Advanced Organocatalytic Synthesis of Axial Chiral Indole-Naphthalene Compounds for Commercial Scale-Up
The landscape of asymmetric catalysis is constantly evolving, driven by the demand for more efficient and sustainable methods to construct complex chiral architectures. A significant breakthrough in this domain is detailed in patent CN110452150B, which discloses a novel preparation method for axial chiral indole-naphthalene compounds. These compounds are not merely structural curiosities; they serve as critical scaffolds for next-generation organocatalysts and ligands. The patented technology introduces a robust organocatalytic strategy that constructs the axial chiral indole-naphthalene structure in a single step from racemic raw materials. This represents a paradigm shift from multi-step sequences to streamlined, one-pot transformations. By leveraging chiral phosphoric acid catalysis, the method achieves exceptional stereocontrol without the need for expensive transition metals. For R&D teams seeking reliable pharmaceutical intermediate suppliers, this technology offers a pathway to high-value chiral building blocks with simplified supply chains. The general reaction scheme illustrates the convergence of an indole derivative and a phenolic component to form the target architecture.
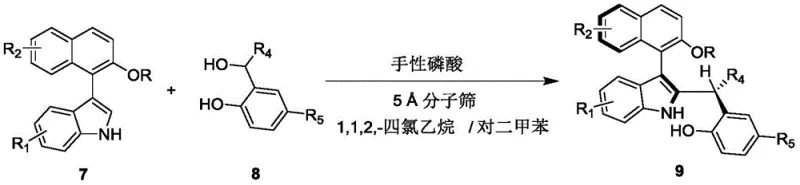
The versatility of this approach is underscored by the broad substrate tolerance, accommodating various substituents on both the indole and naphthalene rings. This flexibility is crucial for medicinal chemists who require diverse libraries for structure-activity relationship (SAR) studies. Furthermore, the operational simplicity—utilizing standard solvents and ambient temperatures—makes this process highly attractive for industrial adoption. As we delve deeper into the technical specifics, it becomes clear that this invention addresses long-standing challenges in the synthesis of axially chiral biaryl systems, offering a solution that balances high yield with superior enantioselectivity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of axial chiral indole-naphthalene compounds has relied heavily on cross-coupling reactions or oxidative couplings that often suffer from significant drawbacks. Traditional methods frequently require pre-functionalized starting materials, such as halogenated aromatics or organometallic reagents, which increase the overall cost and complexity of the synthesis. Moreover, these coupling reactions often necessitate the use of precious metal catalysts like palladium or rhodium, introducing concerns regarding metal residue contamination in the final product—a critical issue for high-purity pharmaceutical intermediates. Another major limitation is the difficulty in controlling stereoselectivity; many conventional routes yield racemic mixtures that require tedious and yield-loss-prone resolution steps. Additionally, the reaction conditions for these older methods can be quite harsh, involving high temperatures or strong bases that limit the functional group tolerance, thereby restricting the structural diversity of the accessible compounds.
The Novel Approach
In stark contrast, the method disclosed in the patent utilizes a dynamic kinetic resolution (DKR) strategy mediated by a chiral phosphoric acid catalyst. This innovative approach allows for the direct conversion of racemic starting materials into enantioenriched products, effectively bypassing the need for separate resolution steps. The reaction proceeds under remarkably mild conditions, typically at temperatures between 20°C and 30°C, which preserves sensitive functional groups and ensures operational safety. The specific synthetic route highlights the use of a specialized spiro-binaphthyl derived chiral phosphoric acid (Compound 6) which creates a highly defined chiral environment. This catalyst facilitates an asymmetric addition reaction that simultaneously forms the carbon-carbon bond and establishes the axial chirality. The result is a process that is not only chemically elegant but also economically superior, offering a streamlined route to complex chiral molecules that was previously unattainable with such efficiency.

Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Asymmetric Addition
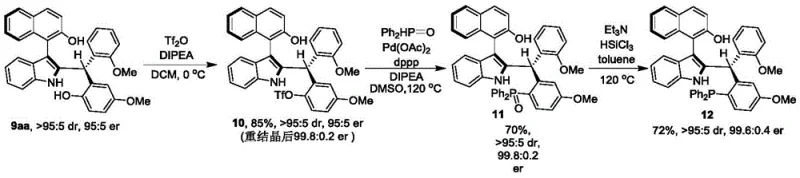
The core of this technological advancement lies in the precise mechanism of the chiral phosphoric acid catalyst. These organocatalysts function through a dual hydrogen-bonding activation mode. The acidic proton of the phosphate group activates the electrophilic species (likely the imine or quinone methide intermediate generated in situ), while the basic phosphoryl oxygen stabilizes the nucleophile or the developing charge in the transition state. In the context of synthesizing axial chiral indole-naphthalenes, the bulky 9-anthracenyl groups on the catalyst backbone (as seen in Compound 6) play a pivotal role. These large substituents create a confined chiral pocket that strictly dictates the approach of the reactants. This steric confinement forces the reaction to proceed through a specific transition state geometry, thereby leading to the preferential formation of one atropisomer over the other. The result is the high enantiomeric ratios (up to 98:2 er) observed in the experimental data. This level of control is essential for producing commercial scale-up of complex polymer additives or active pharmaceutical ingredients where stereochemical purity is non-negotiable.
