Advanced Trifunctional Photoinitiator HTH: Technical Breakthroughs and Commercial Scalability
Introduction to Next-Generation Photoinitiator Technology
The landscape of radiation curing technology is undergoing a significant transformation driven by the demand for higher performance and stricter safety standards, particularly in food packaging and medical applications. Patent CN110724060B introduces a groundbreaking trifunctional photoinitiator, designated as HTH, which addresses the critical limitations of traditional small-molecule initiators regarding migration and volatility. This novel compound, with a molecular formula of C43H44O13 and a molecular weight of 769.82, represents a strategic evolution from monofunctional systems like Darocur 1173 to complex macromolecular architectures. By integrating three active photoinitiating groups into a single molecular framework, this technology ensures that the resulting cured films exhibit exceptional thermal stability and minimal extractable residues. For R&D directors and procurement specialists seeking a reliable photoinitiator supplier, understanding the structural advantages of HTH is paramount for developing next-generation UV curing formulations that comply with rigorous global safety regulations.
The development of HTH is not merely an incremental improvement but a fundamental rethinking of how photoinitiators interact within a polymer matrix. Traditional small molecules, while effective at generating radicals, often suffer from high mobility, leading to potential contamination of the cured product. The patent data highlights that HTH effectively locks the photoactive components into the cross-linked network, drastically reducing the risk of migration into food or biological tissues. This capability positions HTH as a critical component for high-purity photoinitiators required in sensitive industries. Furthermore, the synthesis route described in the patent utilizes established chemical building blocks, suggesting a viable pathway for cost reduction in UV curing manufacturing without compromising on the sophisticated performance metrics required by modern coating and ink applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Conventional photoinitiators, such as the widely used Darocur 1173 and Darocur 2959, have served the industry well for decades but possess inherent drawbacks that become problematic in high-performance applications. The primary issue lies in their relatively low molecular weight, which facilitates high mobility within the cured polymer network. When these small molecules are physically trapped rather than chemically bonded, they can migrate to the surface or leach out upon contact with solvents, hot water, or grease. This phenomenon poses significant sanitation potential safety hazards, especially in food packaging and medical device manufacturing where extractables and leachables are strictly regulated. Additionally, the volatility of these small molecules can lead to odor issues and loss of initiator concentration during storage or high-temperature processing, resulting in inconsistent curing performance and compromised final product quality. The reliance on these monofunctional systems often forces formulators to choose between curing speed and safety, a trade-off that is increasingly unacceptable in the current regulatory environment.
The Novel Approach
The novel approach presented in patent CN110724060B overcomes these deficiencies by engineering a trifunctional architecture that combines the high reactivity of alpha-hydroxy ketones with the steric bulk of a central trimellitic core. By covalently linking one molecule of Darocur 1173 and two molecules of Darocur 2959 through ester linkages, the resulting HTH molecule achieves a molecular weight nearly three times that of its precursors. This increase in size dramatically reduces the diffusion coefficient of the initiator within the resin matrix, effectively anchoring it in place. Unlike polymerizable initiators that rely on vinyl groups which can complicate formulation stability, HTH maintains the simplicity of a cleavage-type initiator while offering the migration resistance of a macromolecule. This design ensures that the photoinitiating activity is preserved, as the alpha-hydroxy ketone groups remain intact and capable of efficient Norrish Type I cleavage, while the physical properties of the cured film are vastly improved. This strategic molecular design offers a compelling solution for the commercial scale-up of complex polymer additives that require both high reactivity and low migration.
