Advanced CoSalen-Catalyzed Oxidation for High-Purity Symmetrical Disulfide Intermediates
The pharmaceutical and agrochemical industries rely heavily on the precise synthesis of sulfur-containing intermediates, particularly symmetrical disulfides, which serve as critical building blocks for active pharmaceutical ingredients and functional materials. A significant technological advancement in this domain is detailed in patent CN101928193A, which discloses a novel catalytic oxidation method utilizing CoSalen complexes. This innovation addresses long-standing challenges in the oxidative coupling of mercaptans and thiophenols by employing air or oxygen as a clean oxidant in the presence of specific cobalt Schiff base catalysts. The process operates under mild conditions, typically ranging from 20°C to 100°C, and demonstrates remarkable versatility across a wide substrate scope including substituted phenyl thiols and heterocyclic mercaptans. By shifting away from harsh stoichiometric oxidants to catalytic aerobic oxidation, this technology offers a pathway to greener, more sustainable manufacturing processes that align with modern environmental regulations while maintaining high product integrity.
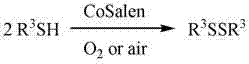
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the development of the CoSalen-catalyzed system, the industrial preparation of symmetrical disulfides was often plagued by significant operational and economic inefficiencies. Traditional methods frequently relied on manganese (III) Schiff base complexes which, while effective, presented substantial difficulties in catalyst preparation, thereby increasing the barrier to entry for large-scale adoption. Furthermore, alternative approaches utilizing metal alkyl compounds in conjunction with copper chloride systems were notoriously sensitive to environmental factors; these reagents required rigorous exclusion of moisture and oxygen, necessitating the use of specialized, expensive reaction vessels and inert atmosphere techniques. Such constraints not only inflated capital expenditure but also introduced complex safety protocols that slowed down production throughput. The reliance on stoichiometric amounts of heavy metal oxidants in older methodologies also generated considerable quantities of hazardous waste, creating a burden on waste treatment facilities and complicating regulatory compliance for manufacturers seeking to minimize their environmental footprint.
The Novel Approach
The methodology described in patent CN101928193A represents a paradigm shift by introducing a robust catalytic system that eliminates the need for sensitive reagents and harsh conditions. By utilizing CoSalen complexes, specifically N,N′-bis(salicylidene)ethylenediamine cobalt and its methoxy-substituted derivatives, the process achieves high conversion rates under ambient pressure using merely air or oxygen. This novel approach simplifies the reactor setup, allowing reactions to proceed in standard glassware or stainless steel reactors without the need for rigorous inert gas protection. The operational simplicity extends to the workup phase, where the removal of the solvent yields the crude product which can be easily purified via recrystallization or standard chromatography. This transition from sensitive, multi-step preparative methods to a direct, aerobic catalytic oxidation significantly reduces the complexity of the manufacturing workflow, making it an attractive option for producers aiming to streamline their supply chains and reduce overall processing time while maintaining exceptional product quality.
Mechanistic Insights into CoSalen-Catalyzed Aerobic Oxidation
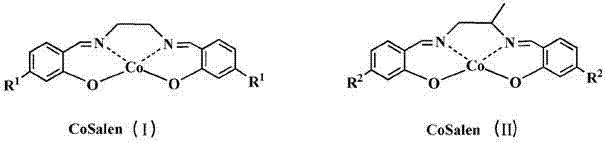
The efficacy of this synthesis route lies in the unique electronic and structural properties of the CoSalen catalyst family. As illustrated in the catalyst structures below, the cobalt center is coordinated within a tetradentate Schiff base ligand framework, which stabilizes the metal in various oxidation states crucial for the redox cycle. The presence of electron-donating groups, such as the methoxy substituents shown in Formula (II), can fine-tune the electron density at the cobalt center, potentially enhancing the activation of molecular oxygen. During the catalytic cycle, the CoSalen complex facilitates the single-electron transfer from the thiol substrate to the oxygen molecule, generating thiyl radicals that subsequently dimerize to form the disulfide bond. This mechanism avoids the formation of over-oxidized byproducts like sulfonic acids, which are common impurities in non-selective oxidation processes. The stability of the Co-N and Co-O bonds within the catalyst structure ensures that the metal does not leach significantly into the product stream, thereby simplifying downstream purification and ensuring the final intermediate meets stringent heavy metal specifications required for pharmaceutical applications.
Impurity control is a critical aspect of this mechanistic pathway, particularly for clients requiring high-purity intermediates for drug synthesis. The selective nature of the CoSalen catalyst minimizes side reactions such as C-S bond cleavage or aromatic ring oxidation, which can occur with stronger, non-selective oxidants. The patent data indicates that by carefully controlling the molar ratio of catalyst to substrate (typically between 1:0.01 and 1:0.1) and monitoring the reaction via TLC, operators can halt the process precisely at the disulfide stage. This kinetic control prevents the accumulation of polysulfides or other sulfur-based impurities that are difficult to separate. Furthermore, the choice of solvent plays a pivotal role in solubilizing the intermediate radical species while maintaining catalyst stability; solvents like ethanol and ethyl acetate provide an optimal balance of polarity and coordinating ability. The result is a reaction profile that consistently delivers products with purity levels often exceeding 98%, reducing the need for extensive and yield-eroding purification steps.
