Advanced Synthesis and Commercialization of Chiral Mitochondria-Targeting Radiation Protection Compounds
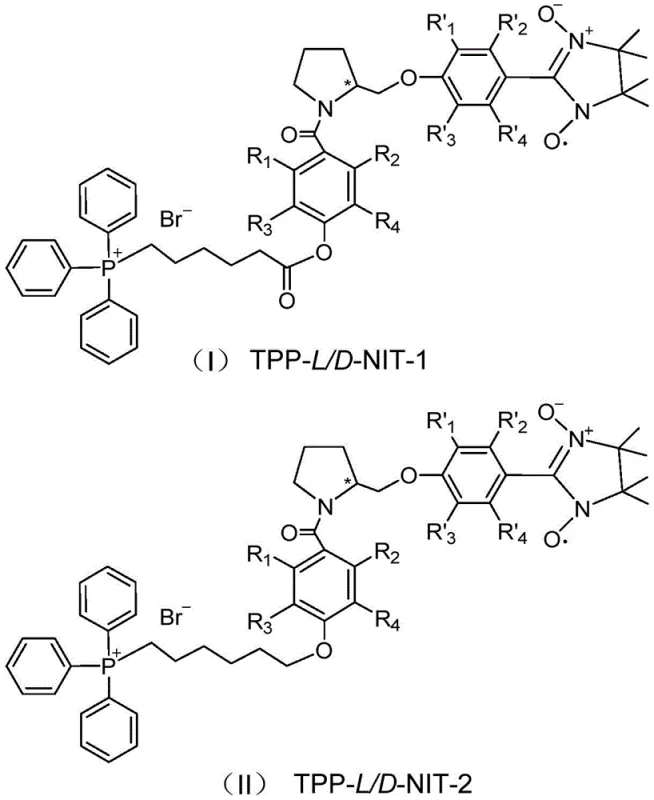
The escalating utilization of nuclear technology across defense, medical, and industrial sectors has precipitated an urgent demand for effective countermeasures against ionizing radiation damage. While traditional agents exist, they often suffer from significant toxicity profiles that limit their clinical utility. A groundbreaking advancement in this field is detailed in patent CN111018913A, which discloses a novel class of chiral mitochondria-targeting nitroxide radical compounds. These molecules, specifically designated as TPP-L/D-NIT-1 and TPP-L/D-NIT-2, represent a paradigm shift in radioprotection by combining a triphenylphosphine cation with a chiral prolinol nitroxide radical. This unique structural architecture not only enhances the scavenging efficiency of harmful free radicals but also ensures exceptional biocompatibility, addressing the critical safety limitations of prior art. For global pharmaceutical developers, this technology offers a robust foundation for creating next-generation radioprotective pharmaceutical compositions with superior safety margins.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the landscape of radioprotective pharmacology has been dominated by compounds such as amifostine (WR2721), which remains the sole agent approved by the US FDA for this indication. However, the clinical deployment of amifostine is severely constrained by its narrow therapeutic index, where the dosage required to achieve a curative effect approaches levels that induce significant toxicity. This precarious balance necessitates rigorous monitoring and limits widespread prophylactic use, particularly in scenarios requiring rapid administration or repeated exposure. Furthermore, conventional free radical scavengers often lack specificity, distributing non-selectively throughout the body rather than concentrating at the subcellular sites most vulnerable to radiation-induced oxidative stress. This lack of targeted delivery results in suboptimal protection of critical organelles like mitochondria, which are the primary generators of reactive oxygen species following ionizing radiation exposure, thereby failing to prevent the cascade of apoptotic signals that lead to tissue and organ damage.
The Novel Approach
In stark contrast to these legacy solutions, the novel approach outlined in the patent data introduces a dual-function molecular design that integrates mitochondrial targeting with catalytic free radical scavenging. By conjugating a lipophilic triphenylphosphine cation with a stable nitroxide radical via a chiral prolinol linker, the new compounds actively accumulate within the mitochondrial matrix driven by the organelle's membrane potential. This targeted accumulation ensures that the antioxidant capacity is delivered precisely where it is needed most, effectively neutralizing reactive oxygen species before they can trigger mitochondrial dysfunction or apoptosis. Moreover, the incorporation of a chiral prolinol unit, which structurally mimics endogenous proline, significantly enhances the biocompatibility of the molecule, reducing the risk of immunogenic reactions or metabolic interference. This strategic molecular engineering results in a radioprotective agent that demonstrates obvious efficacy at the animal level while maintaining a safety profile far superior to existing small molecule scavengers.
