Advanced Enzymatic Synthesis of S-2-Aminobutanamide for Commercial Pharmaceutical Production
The pharmaceutical industry continuously seeks robust and sustainable pathways for producing critical chiral intermediates, and patent CN112195203A presents a significant breakthrough in this domain. This specific intellectual property details a novel enzymatic method for synthesizing (S)-2-aminobutanamide, a pivotal precursor for the antiepileptic drug Levetiracetam. Traditional chemical routes often struggle with environmental hazards and complex purification steps, whereas this biocatalytic approach leverages the high stereoselectivity of specific lipases to achieve superior outcomes. By utilizing a catalytic amount of lipase with the gene sequence SEQ ID NO. 1, the process ensures the formation of the desired S-type chiral configuration with exceptional precision. This innovation addresses the urgent global demand for safer, more efficient, and environmentally friendly manufacturing processes within the fine chemical sector. For R&D directors and supply chain leaders, understanding this technology is crucial for optimizing production pipelines and ensuring the consistent availability of high-quality pharmaceutical intermediates. The method not only simplifies the operational workflow but also aligns with modern green chemistry principles, making it a highly attractive option for large-scale commercial adoption.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical synthesis routes for S-2-aminobutanamide have been plagued by significant safety and efficiency challenges that hinder industrial scalability. Conventional chemical methods often rely on hazardous raw materials such as sodium cyanide and n-propionaldehyde, which pose severe risks to operator safety and require stringent environmental controls. These processes typically involve harsh reaction conditions, including high pressure and alkaline environments, which can lead to unpredictable side reactions and lower overall yields. Furthermore, the need for subsequent splitting treatments to isolate the desired chiral isomer adds considerable complexity and cost to the manufacturing process. The use of expensive starting materials like 2-bromobutyric acid further exacerbates the economic burden, making cost reduction in pharmaceutical intermediates manufacturing difficult to achieve. Additionally, the generation of toxic byproducts and the difficulty in waste treatment create substantial compliance hurdles for production facilities. These limitations collectively result in longer lead times and reduced supply chain reliability, prompting the industry to seek alternative, more sustainable synthetic strategies that can meet rigorous quality and safety standards without compromising economic viability.
The Novel Approach
The enzymatic method described in the patent offers a transformative solution by utilizing biocatalysis to overcome the inherent drawbacks of traditional chemical synthesis. This innovative route employs a specific lipase to catalyze the ammonolysis reaction of (S)-2-aminobutyrate methyl ester, ensuring high stereoselectivity and specificity under mild conditions. By avoiding dangerous reagents and harsh environments, the process significantly enhances operational safety and reduces the environmental footprint associated with production. The use of immobilized lipase allows for easy recovery and reuse of the catalyst, which drastically simplifies the downstream processing and lowers material costs. This approach eliminates the need for complex splitting treatments, as the enzyme naturally favors the formation of the S-type chiral configuration with high optical purity. Consequently, the overall process flow is streamlined, leading to improved efficiency and reduced lead time for high-purity pharmaceutical intermediates. The structural formula of the substrate methyl (S)-2-aminobutyric acid is shown as follows:
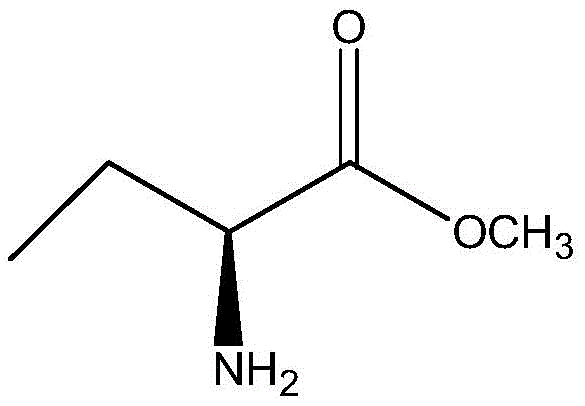
This clear visualization of the starting material highlights the specific chiral center that the enzymatic process targets, ensuring that the integrity of the stereochemistry is maintained throughout the transformation. The simplicity of the substrate structure combined with the specificity of the enzyme creates a highly efficient pathway for generating the target amide, setting a new standard for reliability in pharmaceutical intermediates supplier networks.