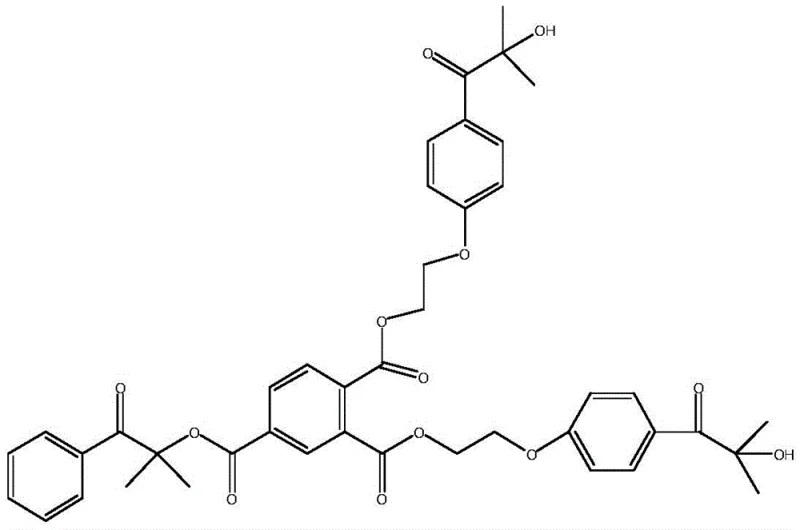
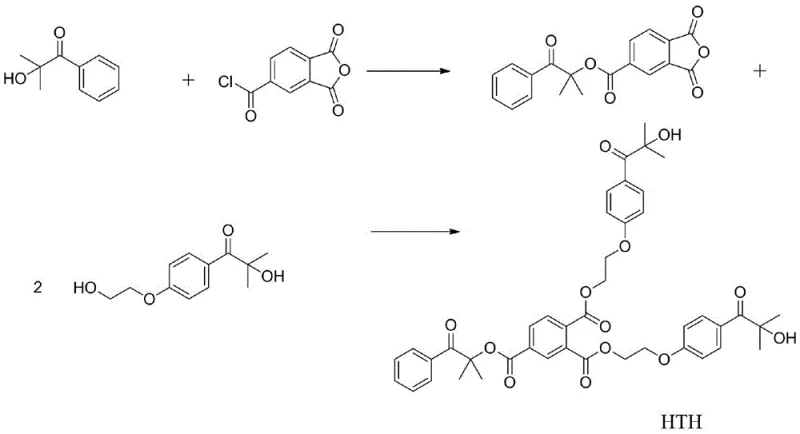
Mechanistic Insights into Esterification and Photoinitiation
The synthesis of HTH relies on a robust two-step esterification strategy that leverages the reactivity of acid chlorides and hydroxyl groups. In the first step, Darocur 1173 reacts with chlorinated trimellitic anhydride in the presence of a base like triethylamine. This reaction selectively forms a mono-ester intermediate that retains a free anhydride group, setting the stage for the subsequent coupling. The second step involves the ring-opening of this anhydride by two equivalents of Darocur 2959, catalyzed by methanesulfonic acid at elevated temperatures around 105°C. This sequential addition allows for precise control over the stoichiometry, ensuring that the final product contains the desired 1:2 ratio of the two initiator types. The use of methanesulfonic acid as a catalyst is particularly advantageous as it is a strong organic acid that promotes esterification without inducing unwanted side reactions on the sensitive alpha-hydroxy ketone moieties. This mechanistic pathway ensures high purity and structural integrity, which are critical for consistent photoinitiation performance.
Upon exposure to UV radiation, the HTH molecule undergoes Norrish Type I cleavage at the alpha-carbon bonds adjacent to the carbonyl groups of the Darocur 1173 and 2959 segments. Because the molecule contains three such active sites, the local concentration of free radicals generated per molecule is significantly higher than that of monofunctional analogs. This high radical flux is instrumental in overcoming oxygen inhibition, a common challenge in UV curing where atmospheric oxygen scavenges initiating radicals. The patent data indicates that HTH exhibits faster polymerization rates for TMPTA compared to Darocur 1173 alone, attributed to this enhanced radical density. Furthermore, the bulky central core may impose steric constraints that influence the recombination of radical pairs, potentially extending the lifetime of the active species. This combination of high radical yield and restricted mobility makes HTH an ideal candidate for reducing lead time for high-purity photoinitiators in demanding industrial applications where rapid throughput and deep cure are essential.
How to Synthesize Trifunctional Photoinitiator HTH Efficiently
The synthesis of HTH is designed to be operationally simple, utilizing standard laboratory glassware and commercially available solvents, which facilitates easy translation to pilot and production scales. The process begins with the formation of the key anhydride intermediate under mild conditions, followed by a thermal coupling step that drives the reaction to completion. Detailed protocols emphasize the importance of purification via silica gel chromatography to remove unreacted starting materials and byproducts, ensuring the final yellow powder meets stringent quality specifications. For process engineers looking to implement this technology, the clear definition of molar ratios and solvent systems in the patent provides a solid foundation for optimization. The detailed standardized synthesis steps are outlined below to guide your technical team in replicating this high-value intermediate.
- React Darocur 1173 with chlorinated trimellitic anhydride in dichloromethane with triethylamine to form the anhydride intermediate.