Mechanistic Insights into Triphenylphosphine-Conjugated Nitroxide Radical Formation
The core innovation of this technology lies in the sophisticated catalytic cycle enabled by the imidazole nitroxide radical moiety. Unlike stoichiometric antioxidants that are consumed upon neutralizing a single free radical, the nitroxide radical structure facilitates a cyclic reaction mechanism wherein the molecule is regenerated after scavenging reactive species. This catalytic mode of action allows for sustained protection over extended periods, providing a continuous shield against the persistent oxidative stress generated by ionizing radiation. The presence of the triphenylphosphine group is equally critical, as it acts as a lipophilic cation that exploits the electrochemical gradient across the inner mitochondrial membrane. This ensures rapid and selective uptake into the mitochondria, which occupy up to 30% of the cell volume and are the epicenter of radiation-induced cellular damage. By concentrating the antioxidant defense system within this vital organelle, the compound effectively preserves ATP synthesis and prevents the release of pro-apoptotic factors, thereby maintaining cellular viability under extreme stress conditions.
Furthermore, the stereochemistry of the prolinol linker plays a pivotal role in the pharmacokinetic and pharmacodynamic properties of the final drug substance. The use of L-prolinol or D-prolinol introduces chirality that closely resembles natural amino acids found in biological systems, thereby facilitating better metabolic integration and reduced off-target effects. This structural similarity minimizes the likelihood of the compound being recognized as a foreign xenobiotic by hepatic enzymes, potentially extending its half-life and improving overall bioavailability. The synthesis pathway meticulously preserves this chiral integrity through mild reaction conditions that avoid racemization, ensuring that the final product retains the specific spatial configuration required for optimal interaction with biological targets. This attention to stereochemical detail underscores the high level of precision engineering involved in developing these advanced radioprotective intermediates for clinical application.
How to Synthesize TPP-L-NIT-1 Efficiently
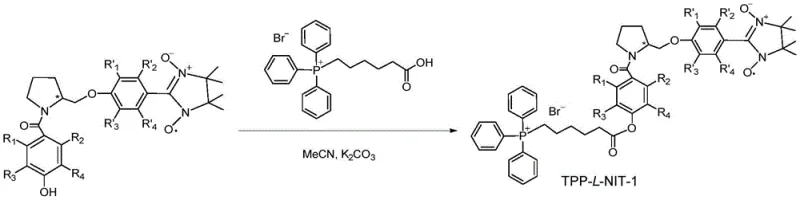
The synthesis of these high-value intermediates follows a modular strategy that allows for precise control over purity and yield at every stage. The process begins with the preparation of the phosphonium salt linker, followed by the independent construction of the chiral nitroxide radical core, and concludes with a convergent coupling step. This divide-and-conquer approach enables manufacturers to optimize each sub-unit separately before final assembly, significantly reducing the complexity of purification and improving overall process robustness. Detailed standardized synthetic steps see the guide below, which outlines the specific reagents, temperatures, and workup procedures required to achieve pharmaceutical-grade quality. Adhering to these protocols ensures consistent reproduction of the chiral centers and the stability of the sensitive nitroxide functionality.
- Synthesize the triphenylphosphine cationic intermediate by reacting triphenylphosphine with 6-bromohexanoic acid or 1,6-dibromohexane under reflux conditions.
- Prepare the chiral nitroxide radical precursor through sequential acylation, etherification, reduction, and oxidation of L-prolinol or D-prolinol.