Mechanistic Insights into Lipase-Catalyzed Aminolysis
The core of this technological advancement lies in the precise mechanistic action of the lipase encoded by SEQ ID NO. 1, which facilitates a highly selective aminolysis reaction. The enzyme acts as a biocatalyst that effectively promotes the formation of an amino group from the amino donor, such as ammonium carbamate or ammonia gas, onto the substrate without disturbing the existing chiral center. This high optical selectivity ensures that the S-type chiral configuration is preserved and enhanced, resulting in a product with a high ee value and chiral purity. The immobilization of the lipase on methacrylic acid porous resin further stabilizes the enzyme, allowing it to maintain high activity levels over extended reaction periods and multiple cycles. This stability is crucial for maintaining consistent product quality and minimizing batch-to-batch variations, which is a key concern for R&D directors focusing on impurity profiles. The reaction proceeds efficiently in alcohol or ether solvents, which effectively dissolve the substrate while providing a safe and manageable reaction environment. The chemical equation of the method for enzymatically synthesizing (S)-2-aminobutanamide of the invention is shown as follows:
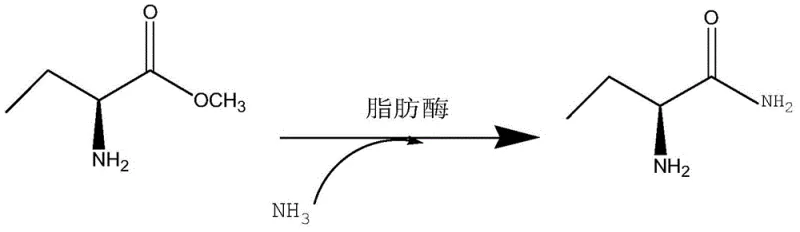
This reaction scheme clearly illustrates the transformation from the ester substrate to the amide product, highlighting the role of ammonia and the lipase catalyst in driving the conversion. The visual representation underscores the simplicity and elegance of the biocatalytic route, where the enzyme facilitates the bond formation with minimal side reactions. This mechanistic clarity provides confidence in the reproducibility of the process, ensuring that the commercial scale-up of complex pharmaceutical intermediates can be achieved with predictable outcomes and high fidelity to the desired molecular structure.
Impurity control is inherently built into this enzymatic process due to the high specificity of the biocatalyst and the mild reaction conditions employed. Unlike chemical methods that often generate a wide range of byproducts requiring extensive purification, the lipase-catalyzed reaction produces a cleaner crude product with fewer impurities. The use of activated carbon for decolorization in the post-treatment step further refines the product quality by adsorbing trace contaminants and improving the visual appearance of the final solid. This reduction in impurity load simplifies the analytical validation process and ensures that the final product meets stringent purity specifications required for pharmaceutical applications. The ability to achieve chiral purity levels exceeding 99.5% without complex chiral resolution steps is a testament to the effectiveness of the enzyme in discriminating between stereoisomers. For quality assurance teams, this means a more robust control strategy with fewer critical process parameters to monitor, leading to greater overall process reliability. The combination of high conversion rates and minimal byproduct formation makes this method particularly suitable for producing high-purity pharmaceutical intermediates that demand exacting quality standards.
How to Synthesize S-2-Aminobutanamide Efficiently
Implementing this synthesis route requires careful attention to reaction parameters and post-processing steps to maximize yield and purity. The process begins with the preparation of the reaction mixture, where the substrate and amino donor are combined in a suitable solvent system under controlled conditions. The addition of the immobilized lipase initiates the catalytic cycle, which must be maintained at an optimal temperature range to ensure maximum enzyme activity and stability. Detailed standard operating procedures are essential to guide the technical team through the reaction monitoring, catalyst recovery, and product isolation phases. The following guide outlines the critical steps involved in executing this synthesis effectively, ensuring that the theoretical benefits of the patent are realized in practical production settings. Adhering to these standardized steps is vital for achieving the high conversion rates and chiral purity demonstrated in the patent examples.
- Prepare the reaction mixture by combining substrate (S)-2-aminobutyrate methyl ester with an amino donor like ammonium carbamate in an alcohol solvent.
- Add immobilized lipase (SEQ ID NO. 1) to the mixture and maintain the temperature between 35°C and 55°C for 24 to 26 hours to ensure complete conversion.
