Advanced One-Pot Synthesis of Quinazoline Dione Intermediates for Commercial Agrochemical Production
The global agrochemical industry is constantly seeking safer and more efficient pathways to produce critical fungicide intermediates, and the recent disclosure in patent CN115490643A offers a transformative solution for the synthesis of 3-dichlorophenyl-6-fluoro-2,4(1H,3H)-quinazolinedione. This compound serves as a pivotal building block for Fluquinconazole, a broad-spectrum triazole fungicide widely used to protect crops such as wheat, barley, and grapes from devastating fungal diseases. Historically, the manufacturing of this quinazoline derivative has been plagued by significant safety hazards and operational complexities, primarily due to the reliance on extremely toxic reagents. The new methodology described in this patent introduces a robust one-pot synthetic strategy that replaces dangerous gaseous reagents with stable liquid alternatives, thereby addressing long-standing concerns regarding worker safety and environmental compliance. By shifting the chemical paradigm from phosgene-based chemistry to a phenyl chloroformate-mediated pathway, this innovation not only mitigates risk but also streamlines the production workflow. For stakeholders in the fine chemical sector, this represents a critical evolution in process chemistry that aligns with modern green manufacturing standards while maintaining high product quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for producing 3-dichlorophenyl-6-fluoro-2,4(1H,3H)-quinazolinedione have historically depended on the use of solid phosgene (triphosgene) and hydrogen chloride gas, creating a formidable barrier to safe and efficient manufacturing. The utilization of solid phosgene introduces severe safety risks, as it is a potent toxin that requires rigorous containment protocols and specialized handling equipment to prevent accidental release and exposure. Furthermore, the subsequent cyclization step typically necessitates the use of hydrogen chloride gas, which is highly corrosive and poses significant threats to respiratory health and equipment integrity. These hazardous conditions mandate the use of expensive, corrosion-resistant reactors and extensive scrubbing systems, driving up capital expenditure and operational costs. Additionally, the multi-step nature of the conventional process often involves isolating unstable intermediates, which can lead to yield losses and increased waste generation. The logistical challenges associated with transporting and storing such dangerous chemicals further complicate the supply chain, making the traditional method increasingly untenable for modern, sustainability-focused production facilities.
The Novel Approach
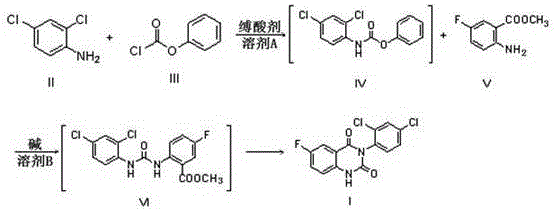
In stark contrast to the perilous legacy methods, the novel approach detailed in the patent employs a sophisticated one-pot technique that fundamentally redesigns the reaction landscape for superior safety and efficiency. This innovative route utilizes 2,4-dichloroaniline and phenyl chloroformate as the primary starting materials, reacting them in the presence of an organic solvent and an acid-binding agent to generate a stable carbamate intermediate in situ. By avoiding the generation of free isocyanates via phosgene, the process eliminates the most significant toxicity hazard associated with the synthesis. The subsequent addition of methyl 2-amino-5-fluorobenzoate and a base facilitates a seamless cyclization within the same reaction vessel, effectively merging what were previously distinct and hazardous steps into a unified operation. This consolidation not only reduces the physical footprint of the reaction but also minimizes solvent usage and waste disposal requirements. The result is a streamlined protocol that delivers high-purity product with impressive yields, demonstrating that safety enhancements do not come at the expense of chemical performance.
Mechanistic Insights into Phenyl Chloroformate-Mediated Cyclization
The core of this technological breakthrough lies in the precise mechanistic execution of the carbamate formation and subsequent intramolecular cyclization, which avoids the harsh conditions of the past. In the first stage, the nucleophilic attack of the amine group in 2,4-dichloroaniline on the carbonyl carbon of phenyl chloroformate is carefully controlled at low temperatures, typically between 0°C and 10°C, to ensure selective formation of the urethane linkage without side reactions. The presence of an acid-binding agent, such as pyridine or triethylamine, is crucial here to scavenge the generated hydrochloric acid, preventing the protonation of the amine and driving the equilibrium forward. This results in the formation of intermediate IV, a stable species that can be carried forward without isolation, preserving the integrity of the reactive functional groups. The choice of solvent in this step, such as toluene or 2-methyltetrahydrofuran, plays a vital role in solubilizing the reactants while maintaining a homogeneous reaction environment conducive to high conversion rates.
Following the formation of the carbamate intermediate, the reaction system undergoes a transformation driven by the addition of a strong base and the second aromatic component, methyl 2-amino-5-fluorobenzoate. The base deprotonates the amide nitrogen or activates the nucleophile, facilitating an attack on the carbonyl center of the carbamate moiety. This triggers a cascade of intramolecular rearrangements and eliminations, ultimately closing the quinazoline ring system to form the target diketone structure. The thermal conditions for this cyclization are remarkably mild, ranging from 50°C to 80°C, which stands in sharp contrast to the extreme conditions often required for phosgene-based cyclizations. This gentle thermal profile helps preserve the structural fidelity of the fluorine and chlorine substituents, ensuring that the final impurity profile is clean and manageable. The ability to control these mechanistic steps within a single pot underscores the elegance of the design, offering a reproducible pathway that is highly attractive for regulatory approval and commercial adoption.