- Couple the phosphonium salt intermediate with the nitroxide radical precursor in anhydrous acetonitrile to obtain the final TPP-L/D-NIT compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain and procurement perspective, the adoption of this synthesis route offers substantial strategic benefits that extend beyond mere technical performance. The reliance on commodity chemicals such as triphenylphosphine, bromohexanoic acid, and prolinol ensures a stable and diversified raw material base, mitigating the risks associated with sourcing exotic or controlled precursors. This accessibility translates directly into enhanced supply chain reliability, as manufacturers are less vulnerable to single-source bottlenecks or geopolitical disruptions affecting specialized reagent markets. Furthermore, the mild reaction conditions employed throughout the synthesis, often proceeding at room temperature or under simple reflux, reduce the energy intensity of the manufacturing process. This operational efficiency not only lowers the carbon footprint of production but also simplifies the engineering requirements for reactor setups, making the technology adaptable to a wide range of existing chemical infrastructure without necessitating costly capital upgrades.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of straightforward purification techniques like crystallization and column chromatography significantly streamline the production workflow. By avoiding complex metal removal steps, which often require specialized resins and extensive validation, manufacturers can achieve substantial cost savings in both material consumption and processing time. Additionally, the high reproducibility of the reaction minimizes batch-to-batch variability, reducing the incidence of failed batches and the associated waste disposal costs. This economic efficiency makes the commercial production of these radioprotective intermediates highly viable, offering a competitive pricing structure for downstream pharmaceutical formulators seeking to develop affordable radioprotection therapies.
- Enhanced Supply Chain Reliability: The synthetic route is designed with scalability in mind, utilizing solvents and reagents that are globally available in bulk quantities. This ensures that production schedules can be maintained consistently, even during periods of high market demand or logistical constraints. The robustness of the chemistry means that scale-up from laboratory to pilot plant does not introduce unforeseen kinetic or thermodynamic hurdles, allowing for a smooth transition to commercial volumes. Consequently, partners can rely on a steady stream of high-quality intermediates, reducing lead times for high-purity mitochondria-targeted compounds and ensuring that critical drug development timelines are met without delay.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste compared to traditional heavy metal-catalyzed routes, aligning with increasingly stringent environmental regulations and corporate sustainability goals. The solvents used, primarily acetonitrile and dichloromethane, are well-understood and can be efficiently recovered and recycled through standard distillation processes. This closed-loop approach to solvent management reduces the environmental impact of manufacturing and lowers the cost of waste treatment. Moreover, the absence of toxic heavy metals in the final product simplifies regulatory filings and quality control testing, accelerating the path to market approval for new drug applications incorporating these advanced radioprotective agents.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. They are derived from a thorough analysis of the background challenges in radioprotection and the specific beneficial effects demonstrated in the patent data. Understanding these aspects is crucial for R&D and procurement teams evaluating the feasibility of integrating these intermediates into their pipeline. The answers provide clarity on the distinct advantages of this chiral nitroxide approach over conventional methods.
Q: How does this novel compound improve upon existing radioprotectors like Amifostine?
A: Unlike Amifostine, which has a narrow therapeutic window close to toxic dosages, this chiral mitochondria-targeting nitroxide radical offers superior biocompatibility due to its prolinol structural unit similarity to biological proline, significantly reducing toxicity risks while maintaining high efficacy.
Q: What is the mechanism behind the mitochondrial targeting capability?
A: The compound utilizes a triphenylphosphine (TPP) cation moiety which leverages the large negative membrane potential of mitochondria to actively accumulate within the organelle, ensuring direct scavenging of reactive oxygen species at the primary site of radiation-induced damage.
Q: Is the synthesis process suitable for large-scale industrial production?
A: Yes, the patented method employs mild reaction conditions, readily available raw materials such as triphenylphosphine and prolinol, and standard purification techniques like column chromatography, ensuring good reproducibility and feasibility for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable TPP-L-NIT-1 Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a dependable supply chain for advanced pharmaceutical intermediates like TPP-L-NIT-1 and TPP-L-NIT-2. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of radioprotective compound meets the highest standards of quality and consistency required for clinical and commercial applications. We understand the complexities involved in handling chiral nitroxide radicals and have optimized our processes to maintain stereochemical integrity throughout the manufacturing lifecycle.
We invite you to collaborate with us to leverage this cutting-edge technology for your radioprotection programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline constraints. By partnering with us, you gain access to specific COA data and comprehensive route feasibility assessments that will de-risk your development process. Contact us today to discuss how we can support your journey from bench-scale discovery to full-scale commercialization with our reliable radioprotective compound supplier capabilities.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