- Purify the intermediate via silica gel column chromatography using petroleum ether and ethyl acetate.
- React the intermediate with two equivalents of Darocur 2959 in DMF with methanesulfonic acid catalyst at 105°C to yield HTH.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of HTH represents a strategic opportunity to enhance product value while mitigating regulatory risks. The shift towards low-migration photoinitiators is not just a technical preference but a market necessity driven by global food safety standards. By sourcing a trifunctional initiator like HTH, companies can future-proof their formulations against tightening regulations regarding extractables. The synthesis route described relies on commodity chemicals like Darocur 1173 and 2959, which are produced in vast quantities globally, ensuring a stable and resilient supply chain. This reliance on established feedstocks minimizes the risk of raw material shortages that often plague niche specialty chemicals. Furthermore, the elimination of complex metal catalysts or exotic reagents simplifies the waste treatment process, aligning with green chemistry principles and reducing the environmental footprint of the manufacturing process.
- Cost Reduction in Manufacturing: The synthesis of HTH offers significant economic advantages through process intensification and raw material efficiency. By combining three functional units into a single molecule, the effective loading rate of the photoinitiator in the final formulation can be optimized, potentially reducing the total mass of additive required to achieve the same curing speed. The use of methanesulfonic acid as a catalyst avoids the need for expensive transition metals, thereby eliminating the costly downstream removal steps associated with metal contamination. Additionally, the high thermal stability of HTH reduces energy losses during processing and minimizes product degradation, leading to higher overall yields and less waste. These factors collectively contribute to a more lean and cost-effective manufacturing process without sacrificing performance.
- Enhanced Supply Chain Reliability: The raw materials for HTH, specifically Darocur 1173 and Darocur 2959, are mature products with well-established global supply networks. This ensures that the production of HTH is not bottlenecked by the availability of obscure precursors. The two-step synthesis is robust and tolerant to minor variations in reaction conditions, which enhances batch-to-b consistency and reliability. For supply chain heads, this translates to predictable lead times and the ability to scale production rapidly in response to market demand. The solid nature of the final product also simplifies logistics and storage compared to liquid initiators, reducing shipping costs and handling risks.
- Scalability and Environmental Compliance: The reaction conditions described in the patent, such as the use of dichloromethane and DMF, are standard in the fine chemical industry, allowing for straightforward scale-up in existing facilities. The workup procedures involving aqueous washes and column chromatography can be adapted to continuous extraction and crystallization processes for large-scale production. Moreover, the non-volatile nature of HTH reduces VOC emissions during the curing process, helping manufacturers meet increasingly strict environmental regulations. The improved thermal stability also means less decomposition during storage and transport, further enhancing the sustainability profile of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of trifunctional photoinitiators in industrial settings. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for decision-making. Understanding these nuances is crucial for R&D teams evaluating the feasibility of switching from traditional initiators to advanced macromolecular systems. The answers below clarify the performance benchmarks and operational parameters associated with HTH.
Q: What are the primary advantages of the trifunctional photoinitiator HTH over monofunctional alternatives?
A: HTH offers significantly reduced migration and volatility due to its higher molecular weight (769.82), while maintaining superior photoinitiation activity compared to Darocur 1173 and 2959.
Q: How does the thermal stability of HTH compare to existing commercial photoinitiators?
A: The temperature of 5% weight loss for HTH is improved by more than 50°C compared to Darocur 2959, indicating excellent thermal stability for high-temperature processing applications.
Q: Is the synthesis of HTH scalable for industrial production?
A: Yes, the synthesis utilizes readily available raw materials like Darocur 1173 and 2959 and employs standard esterification conditions, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable HTH Photoinitiator Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced photoinitiators play in the evolution of the coatings and adhesives industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative technologies like the HTH trifunctional photoinitiator can be seamlessly transitioned from the lab to the factory floor. We are committed to delivering high-purity photoinitiators that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in esterification chemistry and purification techniques allows us to optimize the synthesis of complex molecules like HTH, guaranteeing consistent quality and performance for our global partners.
We invite you to collaborate with us to explore how this novel trifunctional architecture can enhance your product portfolio. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific formulation needs. By partnering with us, you gain access to specific COA data and route feasibility assessments that will accelerate your development timeline. Contact us today to discuss how we can support your supply chain with reliable, high-performance photoinitiator solutions that drive innovation and compliance.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
