Advanced Chiral Macrocyclic Aminophosphine Ligands for High-Purity Pharmaceutical Intermediate Manufacturing
Advanced Chiral Macrocyclic Aminophosphine Ligands for High-Purity Pharmaceutical Intermediate Manufacturing
The landscape of asymmetric catalysis is continuously evolving to meet the stringent demands of the modern pharmaceutical industry, where the production of single-enantiomer compounds is not merely a preference but a regulatory necessity. A significant technological advancement in this domain is documented in patent CN102093420A, which introduces a novel class of chiral macrocyclic aminophosphine ligands featuring a unique P2N4 coordination framework. This innovation addresses critical challenges faced by R&D teams globally, specifically regarding the stability and selectivity of catalysts used in the synthesis of high-value chiral aromatic alcohols. Unlike traditional open-chain ligands that often suffer from conformational flexibility and sensitivity to environmental factors, these macrocyclic structures offer a rigid scaffold that significantly enhances the stereocontrol during catalytic cycles. For procurement and supply chain leaders, understanding the underlying chemistry of such patents is vital, as it directly correlates to the reliability and cost-efficiency of sourcing reliable pharmaceutical intermediates supplier partners who can leverage these advanced synthetic routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral alcohols, which serve as pivotal building blocks for numerous active pharmaceutical ingredients, has relied heavily on open-chain chiral ligands such as DIOP or BINAP derivatives. While effective in specific contexts, these conventional systems often present substantial limitations when applied to large-scale manufacturing environments. One primary drawback is their conformational flexibility, which can lead to less defined chiral pockets around the metal center, resulting in variable enantioselectivity and the formation of unwanted isomers that require costly purification steps. Furthermore, many traditional ligands exhibit high sensitivity to oxygen and moisture, necessitating rigorous inert atmosphere conditions that increase operational complexity and infrastructure costs. The reliance on precious metal catalysts without robust ligand support can also lead to catalyst deactivation over time, reducing turnover numbers and driving up the overall cost reduction in pharmaceutical intermediates manufacturing. These factors collectively create bottlenecks in the supply chain, affecting lead times and consistency.
The Novel Approach
The introduction of the chiral macrocyclic aminophosphine ligand described in the referenced patent represents a paradigm shift towards more robust and efficient catalytic systems. By incorporating both soft phosphine and hard amine donor atoms within a rigid macrocyclic framework, this new ligand class creates a highly stable coordination environment for transition metals. This structural rigidity minimizes conformational freedom, thereby locking the substrate into a precise orientation that maximizes enantioselectivity during the hydrogenation process. The P2N4 coordination motif allows for the formation of chiral metal complexes that are remarkably stable to air and water, eliminating the need for expensive and cumbersome glovebox operations. This stability translates directly into operational simplicity and safety, making the technology highly attractive for commercial scale-up of complex pharmaceutical intermediates. The ability to achieve high conversion rates and optical purity under mild conditions underscores the potential of this approach to revolutionize the production of chiral aromatic alcohols.
Mechanistic Insights into P2N4-Coordinated Asymmetric Hydrogenation
To fully appreciate the value proposition of this technology for R&D directors, one must delve into the mechanistic nuances of the P2N4-coordinated catalytic cycle. The macrocyclic ligand acts as a tetradentate or multidentate chelator, wrapping around the transition metal center, typically rhodium or ruthenium, to form a well-defined chiral cavity. 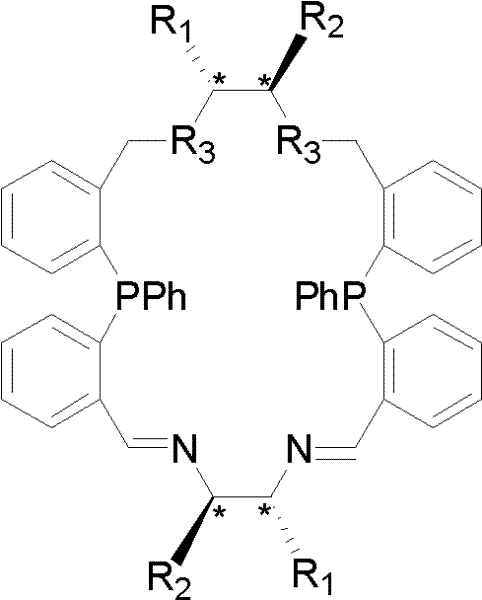 . This rigid structure prevents the ligand from dissociating during the reaction, ensuring that the chiral information is consistently transferred to the substrate throughout the catalytic turnover. The presence of both phosphine and amine functionalities allows for cooperative activation of the substrate and the hydrogen source, facilitating a smooth hydride transfer mechanism. The steric bulk provided by the phenyl groups on the phosphorus atoms further shields the active site, preventing non-selective background reactions. This precise control over the transition state geometry is what enables the system to achieve exceptional enantiomeric excess values, often exceeding industry standards for critical drug intermediates.
