Advanced Synthesis of Planar Pyrazolo Triazine Energetic Molecules for High-Performance Applications
The landscape of high-performance energetic materials is undergoing a significant transformation, driven by the urgent demand for compounds that balance high energy density with exceptional thermal stability and safety. Patent CN113185522A introduces a groundbreaking approach to synthesizing planar pyrazolo triazine energetic molecules, addressing critical limitations found in previous generations of explosives and propellants. This technology leverages a novel cyclization strategy starting from 3,5-diamino-4-nitropyrazole, utilizing malononitrile as a key building block to construct the robust pyrazolo[5,1-c][1,2,4]triazine skeleton. For R&D Directors and Supply Chain Heads in the advanced materials sector, this represents a pivotal shift towards more manageable and scalable synthesis pathways. The resulting compounds, including 8-nitro-3-(1H-tetrazole-5-yl)pyrazolo[5,1-c][1,2,4]triazine-4,7-diamine and its derivatives, demonstrate decomposition temperatures that can exceed 300°C, offering a substantial safety margin over conventional energetic materials like RDX. This patent not only enriches the library of available energetic molecular species but also provides a viable route for the commercial production of next-generation heat-resistant energetic compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
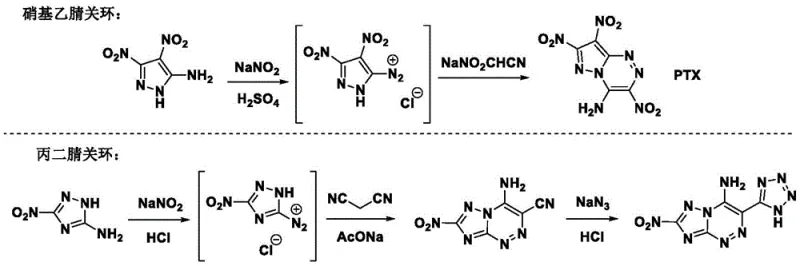
Historically, the synthesis of pyrazolo[5,1-c][1,2,4]triazine backbone energetic molecules has been hindered by the reliance on unstable and hazardous reagents. Previous methods, such as those described in literature involving PTX, often depended on nitroacetonitrile as a key cyclization agent. Nitroacetonitrile is notoriously difficult to prepare on a large scale and poses significant challenges regarding long-term storage and transportation due to its inherent instability. This instability creates a bottleneck for industrial amplification, making it nearly impossible to evaluate these novel energetic molecules in practical weaponry or aerospace applications where consistent supply and safety are paramount. Furthermore, alternative routes using malononitrile cyclization have previously resulted in energetic molecular skeletons with lower thermal stability, often decomposing at temperatures as low as 181°C. Such low decomposition thresholds limit the operational envelope of the final material, rendering them unsuitable for high-temperature environments encountered in modern military and aerospace engineering. The reliance on these problematic reagents and the resulting thermal limitations have stalled the commercial adoption of many promising energetic candidates.
The Novel Approach
The methodology disclosed in CN113185522A overcomes these historical barriers by optimizing the cyclization reaction conditions and reagent selection. By starting from 3,5-diamino-4-nitropyrazole and employing malononitrile under mild acidic conditions, the process achieves rapid construction of the target energetic skeleton without the need for unstable nitroacetonitrile. This strategic substitution ensures that the key reagents are chemically stable, commercially available, and suitable for long-term storage, thereby removing the primary obstacle to scale-up. The reaction conditions are notably mild, avoiding extreme temperatures or pressures that often complicate process safety and equipment requirements. This approach not only simplifies the synthetic steps but also enhances the overall yield and purity of the intermediates. The resulting planar pyrazolo triazine molecules exhibit a high degree of planarity, which contributes to higher crystal density and improved detonation performance. This novel route effectively bridges the gap between laboratory-scale discovery and industrial-scale manufacturing, providing a robust foundation for the production of high-performance energetic materials.
