Advanced Synthesis of Gamma-Piperazinepropyl Methyldimethoxysilane for Industrial Silicone Applications
Advanced Synthesis of Gamma-Piperazinepropyl Methyldimethoxysilane for Industrial Silicone Applications
The global demand for high-performance organosilane coupling agents continues to surge, driven by the need for advanced textile finishing and polymer modification. Patent CN101747364B introduces a transformative methodology for preparing gamma-piperazinepropyl methyldimethoxysilane, a critical intermediate in the production of amino-modified polysiloxane emulsions. This specific chemical architecture imparts exceptional softness, elasticity, and smoothness to high-grade fabrics while maintaining breathability. Unlike traditional synthesis routes that struggle with discoloration and low efficiency, this patented approach leverages a refined amination reaction under strict nitrogen protection and controlled vacuum distillation. For R&D Directors and Procurement Managers seeking a reliable gamma-piperazinepropyl methyldimethoxysilane supplier, understanding the technical nuances of this patent is essential for securing a competitive edge in the specialty chemical market. The process not only guarantees a yellowish transparent product but also establishes a robust framework for commercial scale-up of complex silicone intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of amino-containing silane coupling agents, such as N-(β-aminoethyl)-γ-aminopropyl methyl dimethoxysilane, has been plagued by significant technical bottlenecks that hinder cost reduction in electronic chemical manufacturing and textile auxiliary production. Conventional protocols often suffer from poor reaction selectivity, leading to substantial formation of byproducts that are difficult to separate. The most critical failure point in legacy methods is the thermal instability of the product during purification, which frequently results in a dark, undesirable color and a content purity that fails to meet stringent industry standards. Furthermore, typical yields in these older processes hover around a mere 40%, representing a massive inefficiency in raw material utilization. This low yield directly translates to higher waste generation and increased disposal costs, creating a substantial burden on the supply chain. For Supply Chain Heads, the inconsistency of these conventional methods poses a risk to production continuity, as batch-to-batch variability often necessitates costly rework or rejection of entire lots.
The Novel Approach
The methodology disclosed in CN101747364B fundamentally reengineers the production workflow to overcome these historical deficiencies through precise control of reaction kinetics and purification thermodynamics. By utilizing solid anhydrous piperazine and gamma-chloropropyl methyldimethoxysilane in a specific mass ratio range of 120 to 280:100, the process ensures complete conversion of the chlorosilane while minimizing side reactions. The introduction of a nitrogen-protected environment prevents oxidative degradation, which is the primary cause of product discoloration in open systems. Additionally, the strategic implementation of a two-stage reduced pressure distillation allows for the gentle separation of the target molecule from the solvent and byproducts without exposing it to excessive thermal stress. This novel approach not only elevates the yield to over 90% but also ensures the final product is yellowish and transparent, meeting the highest quality benchmarks for reliable agrochemical intermediate supplier standards and beyond. The ability to recover and reuse the solvent and excess piperazine further enhances the economic viability of this route.
Mechanistic Insights into Piperazine Nucleophilic Substitution
The core chemical transformation in this synthesis is a nucleophilic substitution reaction where the secondary amine groups of the piperazine ring attack the electrophilic carbon attached to the chlorine atom in the gamma-chloropropyl methyldimethoxysilane. This reaction is highly sensitive to temperature and stoichiometry; maintaining the reaction temperature between 100°C and 130°C during the滴 addition phase is critical to balance the reaction rate against the potential for thermal decomposition. The use of anhydrous conditions is paramount, as the presence of moisture could lead to the hydrolysis of the methoxy groups on the silicon atom, resulting in premature condensation and gelation. By carefully controlling the滴 addition time to between 1 and 2 hours, the local concentration of the chlorosilane is kept low, preventing exothermic runaway and ensuring a smooth reaction profile. This precise control is what enables the production of high-purity OLED material precursors and similar sensitive intermediates where structural integrity is non-negotiable.
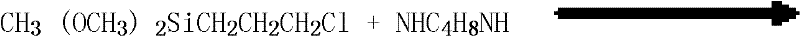
Impurity control is managed through a physical separation strategy rather than complex chemical quenching. The reaction generates piperazine hydrochloride as a stoichiometric byproduct, which precipitates out of the organic solvent phase upon cooling. By filtering the mother liquor at room temperature before distillation, the bulk of the ionic impurities are removed, significantly reducing the load on the distillation column. This step is crucial for achieving the reported content purity of ≥97%. Furthermore, the final vacuum distillation at a deep vacuum of 2 to 6 KPa lowers the boiling point of the product to the 100-130°C range. This low-temperature distillation prevents the "yellowing" phenomenon often associated with amino silanes, ensuring the product remains stable during storage. For R&D teams, this mechanism offers a clear pathway to scaling complex polymer additives without the need for expensive chromatographic purification steps.
