specifics, it becomes evident that this method offers a robust solution for the commercial scale-up of complex heterocycles, ensuring supply chain continuity and superior product quality for downstream applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 2-aminothiazole has relied heavily on the reaction of thiourea with chloroacetaldehyde in aqueous solutions or mixed solvent systems containing water and alcohol. These traditional methodologies are fraught with inherent chemical and engineering deficiencies that compromise both economic efficiency and product integrity. A primary drawback is the intense exothermic nature of the reaction in aqueous media, which necessitates slow, controlled dripping of reagents to manage heat release, thereby extending batch cycle times and limiting reactor throughput. Furthermore, the presence of water and high local temperatures often promotes the hydrolysis and decomposition of thiourea, leading to the formation of complex, colored byproducts that manifest as brown or yellow impurities in the final crude material. To mitigate these issues, manufacturers are forced to employ multiple recrystallization steps or high-vacuum sublimation, which significantly erodes overall yield and escalates production costs. Additionally, the use of volatile organic solvents introduces substantial safety risks and environmental compliance burdens related to solvent recovery and VOC emissions, making these legacy processes increasingly untenable in a modern regulatory landscape.
The Novel Approach
In stark contrast to these legacy limitations, the novel approach outlined in the patent utilizes a solvent-free one-pot strategy driven by a boric acid-borate composite catalyst. This method fundamentally alters the reaction environment by eliminating the solvent medium entirely, thereby preventing the hydrolytic degradation of thiourea that typically occurs in water phases. The reaction is conducted at a controlled temperature range of 90-110°C, where the composite catalyst facilitates a heterogeneous interaction between the solid thiourea and the liquid chloroacetaldehyde acetal. This unique catalytic system not only accelerates the reaction kinetics but also ensures a mild thermal profile with no obvious exothermic heat release, enhancing operational safety and controllability. The result is a dramatic improvement in process metrics, with yields soaring to over 95% and purity levels reaching 97-99%, compared to the 75-80% yields and 80-90% purity typical of older methods. Moreover, the elimination of solvents removes the need for energy-intensive distillation columns for solvent recovery, directly contributing to cost reduction in fine chemical manufacturing and aligning with green chemistry principles.
Mechanistic Insights into Boric Acid-Borate Composite Catalysis
The efficacy of this synthesis hinges on the sophisticated dual-function mechanism of the boric acid-borate composite catalyst, which orchestrates the reaction through both electrophilic activation and physical phase modification. In this system, boric acid serves as a potent electrophilic catalyst, activating the carbonyl equivalent of the chloroacetaldehyde acetal to facilitate nucleophilic attack by the thiourea. Simultaneously, the borate component, such as sodium tetraborate, acts as a fluxing agent that significantly lowers the melting point of the thiourea. This fluxing action allows the thiourea to exist in a molten state under the reaction conditions, creating a liquid-liquid heterogeneous interface that maximizes contact area between reactants without the need for an external solvent. As the reaction progresses and byproduct alcohol is generated, the system eventually transitions towards a more homogeneous phase, driving the reaction to completion. This intricate balance between heterogeneous initiation and homogeneous completion is key to suppressing side reactions and achieving the high selectivity observed in the patent data.
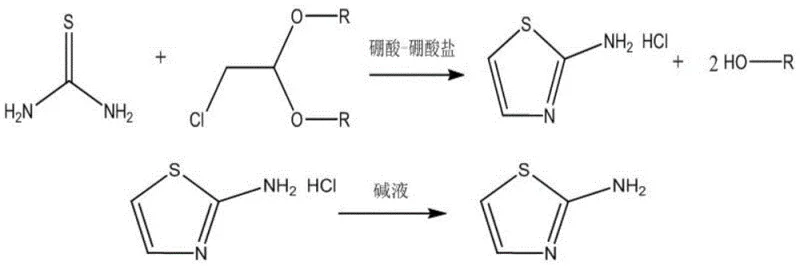
Beyond the catalytic activation, the process incorporates a critical purification mechanism centered on precise pH control during the workup phase. The patent specifies adjusting the pH to a narrow window of 9-10 using a mild alkali such as ammonia water. This specific pH range is vital because a pH that is too low results in incomplete precipitation of the 2-aminothiazole, leading to yield losses in the mother liquor. Conversely, a pH that is too high (over-basic) can cause the precipitated product to discolor or even degrade in the mother liquor, compromising the prized white appearance of the final crystal. By maintaining the pH between 9 and 10 and precipitating at low temperatures (0-10°C), the process ensures maximum crystallization efficiency while preserving the chemical integrity and optical purity of the product. This level of control effectively eliminates the need for subsequent decolorization steps, further streamlining the production workflow and reducing waste generation.
How to Synthesize 2-Aminothiazole Efficiently
Implementing this advanced synthesis route requires careful attention to catalyst preparation and thermal management to fully realize the benefits of the solvent-free protocol. The process begins with the mechanical grinding of boric acid and sodium tetraborate to create a homogeneous composite catalyst powder, which is then mixed with thiourea and chloroacetaldehyde acetal in a standard reactor. The reaction mixture is heated to 90-110°C and maintained for 6-8 hours, allowing the cyclization to proceed to completion without the violent exotherms associated with aqueous methods. Following the reaction, the byproduct alcohol is removed via reduced pressure distillation, and the residue is subjected to a controlled neutralization and crystallization sequence. The detailed standardized synthetic steps, including specific molar ratios and equipment specifications required for GMP-compliant production, are outlined below for technical reference.
