Advanced Two-Step Synthesis of Nitro Compounds for Industrial Scale Manufacturing
The chemical industry is constantly seeking more efficient and safer pathways for synthesizing fundamental building blocks, and the technology disclosed in patent CN112479890A represents a significant leap forward in the preparation of nitro compounds. Traditionally, the production of aliphatic nitro compounds has been plagued by severe safety hazards and environmental concerns associated with direct gas-phase nitration processes. This new methodology introduces a sophisticated two-step synthetic route that bypasses the need for extreme thermal conditions, utilizing readily available alcohol or alkene derivatives as starting materials. By shifting the paradigm from direct hydrocarbon nitration to an activated intermediate strategy, the process achieves superior control over reaction kinetics and product purity. This innovation is particularly critical for manufacturers of specialty solvents and agrochemical intermediates who require consistent quality without the operational risks of high-pressure reactors. The ability to generate high-purity nitro compounds through mild conditions not only enhances workplace safety but also opens new avenues for cost reduction in fine chemical manufacturing by simplifying downstream processing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of nitroalkanes such as 2-nitropropane has relied heavily on the vapor-phase nitration of propane, a process that is inherently dangerous and difficult to control. This conventional method necessitates temperatures exceeding 400°C and involves complex mixtures of nitro products that are challenging to separate, leading to significant yield losses and high energy consumption. Furthermore, the direct nitration of hydrocarbons often results in the formation of toxic by-products and requires extensive waste treatment protocols to manage nitrogen oxide emissions. The equipment required for such high-temperature and high-pressure operations is capital intensive, requiring specialized alloys and rigorous safety monitoring systems that drive up the overall cost of production. Additionally, the poor selectivity of free-radical nitration means that obtaining a single isomer with high purity often requires multiple distillation steps, further eroding profit margins and increasing the carbon footprint of the manufacturing facility.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a stepwise activation strategy that allows for precise molecular construction under much milder conditions. Instead of attacking a stable C-H bond directly, the process first converts a hydroxyl-containing precursor into a highly reactive intermediate, which then undergoes a clean nucleophilic substitution with a nitro source. This method effectively decouples the activation energy requirements, allowing the reaction to proceed at temperatures ranging from -50°C to 200°C, often near ambient conditions. The versatility of this approach is evident in its ability to accommodate a wide range of substrates, producing the general structure shown in Formula I with high fidelity.
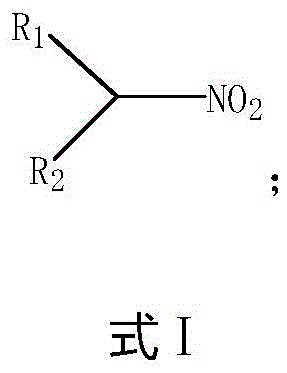
By employing this controlled synthetic pathway, manufacturers can achieve significantly higher yields and purity levels while drastically reducing the complexity of the separation process. The use of liquid-phase reagents also facilitates better heat transfer and mixing, ensuring uniform reaction progress and minimizing the risk of thermal runaway incidents that are common in gas-phase nitration.
Mechanistic Insights into Activated Intermediate Nitration
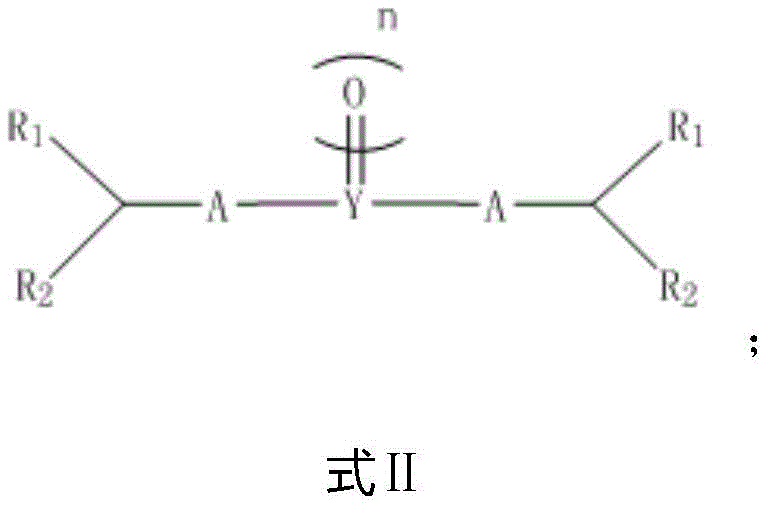
The core mechanistic advantage of this technology lies in the formation of a superior leaving group prior to the introduction of the nitro functionality. In the first step, the starting material, typically an alcohol or an alkene derivative (Compound 1A), reacts with an activating agent (Compound 1B) such as a sulfonyl halide, carbonyl diimidazole, or phosphorus halide. This reaction transforms the poor leaving hydroxyl group into a robust leaving group, such as a sulfonate, carbonate, or phosphate ester, represented structurally by intermediates like Formula II. This activation step is crucial because it lowers the energy barrier for the subsequent nucleophilic attack, enabling the use of milder nitrating agents like sodium nitrite or nitrous acid instead of aggressive mixed acids.