Furthermore, the mechanism inherently suppresses the formation of impurities. By operating under mild, neutral-to-weakly acidic conditions, the process avoids side reactions such as polymerization or decomposition that are common in strong acid or base catalysis. The use of 5 Å molecular sieves in the reaction mixture further enhances the quality of the crude product by sequestering water, which could otherwise hydrolyze sensitive intermediates or deactivate the catalyst. This attention to detail in the reaction design ensures that the final product requires minimal purification, often achievable through standard silica gel chromatography. For procurement managers, this translates to a cleaner process with higher throughput and reduced waste generation, aligning perfectly with modern green chemistry principles and cost reduction in electronic chemical manufacturing or pharma sectors.
How to Synthesize Axial Chiral Indole-Naphthalene Efficiently
The practical implementation of this synthesis is designed for ease of execution in both laboratory and pilot plant settings. The protocol involves dissolving the indole precursor (Formula 7) and the phenolic partner (Formula 8) in a specific binary solvent system comprising 1,1,2,2-tetrachloroethane and p-xylene. The choice of solvent is critical; the mixture provides the optimal polarity and solubility profile to support the catalytic cycle while maintaining the stability of the chiral catalyst. The reaction is initiated by the addition of the chiral phosphoric acid catalyst (typically at 10 mol% loading) and activated 5 Å molecular sieves. The mixture is then stirred at ambient temperature (25°C) for approximately 12 hours. Monitoring via TLC ensures the reaction is driven to completion before workup. The detailed standardized synthesis steps for optimizing yield and enantioselectivity are provided below.
- Prepare the reaction mixture by combining compound of formula 7 and compound of formula 8 in a mixed solvent of 1,1,2,2-tetrachloroethane and p-xylene.
- Add 5 Å molecular sieves and a chiral phosphoric acid catalyst (such as compound 6) to the mixture under stirring.
- Stir the reaction at 20-30°C until completion monitored by TLC, then filter, concentrate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the adoption of this organocatalytic methodology offers distinct strategic advantages over traditional metal-catalyzed routes. The elimination of transition metals such as palladium removes a significant bottleneck in the supply chain, as the sourcing and price volatility of precious metals can disrupt production schedules. Furthermore, the removal of heavy metals simplifies the downstream purification process, eliminating the need for expensive scavenging resins or complex extraction protocols required to meet strict residual metal specifications. This streamlining of the workflow leads to substantial cost savings and faster turnaround times. The use of readily available, commodity-grade solvents and starting materials further enhances the economic viability of the process, making it a robust choice for long-term manufacturing contracts.
- Cost Reduction in Manufacturing: The process significantly lowers the cost of goods sold (COGS) by utilizing organic small molecule catalysts instead of expensive transition metal complexes. Since the catalyst is metal-free, there is no need for costly metal recovery systems or rigorous testing for heavy metal residues, which reduces analytical overhead. Additionally, the high atom economy of the addition reaction means that a larger proportion of the starting material mass is incorporated into the final product, minimizing raw material waste and maximizing yield efficiency.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable organic catalysts and common chemical feedstocks ensures a resilient supply chain. Unlike specialized metal catalysts that may have long lead times or single-source dependencies, the reagents for this process are widely available from multiple global suppliers. The mild reaction conditions also reduce the risk of batch failures due to thermal runaway or equipment corrosion, ensuring consistent delivery schedules and reducing the risk of production downtime.
- Scalability and Environmental Compliance: The protocol is inherently scalable, having been demonstrated to work efficiently with varied substrate loads without loss of selectivity. The absence of toxic heavy metals and the use of recyclable molecular sieves align with stringent environmental regulations, facilitating easier permitting and waste disposal. This eco-friendly profile reduces the environmental compliance burden and supports corporate sustainability goals, making the process suitable for large-scale industrial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these axial chiral compounds. The answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on the capabilities and limitations of the technology. Understanding these details is vital for partners looking to integrate these intermediates into their own development pipelines.
Q: What is the primary advantage of this synthesis method over traditional coupling reactions?
A: This method utilizes dynamic kinetic resolution via asymmetric addition, allowing for the construction of the axial chiral skeleton in a single step from racemic raw materials under mild conditions, avoiding the harsh conditions and limited substrate scope often associated with traditional coupling methods.
Q: What level of enantioselectivity can be achieved with this process?
A: The process demonstrates excellent stereocontrol, achieving enantiomeric ratios (er) as high as 98:2 and diastereomeric ratios (dr) greater than 95:5 across a wide range of substrates, ensuring high optical purity suitable for catalytic applications.
Q: Can the resulting compounds be used directly as catalysts?
A: Yes, the synthesized axial chiral indole-naphthalene compounds serve as valuable precursors that can be derivatized into chiral phosphine catalysts, which have been proven effective in catalyzing asymmetric [4+1] cyclization reactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axial Chiral Indole-Naphthalene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this organocatalytic technology in the field of asymmetric synthesis. As a premier CDMO partner, we possess the technical expertise to translate this patented methodology from bench-scale discovery to full-scale commercial production. Our facilities are equipped to handle the specific solvent systems and purification requirements of this process, ensuring that every batch meets the highest standards of quality. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, guaranteeing a secure supply of these critical chiral intermediates. Our rigorous QC labs enforce stringent purity specifications, ensuring that the optical purity and chemical integrity of the final product are maintained throughout the manufacturing lifecycle.
We invite potential partners to leverage our capabilities to accelerate their drug discovery or catalyst development programs. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to contact our technical procurement team today to request specific COA data for our catalog of chiral intermediates and to discuss route feasibility assessments for your proprietary targets. Let us help you secure a competitive edge through superior chemical innovation and reliable supply chain execution.