How to Synthesize 3-dichlorophenyl-6-fluoro-2,4(1H,3H)-quinazolinedione Efficiently
To implement this advanced synthesis in a laboratory or pilot plant setting, operators must adhere to specific procedural guidelines that maximize yield and safety while minimizing waste. The process begins with the careful preparation of the reaction vessel under an inert atmosphere, followed by the sequential addition of solvents and reagents at controlled temperatures to manage exotherms. Detailed standard operating procedures regarding the stoichiometry of the acid-binding agents and the timing of the base addition are critical for success. For a comprehensive breakdown of the exact molar ratios, solvent volumes, and temperature ramps required to replicate the high yields reported in the patent, please refer to the standardized synthesis guide below.
- React 2,4-dichloroaniline with phenyl chloroformate in an organic solvent with an acid-binding agent at 0-10°C to form the carbamate intermediate.
- Concentrate the reaction mixture and add solvent, methyl 2-amino-5-fluorobenzoate, and a base.
- Heat the mixture to 50-80°C to induce cyclization and crystallization, yielding the final quinazoline dione product with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented one-pot synthesis offers profound strategic benefits that extend far beyond simple chemical curiosity. The elimination of solid phosgene and hydrogen chloride gas from the supply chain removes a major logistical bottleneck, as these substances require specialized transportation and storage infrastructure that is both costly and heavily regulated. By switching to phenyl chloroformate and common organic bases, companies can source raw materials from a much broader vendor base, enhancing supply security and reducing the risk of production stoppages due to reagent shortages. Furthermore, the simplified process flow reduces the number of unit operations required, which directly translates to lower utility consumption and reduced labor hours per batch. This operational efficiency allows for faster turnaround times and greater flexibility in meeting market demand fluctuations without the need for massive capital investment in new safety equipment.
- Cost Reduction in Manufacturing: The economic impact of replacing hazardous gases with stable liquids is substantial, as it negates the need for expensive corrosion-resistant reactors and complex gas scrubbing systems. The one-pot nature of the reaction significantly reduces solvent consumption and waste treatment costs, leading to a leaner and more cost-effective production model. Additionally, the high yields achieved minimize the loss of valuable starting materials, ensuring that the cost of goods sold remains competitive even in volatile raw material markets. These factors combine to create a robust financial case for transitioning to this greener methodology.
- Enhanced Supply Chain Reliability: Sourcing phenyl chloroformate and standard organic solvents is far less restrictive than procuring controlled toxic gases, allowing for more flexible and resilient supply chain planning. The reduced regulatory burden associated with handling non-gaseous reagents simplifies compliance audits and lowers insurance premiums, contributing to overall operational stability. This reliability ensures consistent delivery schedules to downstream customers, strengthening business relationships and market reputation. Companies can thus operate with greater confidence, knowing that their production capabilities are not tethered to the availability of high-risk specialty chemicals.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of toxic emissions make this process inherently scalable, allowing for seamless transition from pilot batches to multi-ton commercial production. The reduction in hazardous waste generation aligns perfectly with increasingly stringent environmental regulations, future-proofing the manufacturing facility against tighter emission standards. This environmental stewardship not only avoids potential fines but also enhances the brand image of the manufacturer as a responsible corporate citizen. Consequently, the process supports sustainable growth and long-term viability in the global agrochemical market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route, drawing directly from the data and claims presented in the patent documentation. Understanding these details is essential for technical teams evaluating the feasibility of adopting this method for their own production lines. The answers provided reflect the specific advantages and operational parameters defined in the intellectual property, ensuring accuracy and relevance for decision-makers.
Q: What are the safety advantages of this new synthesis method compared to traditional routes?
A: The traditional method relies on solid phosgene and hydrogen chloride gas, both of which are highly toxic and corrosive. This new patent utilizes phenyl chloroformate and mild bases, eliminating the need for specialized gas-handling equipment and significantly reducing occupational health hazards.
Q: What yields and purity levels can be expected from this one-pot process?
A: According to the experimental data in the patent, the process consistently achieves total yields ranging from 76% to 85%, with final product purity exceeding 98% after simple filtration and drying, minimizing the need for complex purification steps.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process uses common organic solvents like toluene and acetonitrile and operates under mild temperature conditions (0-10°C followed by 50-80°C). The elimination of hazardous gases and the one-pot nature of the reaction make it highly scalable and cost-effective for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-dichlorophenyl-6-fluoro-2,4(1H,3H)-quinazolinedione Supplier
As the demand for safer and more efficient agrochemical intermediates continues to rise, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM provides a distinct competitive advantage. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 3-dichlorophenyl-6-fluoro-2,4(1H,3H)-quinazolinedione meets the highest industry standards. By leveraging our expertise in green chemistry and process optimization, we help clients navigate the complexities of modern manufacturing while maintaining cost efficiency.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific production requirements. Request a Customized Cost-Saving Analysis today to understand the potential economic benefits for your organization. Our experts are ready to provide specific COA data and route feasibility assessments, ensuring that you have all the necessary information to make informed sourcing decisions. Let us collaborate to build a more sustainable and profitable supply chain for your agrochemical products.