. This rigid structure prevents the ligand from dissociating during the reaction, ensuring that the chiral information is consistently transferred to the substrate throughout the catalytic turnover. The presence of both phosphine and amine functionalities allows for cooperative activation of the substrate and the hydrogen source, facilitating a smooth hydride transfer mechanism. The steric bulk provided by the phenyl groups on the phosphorus atoms further shields the active site, preventing non-selective background reactions. This precise control over the transition state geometry is what enables the system to achieve exceptional enantiomeric excess values, often exceeding industry standards for critical drug intermediates.
Impurity control is another critical aspect where this mechanistic design excels, directly addressing the purity concerns of quality assurance teams. In conventional catalytic systems, ligand decomposition or metal leaching can introduce difficult-to-remove impurities into the final product stream. However, the robust nature of the macrocyclic P2N4 ligand minimizes degradation pathways, ensuring that the catalyst remains intact throughout the reaction duration. The high stability against air and moisture also reduces the risk of oxidation byproducts that often plague sensitive phosphine ligands. Consequently, the downstream purification process is significantly simplified, as there are fewer side products to separate from the desired chiral alcohol. This inherent cleanliness of the reaction profile not only improves the overall yield but also ensures that the final high-purity pharmaceutical intermediates meet the stringent specifications required for regulatory submission. The combination of high selectivity and low impurity generation makes this ligand system a superior choice for GMP manufacturing environments.
How to Synthesize Chiral Macrocyclic Aminophosphine Ligands Efficiently
The synthesis of these advanced ligands is designed to be straightforward and scalable, utilizing readily available starting materials and standard laboratory equipment. The process begins with the condensation of bis(2-formylphenyl)phenylphosphine with a chiral diamine, such as (R,R)-cyclohexyl-1,2-diamine, in a halogenated hydrocarbon solvent. 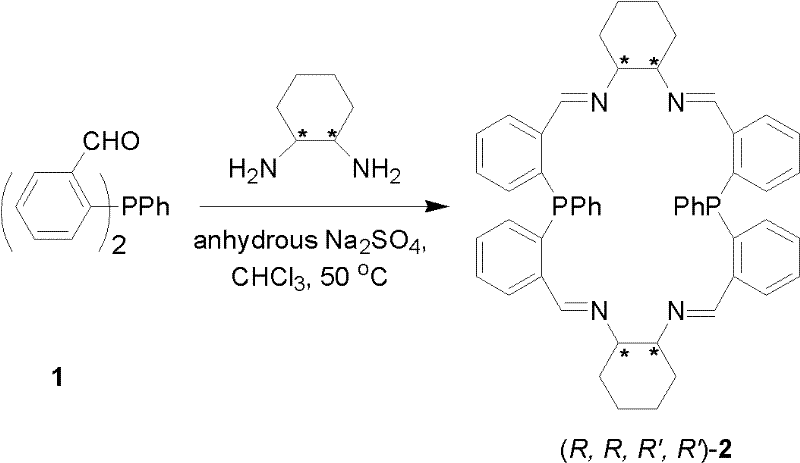 . This step forms the imine-containing macrocyclic precursor, which is isolated as a stable yellow-green solid after simple workup procedures involving filtration and recrystallization. The subsequent reduction of the imine bonds to amine bonds using sodium borohydride yields the final saturated aminophosphine ligand.
. This step forms the imine-containing macrocyclic precursor, which is isolated as a stable yellow-green solid after simple workup procedures involving filtration and recrystallization. The subsequent reduction of the imine bonds to amine bonds using sodium borohydride yields the final saturated aminophosphine ligand. 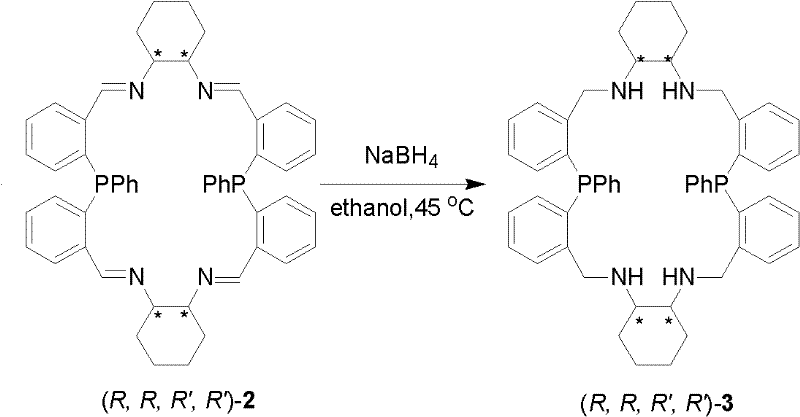 . The entire synthetic route avoids the use of exotic reagents or extreme conditions, making it highly amenable to technology transfer and large-scale production. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and quality control across different manufacturing sites.