- Filter to recover the enzyme, decolorize the filtrate with activated carbon, and distill under reduced pressure to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
This enzymatic technology offers substantial strategic benefits for procurement and supply chain management by addressing key cost and reliability pain points. The elimination of hazardous reagents and the simplification of the process flow directly contribute to a safer working environment and reduced regulatory compliance costs. By utilizing immobilized enzymes that can be recycled, the consumption of expensive catalytic materials is significantly lowered, leading to direct cost savings in raw material procurement. The mild reaction conditions also reduce energy consumption compared to high-temperature or high-pressure chemical processes, further enhancing the economic efficiency of the manufacturing operation. These factors collectively improve the overall cost structure, making the production of S-2-aminobutanamide more competitive in the global market. For supply chain heads, the robustness of the process ensures consistent output and minimizes the risk of production delays caused by safety incidents or equipment failures. This reliability is essential for maintaining continuous supply to downstream drug manufacturers and meeting the demanding schedules of the pharmaceutical industry.
- Cost Reduction in Manufacturing: The enzymatic route eliminates the need for expensive and dangerous chemical reagents like sodium cyanide, which significantly lowers raw material costs and waste disposal expenses. The ability to recover and reuse the immobilized lipase multiple times reduces the recurring cost of catalysts, contributing to substantial long-term savings. Furthermore, the simplified downstream processing reduces the need for complex purification equipment and solvents, lowering capital and operational expenditures. These efficiencies translate into a more favorable cost profile for the final intermediate, allowing for better margin management and competitive pricing strategies in the market.
- Enhanced Supply Chain Reliability: The use of readily available and safe raw materials ensures a stable supply base that is less susceptible to market fluctuations or regulatory restrictions. The robust nature of the enzymatic process minimizes the risk of batch failures, ensuring consistent production volumes and reliable delivery schedules. This stability is crucial for building long-term partnerships with pharmaceutical clients who require guaranteed supply continuity for their drug manufacturing pipelines. By reducing the dependency on hazardous chemicals, the supply chain becomes more resilient to safety audits and environmental regulations, further securing the production timeline.
- Scalability and Environmental Compliance: The mild conditions and aqueous-compatible nature of the biocatalytic process make it highly scalable from laboratory to industrial production without significant re-engineering. The reduction in toxic waste generation aligns with strict environmental regulations, reducing the burden of waste treatment and compliance reporting. This green manufacturing profile enhances the corporate sustainability image and meets the increasing demand for eco-friendly pharmaceutical production methods. The ease of scale-up ensures that production capacity can be expanded rapidly to meet growing market demand without compromising product quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this enzymatic synthesis method. These answers are derived directly from the patent data and provide clarity on the process capabilities and advantages for potential partners. Understanding these details is essential for making informed decisions about adopting this technology for commercial production. The information covers aspects of purity, process safety, and scalability, which are critical for stakeholders evaluating the feasibility of this route. This section aims to resolve uncertainties and provide a comprehensive overview of the value proposition offered by this innovative synthesis method.
Q: What are the advantages of the enzymatic method over chemical synthesis for S-2-aminobutanamide?
A: The enzymatic method avoids harsh conditions and dangerous raw materials like sodium cyanide used in traditional chemical routes. It offers high stereoselectivity, producing S-type chiral configurations with high ee values without the need for complex splitting treatments.
Q: How does the immobilized lipase contribute to process efficiency?
A: Immobilized lipase ensures high enzyme activity and catalytic capability while being easy to recycle and reuse. This significantly reduces the utilization cost of raw and auxiliary materials and simplifies the post-reaction separation process.
Q: What is the expected chiral purity of the product using this method?
A: The method achieves high chiral purity with ee values reaching up to 99.7%. The lipase maintains the chiral configuration of the amino group in the substrate, effectively preventing conversion and ensuring consistent optical selectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable S-2-Aminobutanamide Supplier
NINGBO INNO PHARMCHEM stands ready to leverage this advanced enzymatic technology to deliver high-quality S-2-Aminobutanamide to the global market. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and efficiency. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch meets the highest industry standards. We understand the critical nature of pharmaceutical intermediates and are committed to providing a reliable supply chain that supports your drug development and manufacturing goals. Our technical team is well-versed in the nuances of biocatalytic processes and can optimize the synthesis parameters to maximize yield and purity for your specific requirements.
We invite you to collaborate with us to optimize your supply chain and reduce costs through this innovative synthesis route. Contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your production volumes and quality needs. We are prepared to provide specific COA data and route feasibility assessments to demonstrate the viability of this method for your projects. By partnering with us, you gain access to cutting-edge technology and a dedicated team focused on delivering value and reliability. Let us help you secure a sustainable and efficient supply of this critical intermediate for your pharmaceutical applications.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