Mechanistic Insights into Malononitrile-Mediated Cyclization
The core of this technological advancement lies in the efficient formation of the pyrazolo[5,1-c][1,2,4]triazine ring system through a diazonium salt intermediate. The process begins with the diazotization of 4-nitro-3,5-diaminopyrazole using tert-butyl nitrite and hydrogen chloride in a methanol solution under ice bath conditions. This step generates a reactive diazonium salt which is crucial for the subsequent ring-closing reaction. The introduction of malononitrile in the presence of potassium oxalate and sulfuric acid facilitates the cyclization, forming the 4,7-diamino-8-nitropyrazolo[5,1-c][1,2,4]triazine-1-cyano intermediate. This specific pathway is advantageous because it avoids the formation of complex by-products often associated with less controlled nitration or cyclization reactions. The mild acidic environment ensures that the sensitive nitro and amino groups remain intact during the skeleton construction, preserving the energetic potential of the molecule. Following the formation of the tricyclic core, the cyano group is converted into a tetrazole ring using sodium azide in DMF. This transformation is critical as the tetrazole moiety significantly enhances the nitrogen content and thermal stability of the final product. The mechanistic precision of this route allows for the systematic derivation of various analogues, including N-oxides and nitramines, by further modifying the amino groups on the skeleton.
Impurity control is inherently built into this synthesis design, which is a key consideration for R&D Directors focused on product consistency. The use of stable starting materials like 3,5-diamino-4-nitropyrazole reduces the likelihood of side reactions that generate difficult-to-remove impurities. The intermediate solids, such as the brick-red 4,7-diamino-8-nitropyrazolo[5,1-c][1,2,4]triazine-1-cyano, can be easily isolated by filtration and washing, ensuring high purity before proceeding to the next step. The final tetrazole cyclization is conducted in a homogeneous DMF solution, which promotes complete conversion and minimizes the presence of unreacted precursors. Post-reaction workup involves pH adjustment and precipitation, a technique that effectively separates the target energetic molecule from inorganic salts and solvent residues. The resulting products are yellow powdery solids that can be further purified via recrystallization to achieve transparent flaky or blocky crystals. This high level of purity is essential for ensuring predictable performance in energetic applications, where even trace impurities can alter sensitivity and stability profiles. The robust nature of the reaction mechanism ensures that the process is reproducible, a critical factor for maintaining quality control in commercial production.
How to Synthesize 8-Nitro-3-(1H-Tetrazole-5-Yl)Pyrazolo Triazine Efficiently
The synthesis of the core compound, 8-nitro-3-(1H-tetrazole-5-yl)pyrazolo[5,1-c][1,2,4]triazine-4,7-diamine, follows a streamlined sequence that is amenable to standard chemical manufacturing equipment. The process begins with the preparation of the diazonium salt, followed by the cyclization with malononitrile and the final tetrazole formation. Each step is designed to maximize yield while maintaining safety, with reaction temperatures kept within manageable ranges (0°C to 90°C). The detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and reaction times, are critical for replicating the high yields reported in the patent data. For process engineers, understanding the precise stoichiometry and thermal profiles is essential for scaling this chemistry from gram to ton scale. The following guide outlines the critical operational parameters required to achieve the reported 94% yield in the final tetrazole formation step.
- Diazotization of 4-nitro-3,5-diaminopyrazole using tert-butyl nitrite and HCl under ice bath conditions to form the diazonium salt intermediate.
- Cyclization reaction with malononitrile in the presence of potassium oxalate and sulfuric acid to form the pyrazolo triazine skeleton.
- Tetrazole ring formation by reacting the cyano intermediate with sodium azide in DMF, followed by purification and optional oxidation or nitration steps.
Commercial Advantages for Procurement and Supply Chain Teams
For Procurement Managers and Supply Chain Heads, the shift from unstable nitroacetonitrile to commercially available malononitrile represents a significant reduction in supply chain risk and operational cost. The traditional reliance on difficult-to-store reagents often necessitates specialized logistics and increases the cost of goods sold due to waste and degradation. By utilizing stable, off-the-shelf starting materials, this new synthesis route drastically simplifies the procurement process and enhances supply continuity. The mild reaction conditions further contribute to cost efficiency by reducing energy consumption and minimizing the need for specialized high-pressure or cryogenic equipment. This process optimization translates directly into a more competitive cost structure for the final energetic material, making it a viable option for large-scale applications in defense and aerospace. The ability to source raw materials reliably ensures that production schedules can be met without the delays often associated with custom synthesis of unstable intermediates.
