Advanced Synthesis of Fluorene Oligomers for Next-Generation OLED Display Manufacturing
Advanced Synthesis of Fluorene Oligomers for Next-Generation OLED Display Manufacturing
The rapid evolution of the organic optoelectronics industry demands materials that bridge the gap between the processability of polymers and the purity of small molecules. Patent CN1737080A introduces a groundbreaking class of fluorene oligomer electroluminescent materials that utilize fluorene or anthracene as primary building units to create conjugated segments of varying lengths. These segments are subsequently end-capped with linear or branched groups to finalize the material structure. This innovation addresses the critical instability and efficiency issues plaguing traditional blue-emitting materials in organic light-emitting diodes (OLEDs). By employing a modular synthetic approach, manufacturers can achieve precise control over the electronic properties of the material, ensuring consistent performance in high-end display applications. The strategic design of these oligomers allows for tunable energy gaps and improved solubility, making them highly attractive for solution-processable device fabrication.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polyfluorenes relied heavily on oxidative coupling methods, such as those utilizing iron trichloride (FeCl3). While initially promising, these conventional pathways suffer from severe drawbacks that render the resulting materials unsuitable for commercial high-performance displays. The polymers produced often exhibit low molecular weights and significant branching, which disrupts the conjugation system essential for efficient charge transport. More critically, the residual iron ions from the catalyst act as potent exciton quenchers, strongly absorbing the generated light and drastically reducing the luminous efficiency of the device. Furthermore, the polydispersity inherent in these polymerization reactions leads to batch-to-batch variability, making it nearly impossible to guarantee the strict color purity and lifetime requirements demanded by modern display manufacturers. The tendency of the C-9 position in fluorene to oxidize into carbonyl groups creates deep trap states that further degrade device performance over time.
The Novel Approach
The methodology outlined in the patent represents a paradigm shift towards precision synthesis using Suzuki coupling reactions. Instead of indiscriminate polymerization, this approach constructs the material block-by-block, starting with the synthesis of conjugated fragments containing active bromine end groups. Parallel to this, specific capping groups (S) are synthesized and converted into boration products. The final assembly occurs through a palladium-catalyzed cross-coupling between the brominated segments and the boron-functionalized caps. This strategy ensures that the resulting oligomers have a clearly defined molecular structure with a narrow molecular weight distribution, typically consisting of 1 to 3 repeating units. The elimination of transition metal residues like iron and the ability to purify intermediates at each step result in materials with exceptional purity. This structural definition directly translates to superior electroluminescent properties, including higher quantum efficiency and enhanced operational stability.
Mechanistic Insights into Suzuki-Catalyzed Oligomerization
The core of this synthetic breakthrough lies in the meticulous application of the Suzuki-Miyaura cross-coupling reaction, a powerful tool for forming carbon-carbon bonds between aryl halides and organoboron compounds. The mechanism begins with the oxidative addition of a zero-valent palladium catalyst, such as tetrakis(triphenylphosphine)palladium(0), to the carbon-halogen bond of the fluorene or anthracene derivative. This forms an organopalladium intermediate which then undergoes transmetallation with the organoborate species generated from the capping group precursors. The use of mild bases like sodium carbonate facilitates the activation of the boron species, enabling the transfer of the organic group to the palladium center. Finally, reductive elimination releases the coupled product and regenerates the active palladium catalyst. This cycle is highly tolerant of functional groups, allowing for the incorporation of diverse substituents like long-chain alkyl groups at the C-9 position of fluorene to improve solubility without compromising the electronic integrity of the conjugated backbone.
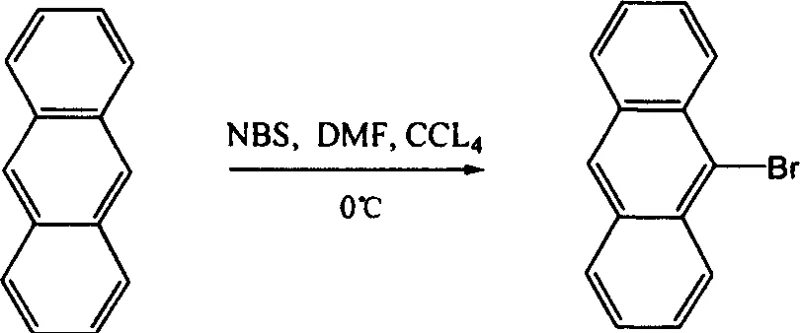
Impurity control is paramount in the production of electroluminescent materials, as even trace contaminants can act as quenching sites. The stepwise nature of this oligomer synthesis allows for rigorous purification of intermediates before the final coupling. For instance, the bromination of anthracene to form 9-bromoanthracene can be carefully monitored to prevent poly-bromination, ensuring a single reactive site. Similarly, the conversion of aryl bromides to boronic acids via lithiation followed by quenching with trialkyl borates must be conducted under strictly anhydrous and oxygen-free conditions to prevent protodeboronation or homocoupling side reactions. By isolating and characterizing intermediates such as the silyl-protected fluorene derivatives or the specific capping groups, manufacturers can ensure that only the desired structural motifs enter the final polymerization step. This level of control effectively eliminates the "trap" states associated with carbonyl defects and metal residues, resulting in a material with a clean emission spectrum and prolonged device lifetime.
