Scalable Metal-Free Production of Benzosiloxaborole Heterocycles for Advanced Pharmaceutical Applications
Scalable Metal-Free Production of Benzosiloxaborole Heterocycles for Advanced Pharmaceutical Applications
The landscape of organometallic chemistry is constantly evolving, driven by the urgent need for more sustainable and efficient synthetic routes in the pharmaceutical industry. A significant breakthrough in this domain is detailed in patent CN114409691A, which discloses a novel preparation method for benzosiloxaboron heterocyclic compounds. These unique structures, containing both silicon and boron elements within a fused ring system, represent a valuable class of intermediates with potential applications in medicinal chemistry and materials science. The disclosed technology leverages boron trichloride (BCl3) to facilitate an intramolecular cyclization of 2-allylphenylsilane derivatives, achieving high conversion rates without the reliance on precious transition metal catalysts. This development addresses critical pain points in modern synthesis, offering a pathway that is not only chemically elegant but also commercially viable for the mass production of high-purity organosilicon boron compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of organosilicon-boron compounds has been a challenging endeavor, often plagued by inefficiencies that hinder large-scale adoption. Traditional methodologies typically rely on multi-step sequences to construct the carbon-silicon and carbon-boron bonds independently, which inherently lowers the overall yield and increases waste generation. Furthermore, many existing protocols depend heavily on transition metal catalysts, such as palladium or rhodium complexes, to drive the coupling reactions. While effective on a small laboratory scale, these metal-catalyzed processes introduce significant downstream processing burdens, particularly the rigorous removal of trace metal residues to meet stringent pharmaceutical purity standards. Additionally, prior art has largely been limited to intermolecular reactions, which suffer from poor selectivity and require complex separation techniques to isolate the desired products from oligomeric byproducts, making them less attractive for cost-sensitive industrial applications.
The Novel Approach
In stark contrast to these cumbersome traditional routes, the methodology outlined in CN114409691A introduces a streamlined, one-pot strategy that fundamentally reshapes the synthetic logic. By utilizing boron trichloride as a key reagent, the process enables a direct intramolecular reaction that simultaneously constructs the heterocyclic framework with remarkable efficiency. This approach eliminates the need for any metal participation, thereby sidestepping the issues of catalyst cost and contamination entirely. The reaction proceeds under mild conditions, specifically at room temperature, which drastically reduces energy consumption compared to thermal or high-pressure alternatives. As illustrated in the general reaction scheme below, the transformation converts readily available 2-allylphenylsilane precursors directly into the target benzosiloxaborole core, showcasing a level of operational simplicity that is highly desirable for a reliable benzosiloxaborole supplier aiming to optimize their manufacturing portfolio.
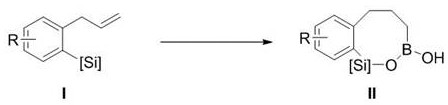
Mechanistic Insights into BCl3-Mediated Intramolecular Cyclization
The core of this technological advancement lies in the unique reactivity of boron trichloride towards the unsaturated carbon-carbon bonds present in the allyl side chain of the substrate. Unlike traditional Lewis acids that might simply coordinate, BCl3 acts as a potent electrophile that activates the alkene moiety, facilitating a cascade of bond-forming events. The silicon atom, bearing a hydride group in the starting material, participates in a concerted or stepwise migration that results in the formation of the new silicon-oxygen-boron linkage. This intramolecular nature ensures high regioselectivity, as the reacting centers are held in close proximity by the molecular backbone, effectively suppressing the formation of intermolecular oligomers that often plague similar intermolecular borylation reactions. The resulting seven-membered heterocyclic ring is stabilized by the strong affinity between the boron and oxygen atoms formed during the aqueous workup, locking the structure into a stable configuration suitable for further functionalization.
From an impurity control perspective, this mechanism offers distinct advantages that are crucial for producing high-purity organosilicon boron compounds. Because the reaction is driven by the intrinsic geometry of the substrate and the specific electrophilicity of BCl3, side reactions such as polymerization or non-specific halogenation are minimized. The absence of transition metals means there is no risk of metal-induced decomposition pathways or the formation of metal-complexed impurities that are notoriously difficult to remove. Furthermore, the mild reaction conditions prevent thermal degradation of sensitive functional groups that might be present on the aromatic ring, such as the methyl substituent shown in specific examples. This inherent cleanliness of the reaction profile translates directly into a simpler purification workflow, often requiring only standard column chromatography to achieve analytical purity, which is a significant benefit for reducing lead time for high-purity intermediates in a commercial setting.