- Cost Reduction in Manufacturing: The elimination of unstable and hard-to-prepare reagents like nitroacetonitrile removes a major cost driver from the manufacturing process. Malononitrile is a commodity chemical with a stable supply chain, leading to substantial cost savings in raw material procurement. Additionally, the mild reaction conditions reduce energy costs associated with heating and cooling, while the high yields minimize waste disposal expenses. The simplified purification steps, primarily involving filtration and washing, further lower the operational expenditure compared to complex chromatographic separations. These factors combine to create a highly cost-effective manufacturing route that improves the overall margin profile for producers of advanced energetic materials.
- Enhanced Supply Chain Reliability: The use of commercially available and stable starting materials ensures a robust and resilient supply chain. Unlike custom reagents that may have long lead times or limited suppliers, malononitrile and 3,5-diamino-4-nitropyrazole can be sourced from multiple global vendors. This diversity in supply sources mitigates the risk of production stoppages due to raw material shortages. Furthermore, the stability of the intermediates allows for safer storage and transportation, reducing the logistical constraints often imposed on hazardous energetic precursors. This reliability is crucial for maintaining continuous production lines and meeting the demanding delivery schedules of downstream customers in the defense and aerospace sectors.
- Scalability and Environmental Compliance: The synthesis route is designed with scalability in mind, utilizing standard reaction vessels and workup procedures that are easily transferred from pilot plant to full-scale production. The avoidance of extreme conditions and hazardous reagents simplifies the safety assessment and regulatory approval process for new manufacturing facilities. Waste streams are primarily aqueous and organic solvents that can be managed through standard treatment protocols, ensuring compliance with environmental regulations. The high atom economy of the cyclization reaction also contributes to a greener process profile, aligning with the increasing industry focus on sustainable manufacturing practices. This scalability ensures that the technology can meet growing market demand without compromising on safety or environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these planar pyrazolo triazine energetic molecules. The answers are derived directly from the patent data and technical specifications, providing clarity on the performance and manufacturability of these advanced materials. Understanding these details is essential for stakeholders evaluating the integration of this technology into their existing product lines or research programs. The focus is on practical implementation, safety profiles, and the strategic advantages offered by this novel synthetic approach.
Q: What are the thermal stability advantages of these new energetic molecules?
A: The synthesized planar pyrazolo triazine molecules exhibit initial decomposition temperatures exceeding 300°C for certain derivatives, significantly outperforming traditional RDX explosives in thermal stability while maintaining high energy density.
Q: Why is malononitrile preferred over nitroacetonitrile in this synthesis?
A: Malononitrile offers superior chemical stability and long-term storage capabilities compared to nitroacetonitrile, which is difficult to amplify and store. This switch facilitates safer and more reliable commercial scale-up.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the reaction conditions are mild, and the starting materials are commercially available. The process avoids hazardous reagents that hinder amplification, making it highly suitable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrazolo Triazine Supplier
The development of planar pyrazolo triazine energetic molecules marks a significant milestone in the field of high-energy density materials, offering a blend of thermal stability and performance that meets the rigorous demands of modern applications. NINGBO INNO PHARMCHEM, as a leading CDMO expert, possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this technology to market. Our facilities are equipped to handle the specific requirements of energetic material synthesis, ensuring stringent purity specifications and rigorous QC labs are in place to guarantee product consistency. We understand the critical nature of supply chain reliability in the defense and aerospace sectors and are committed to delivering high-quality intermediates that meet your exact specifications.
We invite you to collaborate with us to optimize your supply chain and leverage the benefits of this advanced synthesis route. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs, demonstrating how this technology can reduce your overall manufacturing costs. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your projects. By partnering with us, you gain access to a reliable source of high-purity pyrazolo triazine derivatives that can enhance the performance of your final products.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