How to Synthesize Fluorene Oligomer Efficiently
The synthesis of these advanced materials requires a disciplined approach to reaction conditions and reagent quality. The process generally involves three distinct phases: the preparation of the halogenated conjugated core, the synthesis of the boron-functionalized end caps, and the final coupling. Each step demands strict exclusion of moisture and oxygen, typically achieved through Schlenk line techniques or glovebox operations. The choice of solvent, often toluene or tetrahydrofuran, and the precise stoichiometry of the palladium catalyst are critical for maximizing yield and minimizing homocoupling byproducts. Detailed standard operating procedures for scaling this chemistry from gram-scale laboratory synthesis to industrial production are essential for maintaining consistency.
- Synthesize conjugated segments containing active bromine end groups using fluorene or anthracene building blocks.
- Prepare capping groups (S) containing bromine end groups and convert them into boration products using butyllithium.
- Perform the final Suzuki coupling reaction between the brominated conjugated segments and the boration capping groups using a palladium catalyst to obtain the target oligomer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from ill-defined polymers to structured oligomers offers tangible strategic benefits beyond mere performance metrics. The ability to source materials with guaranteed purity specifications reduces the risk of downstream device failure, which is a costly liability in the consumer electronics sector. Furthermore, the modular nature of the synthesis allows for flexible manufacturing; the same conjugated core can be paired with different end-caps to tune emission colors or solubility profiles without redesigning the entire supply chain. This flexibility enhances supply chain resilience, as raw material sourcing can be optimized for specific sub-components rather than relying on a single, complex monolithic polymer precursor.
- Cost Reduction in Manufacturing: The elimination of expensive and difficult-to-remove transition metal catalysts like iron significantly lowers downstream processing costs. Traditional methods require extensive purification steps to remove metal residues that quench luminescence, whereas the Suzuki coupling uses palladium which can be efficiently scavenged or used in lower quantities due to high turnover numbers. Additionally, the higher yield and selectivity of the stepwise oligomerization reduce waste generation and solvent consumption, leading to substantial cost savings in raw material utilization and waste disposal. The defined structure also means less material is wasted on non-emissive fractions that often plague polydisperse polymer batches.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as fluorene, anthracene, and common boron reagents ensures a stable and robust supply chain. Unlike proprietary monomers that may have single-source bottlenecks, the building blocks for these oligomers are widely available in the global fine chemical market. The synthetic route is robust and scalable, reducing the lead time for high-purity electroluminescent materials. By decoupling the synthesis of the core and the end-caps, production can be parallelized, further accelerating time-to-market for new display formulations and ensuring continuity of supply even if one part of the synthesis faces temporary disruptions.
- Scalability and Environmental Compliance: The Suzuki reaction is well-established in industrial settings, facilitating a smoother scale-up from pilot plant to commercial production volumes. The reaction conditions are relatively mild compared to harsh oxidative polymerizations, reducing energy consumption and safety risks associated with exothermic runaway reactions. Moreover, the ability to recycle solvents and the reduced generation of heavy metal waste align with increasingly stringent environmental regulations. This compliance minimizes the regulatory burden on manufacturing sites and supports the sustainability goals of major electronics brands seeking greener supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these fluorene-based oligomers. Understanding these nuances is critical for R&D teams evaluating material integration and procurement officers assessing vendor capabilities. The answers are derived directly from the technical specifications and experimental data provided in the underlying patent literature, ensuring accuracy and relevance to real-world manufacturing scenarios.
Q: Why are fluorene oligomers preferred over polyfluorenes for blue emission?
A: Polyfluorenes synthesized via oxidative coupling often suffer from low molecular weight, serious branching, and residual iron ions that absorb excitons, reducing luminous efficiency. Oligomers offer defined structures, higher purity, and avoid these trap states, leading to stable blue emission.
Q: What is the primary advantage of the Suzuki coupling method in this context?
A: The Suzuki reaction allows for precise control over molecular weight and conjugation length with narrow distribution. It operates under non-toxic, easily controlled conditions, yielding materials with very high purity compared to traditional oxidative polymerization.
Q: Can these materials be used for applications other than OLEDs?
A: Yes, besides organic electroluminescent devices, these fluorene oligomers are suitable for photovoltaic cells, information storage devices, and thin-film transistors due to their excellent charge transport and optical properties.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluorene Oligomer Supplier
The synthesis of high-performance fluorene oligomers represents a sophisticated intersection of organic synthesis and materials science, requiring a partner with deep technical expertise and robust manufacturing capabilities. NINGBO INNO PHARMCHEM stands at the forefront of this field, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with state-of-the-art reactors capable of handling air-sensitive chemistry and rigorous QC labs that enforce stringent purity specifications essential for OLED applications. We understand that the difference between a lab curiosity and a commercial product lies in the details of process optimization and impurity control, areas where our team excels.
We invite you to collaborate with us to optimize your material supply chain and reduce your overall cost of goods. Whether you require custom synthesis of specific oligomer variants or large-scale production of standard intermediates, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing prowess can accelerate your product development cycle.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