How to Synthesize Gamma-Piperazinepropyl Methyldimethoxysilane Efficiently
Implementing this synthesis route requires strict adherence to the operational parameters defined in the patent embodiments to ensure reproducibility and safety. The process begins with the dissolution of solid piperazine in a high-boiling solvent such as xylene or toluene under a nitrogen blanket, followed by the controlled addition of the chlorosilane. The reaction mixture is then maintained at an elevated temperature to drive the substitution to completion. Following the reaction, the mixture is cooled to facilitate the precipitation of the salt byproduct, which is removed via filtration. The filtrate is then subjected to a sequential distillation process to recover solvents and isolate the final product. Detailed standardized synthesis steps see the guide below.
- Dissolve solid anhydrous piperazine in a solvent like xylene under nitrogen protection and heat to 90-120°C.
- Slowly drip gamma-chloropropyl methyldimethoxysilane into the reaction kettle at 100-130°C and stir for 2-6 hours.
- Filter off the piperazine hydrochloride byproduct, then perform reduced pressure distillation to collect the product fraction at 100-130°C.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers profound advantages that directly address the pain points of cost and reliability in the fine chemical sector. The elimination of complex catalytic systems and the reliance on straightforward thermal amination simplifies the manufacturing process, reducing the need for specialized equipment and skilled labor. The high yield achieved minimizes raw material waste, which is a primary driver of variable costs in chemical production. Moreover, the ability to recover and recycle the solvent and excess reactant creates a closed-loop system that drastically reduces the consumption of fresh materials. For Procurement Managers, this translates into a more stable cost structure and protection against volatility in raw material pricing. The robustness of the process also ensures consistent quality, reducing the risk of downstream production failures for customers using this intermediate in their own formulations.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by eliminating the need for expensive transition metal catalysts and complex purification media. The high reaction yield means that less raw material is required per unit of product, directly lowering the bill of materials. Furthermore, the integrated solvent recovery system allows for the reuse of xylene or toluene, substantially reducing waste disposal fees and fresh solvent procurement costs. This efficiency makes the commercial scale-up of complex silicone intermediates economically viable even in markets with strict environmental regulations.
- Enhanced Supply Chain Reliability: The simplicity of the reaction setup, involving standard reactors and distillation columns, ensures that production can be easily scaled or replicated across different manufacturing sites. The use of readily available raw materials like anhydrous piperazine and gamma-chloropropyl methyldimethoxysilane mitigates the risk of supply bottlenecks associated with exotic reagents. By reducing lead time for high-purity silicone additives, manufacturers can respond more agilely to market demand fluctuations. The stability of the process also guarantees consistent batch quality, which is critical for maintaining long-term contracts with downstream textile and polymer manufacturers.
- Scalability and Environmental Compliance: The closed nitrogen-protected system minimizes the release of volatile organic compounds (VOCs) into the atmosphere, aligning with modern environmental compliance standards. The vacuum distillation process is energy-efficient compared to atmospheric distillation, as it operates at lower temperatures. The solid byproduct, piperazine hydrochloride, is easily separated and can potentially be processed for salt recovery or safe disposal, simplifying waste management. This environmental friendliness enhances the sustainability profile of the supply chain, appealing to eco-conscious global brands.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of gamma-piperazinepropyl methyldimethoxysilane. These answers are derived directly from the experimental data and technical specifications outlined in the patent literature. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this intermediate into their supply chains. The information provided here serves as a foundational reference for further technical discussions with our engineering team.
Q: Why is the product color superior in this new method compared to conventional amino silane synthesis?
A: Conventional methods often result in dark-colored products with yields around 40% due to side reactions and thermal degradation. This patent utilizes a specific vacuum distillation protocol (2-6 KPa) and immediate filtration of the hydrochloride salt, preventing thermal discoloration and ensuring a yellowish transparent appearance.
Q: How does the solvent recovery process impact manufacturing costs?
A: The process employs a two-stage vacuum distillation. The first stage (40-70 KPa) recovers the solvent (e.g., xylene or toluene) and excess piperazine for reuse. This closed-loop recovery significantly reduces raw material consumption and waste disposal costs.
Q: What are the critical purity specifications achieved by this route?
A: By optimizing the mass ratio of piperazine to the chlorosilane (120-280:100) and controlling the reaction temperature strictly between 100-140°C, the method achieves a content purity of ≥97% with yields exceeding 90%, suitable for high-grade fabric finishing agents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gamma-Piperazinepropyl Methyldimethoxysilane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality silane coupling agents play in the performance of downstream silicone and textile products. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the laboratory success of patent CN101747364B is translated into industrial reality. Our facilities are equipped with rigorous QC labs and stringent purity specifications to guarantee that every batch of gamma-piperazinepropyl methyldimethoxysilane meets the exacting standards required for high-grade fabric finishing. We are committed to delivering not just a chemical product, but a reliable supply chain solution that enhances your manufacturing efficiency.
We invite you to collaborate with us to optimize your sourcing strategy for this vital intermediate. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and application needs. We encourage you to contact us to request specific COA data and route feasibility assessments, ensuring that our capabilities align perfectly with your project timelines. By partnering with us, you gain access to a stable source of high-purity silicone additives that will drive the quality of your final products.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