- Prepare the composite catalyst by grinding boric acid and sodium tetraborate (molar ratio 1: 1 to 1:1.5) into a fine powder to ensure homogeneity.
- Mix thiourea, chloroacetaldehyde acetal, and the catalyst in a reactor, then heat to 90-110°C for 6-8 hours under solvent-free conditions to initiate the heterogeneous reaction.
- Distill off byproduct alcohol under reduced pressure, adjust pH to 9-10 using ammonia water at 0-10°C to precipitate white crystals, and dry under vacuum.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this solvent-free catalytic technology translates into tangible strategic advantages that extend far beyond simple yield improvements. The elimination of organic solvents from the reaction matrix fundamentally reshapes the cost structure of production by removing the capital and operational expenses associated with solvent storage, recovery, and disposal infrastructure. This simplification of the process flow reduces the complexity of the manufacturing plant, lowering maintenance costs and minimizing the risk of production downtime due to solvent handling issues. Furthermore, the ability to produce a white, high-purity product directly from the crystallization step eliminates the need for costly and time-consuming recrystallization or sublimation purification stages. This reduction in downstream processing steps significantly shortens the overall batch cycle time, enhancing the agility of the supply chain and allowing for faster response to market demand fluctuations without compromising on quality standards.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, primarily driven by the complete removal of solvent-related costs. In traditional processes, a significant portion of the operating budget is consumed by the purchase of solvents, the energy required to distill and recover them, and the environmental fees associated with their disposal. By operating under solvent-free conditions, the process eradicates these variable costs entirely. Additionally, the high selectivity of the boric acid-borate catalyst minimizes the formation of byproducts, which means that raw material utilization is maximized. The reduction in thiourea dosage, as noted in the patent, further contributes to raw material savings. When combined with the energy savings from avoiding solvent recovery and multi-step purification, the overall cost of goods sold (COGS) is substantially reduced, offering a highly competitive pricing structure for bulk purchasers.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the inherent safety and simplicity of the new method. Traditional solvent-based reactions often face regulatory scrutiny and logistical hurdles regarding the transport and storage of flammable liquids. The solvent-free nature of this process mitigates these risks, ensuring smoother regulatory compliance and fewer interruptions due to safety audits or environmental incidents. Moreover, the use of readily available and stable raw materials like boric acid and sodium tetraborate ensures that the supply of catalyst components is secure and不受 geopolitical volatility. The robustness of the reaction conditions, which do not require extreme pressures or cryogenic temperatures, also means that the process can be easily replicated across different manufacturing sites, providing redundancy and flexibility in the global supply network.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces unforeseen challenges, particularly regarding heat transfer and mixing efficiency in large vessels. The mild thermal profile of this solvent-free reaction, characterized by the absence of intense exothermic heat release, makes it exceptionally amenable to scale-up from pilot plants to multi-ton commercial reactors. The lack of solvent vapor pressure issues simplifies reactor design and operation at scale. From an environmental perspective, the process aligns perfectly with increasingly stringent global regulations on VOC emissions and wastewater discharge. By generating minimal waste and avoiding the release of volatile organics, manufacturers can achieve significant sustainability goals, enhancing their corporate social responsibility profile and meeting the green procurement criteria of major multinational pharmaceutical clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this novel 2-aminothiazole synthesis method. These insights are derived directly from the experimental data and comparative examples provided in the patent literature, offering a transparent view of the technology's capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing supply chains or for quality assurance professionals assessing the consistency of the final product specifications.
Q: Why is the boric acid-borate composite catalyst critical for this synthesis?
A: The composite catalyst serves a dual function: boric acid acts as an electrophilic catalyst to promote cyclization, while the borate component acts as a fluxing agent that lowers the melting point of thiourea, enabling a efficient heterogeneous reaction without solvents.
Q: How does this method improve product color compared to traditional aqueous methods?
A: Traditional aqueous methods often result in brown-yellow products due to intense exothermic reactions and thiourea decomposition. This solvent-free process controls reaction heat effectively and utilizes precise pH regulation (9-10) during precipitation to prevent discoloration, yielding a pure white product.
Q: What are the scalability advantages of eliminating organic solvents?
A: Eliminating solvents removes the need for complex solvent recovery systems and reduces fire hazards associated with volatile organics. This simplifies the equipment requirements, lowers CAPEX for large-scale reactors, and significantly streamlines the downstream purification process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Aminothiazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced manufacturing technologies like the solvent-free boric acid-borate catalytic system is key to maintaining competitiveness in the global fine chemical market. As a leading CDMO partner, we possess the technical expertise and infrastructure to seamlessly adopt and optimize such innovative pathways for our clients. Our facilities are equipped to handle diverse synthetic routes, and we have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering 2-aminothiazole that meets stringent purity specifications, utilizing our rigorous QC labs to ensure every batch complies with the highest international standards for pharmaceutical and electronic grade intermediates. Our dedication to process excellence ensures that we can consistently provide the high-purity 2-aminothiazole required for your most demanding applications.
We invite you to collaborate with us to leverage these technological advancements for your specific project needs. Our team is ready to provide a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this greener, more efficient synthesis route for your supply chain. We encourage you to contact our technical procurement team today to request specific COA data from our recent batches and to discuss route feasibility assessments tailored to your volume requirements. By partnering with NINGBO INNO PHARMCHEM, you secure not just a supplier, but a strategic ally dedicated to driving value and innovation in your chemical sourcing strategy.