. The entire synthetic route avoids the use of exotic reagents or extreme conditions, making it highly amenable to technology transfer and large-scale production. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and quality control across different manufacturing sites.
- Condense bis(2-formylphenyl)phenylphosphine with chiral cyclohexyl diamine in halogenated hydrocarbon with dehydrating agent.
- Isolate the intermediate imine phosphine ligand via filtration, washing, and recrystallization to obtain yellow-green crystals.
- Reduce the imine intermediate using sodium borohydride in alcohol solvent to yield the final stable amine phosphine ligand.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ligand technology offers tangible benefits that extend beyond mere chemical performance. The primary advantage lies in the drastic simplification of the manufacturing process, which directly impacts the total cost of ownership. Because the catalyst system is stable to air and moisture, facilities do not need to invest in specialized inert atmosphere infrastructure for every reaction vessel, leading to substantial capital expenditure savings. Furthermore, the high turnover numbers and selectivity reduce the amount of catalyst required per kilogram of product, lowering the raw material costs associated with precious metals. The elimination of complex purification steps to remove isomers or decomposition byproducts also reduces solvent consumption and waste generation, aligning with sustainability goals and reducing disposal costs. These factors combine to create a more resilient and cost-effective supply chain for critical chiral building blocks.
- Cost Reduction in Manufacturing: The implementation of this robust catalytic system eliminates the need for expensive heavy metal scavengers often required to clean up unstable catalyst residues, resulting in significant process cost optimization. By utilizing simple transition metal complexes that are easily sourced, manufacturers can avoid the price volatility associated with specialized proprietary catalysts. The high efficiency of the reaction means less energy is consumed per unit of product, as reactions can proceed at moderate temperatures without prolonged heating or cooling cycles. Additionally, the reduced need for extensive chromatographic purification lowers the consumption of silica gel and organic solvents, which are major cost drivers in fine chemical production. Overall, the process economics are favorable due to the streamlined workflow and higher throughput capabilities.
- Enhanced Supply Chain Reliability: The stability of the ligand and the resulting catalyst ensures consistent batch-to-batch performance, which is crucial for maintaining uninterrupted supply to downstream drug manufacturers. Since the reagents are not hypersensitive to shipping conditions or short-term exposure to ambient air, logistics become simpler and less prone to disruption. This reliability reduces the risk of production delays caused by catalyst failure or the need to requalify batches due to purity issues. Suppliers leveraging this technology can offer more predictable lead times, allowing pharmaceutical companies to optimize their inventory levels and reduce safety stock requirements. The robustness of the chemistry essentially de-risks the supply chain for high-value chiral intermediates.
- Scalability and Environmental Compliance: The synthetic route is inherently scalable, having been demonstrated to work effectively with increased substrate loading without loss of selectivity or yield. This scalability ensures that the technology can meet the growing demand for chiral drugs without requiring disproportionate increases in manufacturing footprint. From an environmental perspective, the use of safer solvents like isopropanol in the catalytic step and the reduction of waste streams contribute to a greener manufacturing profile. The process aligns well with modern environmental regulations regarding volatile organic compound emissions and hazardous waste disposal. This compliance facilitates smoother regulatory approvals and enhances the corporate social responsibility profile of the manufacturing partner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral ligand technology in industrial settings. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this chemistry into their existing production pipelines. It clarifies the operational parameters and expected outcomes, ensuring alignment between technical capabilities and business objectives.
Q: What are the stability advantages of this macrocyclic ligand compared to open-chain variants?
A: The macrocyclic structure provides enhanced rigidity and stability against air and moisture, simplifying handling and storage requirements compared to sensitive open-chain ligands.
Q: Can this ligand system be scaled for industrial production of chiral alcohols?
A: Yes, the catalytic system operates under mild conditions with simple transition metal complexes, demonstrating high conversion rates suitable for commercial scale-up.
Q: What level of enantioselectivity can be expected in asymmetric hydrogenation?
A: Experimental data indicates optical purity reaching up to 99% ee for various aromatic ketone substrates, ensuring high-quality chiral intermediate output.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Macrocyclic Aminophosphine Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the P2N4 macrocyclic ligand system in driving the next generation of pharmaceutical synthesis. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative lab-scale chemistry can be successfully translated into robust manufacturing processes. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of analyzing complex chiral mixtures to guarantee the highest quality standards. We understand that the transition from patent to production requires not just chemical expertise but also a deep commitment to reliability and continuous improvement. Our team is prepared to handle the nuances of air-sensitive chemistry and complex purification requirements to deliver intermediates that meet your exact needs.
We invite you to collaborate with us to explore how this technology can optimize your specific synthesis routes and reduce your overall production costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your project requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your supply chain goals. By partnering with us, you gain access to a wealth of chemical knowledge and manufacturing capacity designed to accelerate your drug development timelines. Let us help you turn complex chemical challenges into competitive commercial advantages through our expert engineering and production services.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