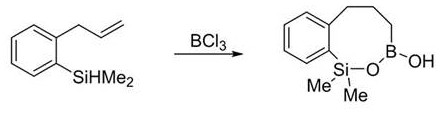
How to Synthesize Benzosiloxaborole Efficiently
Implementing this synthesis in a practical setting requires adherence to specific operational parameters to maximize yield and safety. The process begins with the dissolution of the 2-allylphenylsilane substrate in 1,2-dichloroethane, a solvent chosen for its ability to solubilize both the organic starting material and the inorganic boron reagent while maintaining stability under reaction conditions. The addition of boron trichloride must be controlled carefully, typically using 1.2 equivalents relative to the substrate to ensure complete conversion without excessive reagent waste. Following the reaction period, a straightforward aqueous quench hydrolyzes the intermediate boron-chlorine species to the final boron-hydroxyl form, allowing for easy phase separation. For a detailed breakdown of the specific stoichiometric ratios, stirring times, and purification techniques validated in the patent examples, please refer to the standardized protocol below.
- Dissolve the 2-allylphenylsilane substrate in 1,2-dichloroethane solvent within a reaction flask under inert atmosphere.
- Add boron trichloride (BCl3) dropwise to the stirred solution at room temperature, maintaining a molar ratio of approximately 1.2 equivalents.
- Stir the reaction mixture for 3 hours, followed by aqueous quenching, extraction with dichloromethane, and purification via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift from metal-catalyzed to metal-free synthesis represents a strategic opportunity to optimize both cost structures and operational reliability. The elimination of precious metal catalysts removes a volatile cost component from the bill of materials, as prices for metals like palladium can fluctuate wildly based on geopolitical factors. Moreover, the simplified downstream processing reduces the consumption of specialized scavenging resins and filtration media, leading to substantial cost savings in the overall manufacturing process. The robustness of the reaction conditions also implies a lower risk of batch failure due to equipment sensitivity, ensuring a more consistent supply of critical intermediates for downstream drug synthesis.
- Cost Reduction in Manufacturing: The most immediate financial impact comes from the complete removal of transition metal catalysts, which are often the single most expensive reagent in fine chemical synthesis. By replacing these with commodity chemicals like boron trichloride, the raw material costs are drastically simplified and reduced. Additionally, the mild room temperature operation eliminates the need for energy-intensive heating or cooling systems, further lowering the utility overheads associated with production. The high conversion rates, reaching up to 80% in optimized cases, mean that less starting material is wasted, improving the overall atom economy and reducing the cost per kilogram of the final active ingredient.
- Enhanced Supply Chain Reliability: Relying on widely available reagents such as 1,2-dichloroethane and boron trichloride mitigates the risk of supply disruptions that often accompany specialized ligands or custom catalysts. This accessibility ensures that production schedules can be maintained without waiting for long-lead-time imports. Furthermore, the operational simplicity of the process allows for greater flexibility in manufacturing scheduling, as the reaction does not require complex setup or extended equilibration times. This agility enables suppliers to respond more rapidly to fluctuations in demand, securing the continuity of supply for critical pharmaceutical pipelines.
- Scalability and Environmental Compliance: The transition to a metal-free process aligns perfectly with increasingly stringent environmental regulations regarding heavy metal discharge. By avoiding the use of toxic transition metals, the wastewater treatment burden is significantly lightened, reducing the costs and complexities associated with environmental compliance. The process is inherently scalable, as the exothermic nature of the reaction can be easily managed in larger reactors through controlled dosing, facilitating the commercial scale-up of complex heterocyclic intermediates from pilot plant to multi-ton production without the need for re-engineering the core chemistry.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of integrating this technology into their existing workflows, we have compiled answers to common inquiries based on the patent data. These questions address the practical aspects of implementation, including yield expectations, safety considerations, and compatibility with various substrates. Understanding these details is essential for making informed decisions about adopting this novel synthetic route for your specific project needs.
Q: What are the primary advantages of this BCl3-mediated method over traditional transition metal catalysis?
A: The primary advantage is the complete elimination of transition metal catalysts, which removes the need for expensive and time-consuming heavy metal scavenging steps, thereby significantly reducing production costs and simplifying the purification process for pharmaceutical grade intermediates.
Q: What yields can be expected from this intramolecular cyclization process?
A: Experimental data indicates that conversion rates are highly efficient, with isolated yields reaching up to 80% for dimethyl-substituted substrates under mild room temperature conditions, demonstrating excellent atom economy.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process utilizes common solvents like 1,2-dichloroethane and operates at ambient temperatures without requiring specialized high-pressure or cryogenic equipment, making it inherently scalable for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzosiloxaborole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the metal-free cyclization technology described in CN114409691A for the next generation of pharmaceutical intermediates. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this laboratory-scale discovery can be fully realized in an industrial setting. We are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch of benzosiloxaborole derivatives we produce. Our commitment to quality assurance means that we can deliver materials that meet the exacting standards required for clinical trial supplies and commercial API manufacturing.
We invite you to collaborate with us to leverage this efficient synthetic route for your upcoming projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to reach out today to obtain specific COA data for our current inventory and to discuss route feasibility assessments for your target molecules. Let us help you streamline your supply chain and reduce your time-to-market with our advanced manufacturing capabilities.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
