Advanced Metal-Free Synthesis of 3-Selenoindole Derivatives for Commercial Scale-Up
Advanced Metal-Free Synthesis of 3-Selenoindole Derivatives for Commercial Scale-Up
The landscape of heterocyclic chemistry is continuously evolving, driven by the demand for more efficient and environmentally benign synthetic routes for bioactive scaffolds. A significant breakthrough in this domain is detailed in patent CN111848488A, which discloses a novel methodology for the synthesis of 3-selenoindole derivatives. These compounds are of paramount interest in medicinal chemistry due to their potent antiproliferative and antitumor activities, making them valuable candidates for drug discovery pipelines. The disclosed method utilizes a metal-free approach, employing 2-styrylaniline derivatives and organic diselenides as raw materials in the presence of a non-metallic additive, specifically N-fluorobisbenzenesulfonamide (NFSI). This one-pot cyclization strategy represents a substantial leap forward in process chemistry, offering a pathway that is not only operationally simple but also highly selective and universally applicable across a wide range of substrates.

For procurement managers and supply chain directors seeking a reliable pharmaceutical intermediate supplier, the implications of this technology are profound. By shifting away from traditional metal-catalyzed processes, manufacturers can achieve significant cost reduction in fine chemical manufacturing while ensuring the production of high-purity 3-selenoindole derivatives that meet stringent regulatory standards. The elimination of transition metals simplifies the downstream purification process, directly impacting the bottom line by reducing waste treatment costs and shortening production cycles. Furthermore, the robustness of this reaction under air atmosphere removes the capital expenditure associated with maintaining inert gas systems, making it an ideal candidate for the commercial scale-up of complex heterocycles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-selenoindoles has relied heavily on the direct selenylation of pre-existing indole frameworks or the cyclization of 2-alkynylaniline derivatives. While effective in laboratory settings, these conventional methods suffer from significant drawbacks when translated to an industrial context. Direct selenylation often requires expensive, pre-functionalized indole starting materials, which limits the structural diversity and increases the overall cost of goods. Moreover, many of these protocols necessitate the use of stoichiometric amounts of strong bases or harsh reaction conditions, such as dry solvents and prolonged reaction times under inert atmospheres. The reliance on transition metal catalysts, while common, introduces a critical bottleneck: the removal of trace metal residues to meet pharmaceutical purity specifications is a resource-intensive process that often involves specialized scavengers and multiple purification steps, thereby eroding profit margins and extending lead times.
The Novel Approach
In stark contrast, the methodology described in patent CN111848488A leverages readily available 2-styrylaniline derivatives as starting materials, which are generally more accessible and cost-effective than their alkyne counterparts. The core innovation lies in the use of NFSI as a non-metallic additive to activate the diselenide species, facilitating a tandem cyclization and selenylation sequence in a single pot. This approach operates under mild conditions, typically at temperatures between 100-120°C, and crucially, it proceeds efficiently under ambient air without the need for moisture exclusion. The reaction demonstrates exceptional functional group tolerance, accommodating both electron-donating and electron-withdrawing substituents on the aromatic rings with high yields, often exceeding 90%. This versatility allows for the rapid generation of diverse chemical libraries, accelerating the drug discovery process while maintaining a lean and agile manufacturing footprint.
Mechanistic Insights into NFSI-Mediated Electrophilic Cyclization
Understanding the underlying mechanism is crucial for R&D directors evaluating the feasibility of this route for large-scale production. The reaction initiates with the interaction between diphenyl diselenide and NFSI, generating a highly reactive electrophilic selenium species, denoted as PhSeX (where X is F or the sulfonamide group). This electrophile attacks the carbon-carbon double bond of the 2-styrylaniline substrate, forming a cyclic selenium ion intermediate. Subsequently, the intramolecular nucleophilic attack by the amino group triggers the opening of the three-membered ring, leading to the formation of a key indoline intermediate. The presence of pyridine as a solvent plays a dual role: it acts as a base to facilitate proton elimination and assists in the removal of the selenium moiety to regenerate the electrophilic species, ultimately yielding the 2-phenylindole intermediate before the final electrophilic addition forms the target 3-selenoindole product.
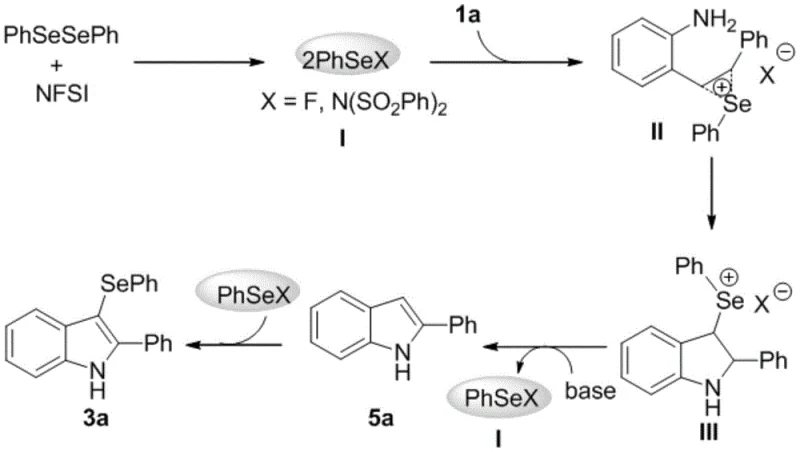
This mechanistic pathway offers distinct advantages regarding impurity control and process safety. Because the reaction does not involve radical chains or unstable organometallic intermediates, the risk of exothermic runaways is minimized, enhancing operational safety. Furthermore, the regeneration of the electrophilic selenium species suggests a catalytic-like efficiency in the activation step, although stoichiometric NFSI is used to drive the equilibrium. The absence of metal catalysts means that the impurity profile is significantly cleaner, devoid of heavy metal contaminants that often complicate regulatory filings. For process chemists, this translates to a more predictable crystallization behavior and simpler isolation protocols, ensuring that the final API intermediate consistently meets the rigorous quality thresholds required by global health authorities.