Once the intermediate is formed, the nitration step proceeds via a classic SN2-type mechanism where the nitro anion or equivalent species displaces the activated leaving group. This pathway ensures high regioselectivity, preventing the formation of unwanted isomers that plague free-radical mechanisms. Furthermore, the choice of solvent plays a pivotal role in stabilizing the transition state; polar aprotic solvents like dimethylformamide or acetonitrile can enhance the nucleophilicity of the nitro source, driving the reaction to completion with minimal side reactions. The result is a clean reaction profile that simplifies the impurity spectrum, making the final purification via recrystallization highly effective and efficient.
How to Synthesize 2-Nitropropane Efficiently
The synthesis of specific nitro compounds like 2-nitropropane using this patented method involves a straightforward protocol that balances reaction efficiency with operational simplicity. The process begins with the activation of isopropanol using a reagent like sulfuric acid or sulfuryl chloride to form the corresponding sulfate or sulfonate intermediate. This intermediate is then treated with a nitrite salt in a suitable solvent system, allowing the nitro group to be installed under controlled thermal conditions. The detailed standardized synthesis steps for optimizing yield and purity are outlined in the guide below.
- React compound 1A (alcohol or alkene derivative) with compound 1B (activating agent like sulfonyl halide or carbonyl compound) to form intermediate 2A.
- Treat the isolated intermediate 2A with a nitrating reagent such as nitrite or nitrous acid under mild temperature conditions.
- Purify the crude nitro product through low-temperature recrystallization and rotary evaporation to obtain the refined final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers transformative benefits that extend far beyond simple chemical yield improvements. The shift from high-pressure gas processing to liquid-phase batch chemistry fundamentally alters the capital expenditure landscape, allowing for the use of standard glass-lined or stainless steel reactors that are widely available and easier to maintain. This compatibility with existing infrastructure means that production capacity can be ramped up rapidly without the need for specialized high-pressure vessel certifications, thereby reducing lead time for high-purity nitro compound deployment. Moreover, the reliance on commodity chemicals like alcohols and common inorganic salts as raw materials insulates the supply chain from the volatility associated with petrochemical feedstocks used in traditional cracking and nitration processes.
- Cost Reduction in Manufacturing: The elimination of extreme temperature and pressure requirements leads to substantial energy savings, as the process does not require massive furnaces or compressors to drive the reaction. By avoiding the use of expensive transition metal catalysts or hazardous mixed acids, the raw material costs are significantly lowered, and the expense associated with neutralizing and disposing of corrosive waste streams is drastically reduced. The high selectivity of the reaction minimizes the loss of valuable starting materials to by-products, ensuring that a greater proportion of the input mass is converted into saleable product, which directly improves the gross margin for the manufacturer.
- Enhanced Supply Chain Reliability: Because the starting materials are common industrial alcohols and inorganic reagents, sourcing is stable and less susceptible to the geopolitical disruptions that often affect specialized hydrocarbon feedstocks. The mild reaction conditions also reduce the frequency of unplanned shutdowns caused by equipment stress or safety interlocks, ensuring a consistent and reliable flow of product to downstream customers. This stability is critical for long-term contracts in the pharmaceutical and agrochemical sectors, where continuity of supply is often valued higher than marginal price fluctuations.
- Scalability and Environmental Compliance: The liquid-phase nature of the reaction makes scaling from pilot plant to commercial production linear and predictable, avoiding the non-linear heat transfer issues common in gas-phase nitration. From an environmental perspective, the process generates fewer nitrogen oxide emissions and avoids the creation of complex tarry by-products, simplifying wastewater treatment and helping facilities meet increasingly stringent environmental regulations. The ability to recycle solvents like ethanol or acetone further enhances the sustainability profile of the operation, aligning with corporate goals for green chemistry and reduced carbon intensity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nitration technology, derived directly from the patent specifications and experimental data. Understanding these details is essential for R&D teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers provided reflect the specific advantages of the activated intermediate method over legacy technologies.
Q: What are the advantages of this nitration method over traditional propane nitration?
A: Unlike traditional propane nitration which requires hazardous high-temperature and high-pressure conditions, this method utilizes mild reaction parameters (-50°C to 200°C) and liquid-phase reagents, significantly improving operational safety and simplifying product separation.
Q: What types of intermediates are formed during the synthesis?
A: The process generates activated intermediates such as sulfonates, carbonates, or phosphates (Formula II-VI), which serve as excellent leaving groups for the subsequent nucleophilic substitution by nitro species.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the use of common solvents like ethanol and acetone, combined with standard stainless steel reactors and ambient pressure capabilities, makes this route highly scalable and cost-effective for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nitro Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting advanced synthetic methodologies like the one described in CN112479890A to enhance product quality and operational efficiency. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition to this newer, safer technology is seamless and successful. Our rigorous QC labs and commitment to stringent purity specifications mean that we can deliver nitro compounds that meet the exacting standards required for sensitive applications in electronics and pharmaceuticals. We are dedicated to leveraging our technical expertise to optimize this two-step process, maximizing yield while minimizing environmental impact for our global clientele.
We invite you to collaborate with us to explore how this innovative synthesis route can optimize your supply chain and reduce your overall manufacturing costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your project goals and timeline expectations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
