Revolutionizing Aryl Azide Production: A Green Copper-Catalyzed Route for Commercial Scale-Up
The landscape of organic synthesis for nitrogen-containing heterocycles and active pharmaceutical ingredients (APIs) is undergoing a significant transformation driven by the need for greener, safer, and more cost-effective methodologies. Patent CN102180755A introduces a groundbreaking approach to the synthesis of aryl azide compounds, a critical class of intermediates widely utilized in click chemistry, drug discovery, and material science. This technology departs from conventional hazardous routes by employing a cuprous catalyst system in an aqueous environment, utilizing readily available aryl boronic acids as the starting materials. The core innovation lies in the substitution reaction between aryl boronic acids and alkali metal azides (MN3, where M is Li, Na, K, Rb, or Cs) facilitated by cuprous oxide and ammonia water. This method not only addresses the severe safety concerns associated with traditional azide synthesis, such as the handling of explosive hydrazoic acid derivatives, but also aligns perfectly with modern green chemistry principles by using water as the primary solvent. For R&D directors and process chemists, this represents a robust platform for generating high-purity building blocks with exceptional functional group tolerance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of organic azides has been plagued by significant operational hazards and environmental drawbacks that complicate large-scale manufacturing. Traditional methods often rely on the diazotization of aromatic amines followed by treatment with sodium azide, a process that generates unstable diazonium salts prone to violent decomposition. Alternatively, nucleophilic substitution reactions using halogenated aromatics often require harsh conditions, polar aprotic solvents like DMF or DMSO, and elevated temperatures, which can lead to poor selectivity and difficult purification profiles. Furthermore, methods involving iodine azide or other hypervalent iodine reagents, while effective on a small scale, pose unacceptable explosion risks in industrial settings due to the shock sensitivity of the reagents. These legacy processes frequently result in low atom economy, generate substantial toxic waste streams requiring expensive disposal, and limit the scope of substrates due to incompatibility with sensitive functional groups. Consequently, procurement teams face inflated costs related to safety infrastructure, waste management, and the sourcing of specialized, high-risk reagents.
The Novel Approach
In stark contrast, the methodology disclosed in CN102180755A offers a paradigm shift by leveraging the stability and availability of aryl boronic acids. This novel route operates under remarkably mild conditions, typically between 10-30°C, and utilizes atmospheric pressure in an open system, eliminating the need for expensive high-pressure reactors or inert gas manifolds. The use of water as the solvent not only drastically reduces raw material costs but also simplifies the work-up procedure, as the organic product can often be separated via simple extraction, leaving the inorganic salts and catalyst in the aqueous phase. 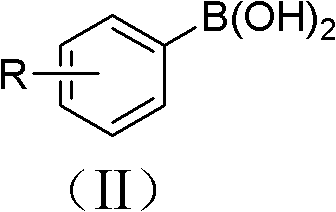 As illustrated by the general structure of the starting materials, the process accommodates a wide variety of substituents on the aromatic ring. The transformation yields the target aryl azide structure, shown below, with high efficiency and purity.
As illustrated by the general structure of the starting materials, the process accommodates a wide variety of substituents on the aromatic ring. The transformation yields the target aryl azide structure, shown below, with high efficiency and purity. 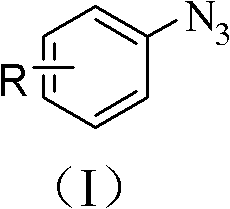 This transition from hazardous halides or amines to stable boronic acids in water fundamentally changes the risk profile and economic feasibility of producing these valuable intermediates.
This transition from hazardous halides or amines to stable boronic acids in water fundamentally changes the risk profile and economic feasibility of producing these valuable intermediates.
Mechanistic Insights into Cu-Catalyzed Azidation
The mechanistic underpinning of this synthesis involves a copper-mediated oxidative coupling that proceeds efficiently in the presence of ammonia water. The cuprous catalyst, preferably cuprous oxide (Cu2O), acts as a Lewis acid to activate the boronic acid species, facilitating the transmetallation or coordination necessary for the subsequent nucleophilic attack by the azide anion. Ammonia water serves a dual purpose: it acts as a ligand to stabilize the copper species in the aqueous medium and potentially assists in the activation of the boron center. The reaction tolerates oxygen, proceeding smoothly in an open system, which suggests a radical or oxidative mechanism that does not require rigorous exclusion of air, a significant advantage for scale-up operations. The catalytic cycle is robust enough to handle electron-rich and electron-deficient substrates alike, ensuring consistent performance across a broad library of chemical space. This mechanistic resilience allows for the synthesis of complex molecules containing sensitive moieties such as aldehydes, esters, and nitro groups without the need for protective group manipulation, thereby shortening synthetic routes and improving overall yield.
Impurity control is inherently superior in this aqueous system due to the phase separation characteristics of the reaction mixture. Byproducts, primarily inorganic boron salts and excess azide, remain dissolved in the aqueous layer, while the lipophilic aryl azide product partitions into the organic extraction solvent. This natural segregation minimizes the carryover of metal contaminants and inorganic salts into the final product, reducing the burden on downstream purification steps like column chromatography. The patent data indicates that simple extraction followed by washing is often sufficient to obtain products of high purity, as evidenced by the clean NMR spectra reported in the examples. Furthermore, the ability to recycle the catalytic system by replenishing alkali salts after extraction demonstrates a closed-loop potential that further minimizes metal waste and ensures consistent batch-to-batch quality, a critical factor for regulatory compliance in pharmaceutical manufacturing.