How to Synthesize 2-Phenyl-3-(phenylselenoyl) Indole Efficiently
The practical execution of this synthesis is designed for simplicity and reproducibility, making it highly attractive for contract development and manufacturing organizations (CDMOs). The standard protocol involves combining the 2-styrylaniline substrate and diphenyl diselenide in pyridine solvent, followed by the addition of NFSI. The mixture is then heated to approximately 110°C for 4 hours under air. Upon completion, the reaction is quenched with saturated sodium bicarbonate, and the product is extracted and purified via standard column chromatography. This straightforward workflow minimizes the need for specialized equipment or hazardous reagents. For a detailed breakdown of the standardized operating procedures and specific molar ratios optimized for different substrates, please refer to the technical guide below.
- Mix 2-styrylaniline derivative and organic diselenide in pyridine solvent with NFSI additive.
- Heat the reaction mixture to 110°C under air atmosphere for 4 hours to facilitate cyclization.
- Quench with saturated sodium bicarbonate, extract, dry, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this metal-free synthesis route offers compelling benefits that align with modern supply chain resilience goals. The primary advantage lies in the drastic simplification of the manufacturing process, which directly correlates to reduced operational expenditures. By eliminating the need for expensive transition metal catalysts and the associated purification infrastructure, manufacturers can achieve substantial cost savings without compromising on product quality. Additionally, the use of commodity chemicals like pyridine and commercially available diselenides ensures a stable supply chain, mitigating the risks associated with sourcing specialized or scarce reagents. The robustness of the reaction under air atmosphere further reduces dependency on utility-intensive inert gas systems, lowering the overall energy footprint of the production facility.
- Cost Reduction in Manufacturing: The exclusion of precious metal catalysts such as palladium or copper removes a significant cost driver from the bill of materials. More importantly, it obviates the need for costly metal scavenging resins and extensive analytical testing for residual metals, which are mandatory for pharmaceutical ingredients. This streamlined downstream processing results in a leaner cost structure, allowing for more competitive pricing in the global market while maintaining healthy margins for suppliers.
- Enhanced Supply Chain Reliability: The starting materials, specifically 2-styrylaniline derivatives and diaryl diselenides, are widely available from multiple chemical vendors, reducing the risk of single-source bottlenecks. The reaction's tolerance to air and moisture means that production is less susceptible to interruptions caused by utility failures or strict environmental controls. This inherent robustness ensures consistent batch-to-batch quality and reliable delivery schedules, which is critical for maintaining uninterrupted drug development timelines for our partners.
- Scalability and Environmental Compliance: The protocol has been validated for scalability, with patent data demonstrating successful gram-scale reactions yielding consistent results. The absence of toxic heavy metals simplifies waste management and disposal, aligning with increasingly stringent environmental regulations and green chemistry principles. This eco-friendly profile not only reduces compliance costs but also enhances the sustainability credentials of the final product, a factor that is becoming increasingly important for corporate social responsibility initiatives in the pharmaceutical sector.
Frequently Asked Questions (FAQ)
To address common technical inquiries regarding this synthesis platform, we have compiled answers based on the specific experimental data and mechanistic studies provided in the patent literature. These insights are intended to clarify the operational parameters and potential applications of this technology for prospective partners. Understanding these nuances is essential for evaluating the fit of this chemistry within your specific project requirements and for planning effective technology transfer strategies.
Q: What are the advantages of using NFSI over metal catalysts for selenylation?
A: Using N-fluorobisbenzenesulfonamide (NFSI) eliminates the need for expensive transition metal catalysts, thereby removing the costly and complex heavy metal removal steps required for pharmaceutical grade purity compliance.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the patent data confirms successful scale-up experiments where substrate loading was increased to 6 mmol with maintained high yields (85%), demonstrating robustness for commercial manufacturing.
Q: Does the reaction require inert gas protection?
A: No, a key advantage of this protocol is its insensitivity to air and moisture, allowing the reaction to proceed efficiently under ambient air atmosphere without the need for nitrogen or argon shielding.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Selenoindole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free cyclization technology for the next generation of selenium-containing therapeutics. As a dedicated CDMO partner, we possess the technical expertise and infrastructure to translate this innovative laboratory protocol into a robust, GMP-compliant manufacturing process. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from clinical trials to full-scale commercialization. We operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee stringent purity specifications for every batch of 3-selenoindole derivatives we produce, safeguarding the integrity of your drug development pipeline.
We invite you to collaborate with us to optimize your supply chain and accelerate your time-to-market. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. By leveraging our expertise in process optimization and scale-up engineering, we can help you navigate the complexities of selenium chemistry with confidence. Please contact us today to request specific COA data and comprehensive route feasibility assessments for your target molecules, and let us demonstrate how our advanced synthesis capabilities can drive value for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