How to Synthesize Symmetrical Disulfides Efficiently
The practical implementation of this technology involves a straightforward sequence of mixing, reacting, and isolating, designed to be easily scalable from laboratory benchtop to industrial pilot plants. The process begins with the dissolution of the thiol substrate and the pre-synthesized CoSalen catalyst in a suitable organic solvent, followed by the introduction of an oxygen source. Reaction conditions are mild, typically requiring temperatures between 20°C and 100°C depending on the specific substrate reactivity, with reaction times ranging from 0.3 to 10 hours. The versatility of the method allows for the use of diverse solvents including alcohols, esters, and halogenated hydrocarbons, providing flexibility for process optimization. Detailed standardized operating procedures regarding specific stoichiometry, temperature ramps, and purification protocols are essential for ensuring batch-to-batch consistency and maximizing yield.
- Prepare the reaction mixture by combining the thiol substrate (Formula III) and the CoSalen catalyst (Formula I or II) in an organic solvent such as ethanol or ethyl acetate.
- Introduce air or oxygen into the reaction vessel and maintain the temperature between 20°C and 100°C, monitoring progress via TLC until completion.
- Upon completion, cool the reaction, remove the solvent via evaporation, and purify the crude symmetrical disulfide through recrystallization or column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the CoSalen-catalyzed oxidation method offers tangible strategic benefits that extend beyond mere chemical efficiency. The primary advantage lies in the drastic simplification of the raw material portfolio; by replacing expensive, air-sensitive metal alkyls and complex manganese catalysts with stable, easily synthesized CoSalen complexes and ubiquitous air, companies can significantly reduce their dependency on specialized chemical suppliers. This shift mitigates supply risk and enhances the resilience of the production line against market volatility. Moreover, the use of air or oxygen as the terminal oxidant eliminates the cost associated with purchasing and disposing of stoichiometric chemical oxidants, leading to substantial cost savings in raw material procurement. The simplified reactor requirements also mean that existing manufacturing infrastructure can often be utilized without major retrofitting, accelerating the time-to-market for new disulfide-based intermediates.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the elimination of costly reagents and the reduction of waste disposal fees. Since the catalyst loading is low and the oxidant is essentially free (air), the variable cost per kilogram of product is significantly lowered compared to traditional methods. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, further contributing to overall operational expenditure savings. The high selectivity of the reaction minimizes the loss of valuable starting materials to byproducts, improving the overall atom economy of the process.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of commodity chemicals as solvents and oxidants, which are readily available globally and less prone to shortage than specialized organometallic reagents. The robustness of the CoSalen catalyst allows for longer shelf life and easier storage conditions, reducing inventory management complexities. This reliability ensures that production schedules can be maintained consistently, reducing lead times for high-purity symmetrical disulfides and enabling manufacturers to respond more agilely to customer demand fluctuations.
- Scalability and Environmental Compliance: From an environmental perspective, the process generates minimal hazardous waste, aligning with increasingly strict global regulations on chemical manufacturing emissions. The absence of heavy metal waste streams associated with stoichiometric oxidants simplifies effluent treatment and reduces the environmental compliance burden. This green chemistry profile not only lowers regulatory risk but also enhances the corporate sustainability image, which is increasingly important for securing contracts with major multinational pharmaceutical partners who prioritize eco-friendly supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic oxidation technology. These insights are derived directly from the experimental data and claims within patent CN101928193A, providing a factual basis for evaluating the feasibility of this method for your specific production needs. Understanding these nuances is crucial for R&D teams planning process validation and for procurement teams assessing vendor capabilities.
Q: What are the advantages of CoSalen catalysts over traditional metal alkyl systems?
A: Unlike traditional metal alkyl and copper chloride systems which are highly sensitive to water and air requiring special containers, CoSalen complexes are stable, easy to prepare, and function effectively under ambient atmospheric conditions.
Q: Is the oxidant used in this process cost-effective for large scale production?
A: Yes, the process utilizes air or molecular oxygen as the oxidant, which are significantly cheaper and more environmentally benign compared to stoichiometric chemical oxidants, drastically reducing raw material costs.
Q: What level of purity can be achieved with this synthesis method?
A: The method yields symmetrical disulfides with exceptionally low impurity content, often achieving purity levels exceeding 98% after simple purification steps like recrystallization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Symmetrical Disulfide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your final drug products. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to full-scale manufacturing is seamless and efficient. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of symmetrical disulfide meets the highest industry standards. Our commitment to process excellence means we can leverage advanced catalytic technologies like the CoSalen system to deliver cost-effective solutions without compromising on quality or delivery timelines.
We invite you to collaborate with us to optimize your supply chain for sulfur-containing intermediates. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity targets. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can support your long-term strategic goals.