How to Synthesize Phenyl Azide Efficiently
The practical execution of this synthesis is straightforward and amenable to both laboratory and pilot-plant environments. The general protocol involves charging a reaction vessel with the aryl boronic acid, sodium azide, cuprous oxide catalyst, and aqueous ammonia in water. The mixture is stirred at ambient temperature (typically 15-25°C) for a period ranging from 3 to 48 hours, depending on the electronic nature of the substrate. Upon completion, the reaction is quenched or worked up by adding a base such as sodium hydroxide solution to ensure complete deprotonation and phase separation, followed by extraction with ethyl acetate. The detailed standardized operating procedures, including specific molar ratios (e.g., catalyst:ammonia:boronic acid:azide at 0.1:2.5:1:5) and precise temperature controls for various substrates, are critical for maximizing yield and safety.
- Prepare the reaction mixture by combining aryl boronic acid, sodium azide (NaN3), cuprous oxide (Cu2O) catalyst, and aqueous ammonia in water within a reaction vessel equipped with stirring.
- Maintain the reaction temperature between 10-30°C under atmospheric pressure and open to air, stirring for 3 to 48 hours depending on the substrate reactivity.
- Upon completion, perform an aqueous work-up by extracting the product with ethyl acetate, washing the organic phase, and purifying via standard chromatography or crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this technology translates into tangible strategic benefits beyond mere chemical curiosity. The shift to water as a solvent eliminates the recurring cost of purchasing, recovering, and disposing of large volumes of volatile organic compounds (VOCs), leading to substantial operational expenditure reductions. The use of commodity chemicals like cuprous oxide and sodium azide, which are globally sourced and price-stable, mitigates supply chain volatility compared to specialized organometallic reagents. Moreover, the mild reaction conditions reduce energy consumption for heating and cooling, contributing to a lower carbon footprint and aligning with corporate sustainability goals. The simplified work-up process reduces the demand for silica gel and chromatography solvents, further driving down the cost of goods sold (COGS) and accelerating the throughput of manufacturing campaigns.
- Cost Reduction in Manufacturing: The elimination of expensive anhydrous solvents and the ability to operate at room temperature significantly lowers utility and raw material costs. By avoiding the need for cryogenic conditions or high-pressure equipment, capital expenditure for reactor infrastructure is minimized. The high tolerance for functional groups reduces the number of synthetic steps required to reach the target intermediate, effectively compounding the cost savings by improving overall process mass intensity (PMI). Additionally, the potential for catalyst recycling means that the effective loading of copper per kilogram of product can be drastically reduced over multiple batches, optimizing the utilization of this metal resource.
- Enhanced Supply Chain Reliability: Sourcing aryl boronic acids is generally more reliable and diversified than sourcing specialized halogenated precursors or unstable diazonium salts, as boronic acids are staple reagents in the fine chemical industry. The robustness of the reaction against moisture and oxygen means that storage and handling requirements for intermediates are less stringent, reducing the risk of spoilage during logistics. This stability allows for larger batch sizes and longer campaign runs without the fear of reagent degradation, ensuring a steady flow of materials to downstream synthesis units. The reduced hazard profile also simplifies regulatory approvals for transport and storage, removing bottlenecks that often delay project timelines.
- Scalability and Environmental Compliance: The aqueous nature of the reaction inherently limits the generation of hazardous organic waste, simplifying effluent treatment and ensuring compliance with increasingly strict environmental regulations. The process avoids the formation of explosive byproducts, enhancing plant safety and reducing insurance premiums associated with high-risk chemical operations. Scalability is facilitated by the exotherm control inherent in water-based systems, allowing for safe expansion from gram to ton scales without complex engineering controls. This ease of scale-up ensures that supply can meet surging demand for key intermediates without the long lead times typically associated with developing new high-hazard processes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this copper-catalyzed azidation technology. These insights are derived directly from the experimental data and scope defined in the patent literature, providing a realistic view of the process capabilities. Understanding these nuances is essential for integrating this method into existing production workflows and assessing its fit for specific project requirements.
Q: What are the primary safety advantages of this azide synthesis method compared to traditional routes?
A: Unlike traditional methods that often involve hazardous diazotization or the use of explosive iodine azide, this patent utilizes stable aryl boronic acids and alkali metal azides in a benign aqueous medium, significantly reducing explosion risks and handling hazards.
Q: Can the copper catalyst system be recycled to reduce production costs?
A: Yes, the patent explicitly demonstrates that after product extraction, the remaining aqueous catalytic system can be reused for subsequent batches by simply replenishing the alkali salt and substrates, maintaining catalytic activity over multiple cycles.
Q: What is the functional group tolerance of this copper-catalyzed azidation?
A: The method exhibits high tolerance for diverse functional groups including halogens, nitro, aldehyde, ester, and hydroxyl groups on the aromatic ring, making it suitable for synthesizing complex pharmaceutical intermediates without extensive protecting group strategies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenyl Azide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the copper-catalyzed aqueous synthesis route for aryl azides and have integrated this chemistry into our advanced process development portfolio. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of Phenyl Azide meets the exacting standards required for pharmaceutical applications. Our commitment to quality assurance means that we can deliver high-purity intermediates with consistent impurity profiles, supporting your regulatory filings and clinical trial timelines with confidence.
We invite you to collaborate with us to leverage this green and efficient synthesis technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this aqueous copper-catalyzed route for your supply chain. Please contact us to request specific COA data for our catalog of aryl azides or to discuss route feasibility assessments for custom targets. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable supply of critical intermediates produced via sustainable, cost-effective, and safe manufacturing processes.
