Scalable Synthesis of 3,5-Dimethyl-1H-Pyrrole-2,4-Dicarbaldehyde for Advanced Pharmaceutical Applications
Scalable Synthesis of 3,5-Dimethyl-1H-Pyrrole-2,4-Dicarbaldehyde for Advanced Pharmaceutical Applications
The development of complex heterocyclic building blocks remains a cornerstone of modern medicinal chemistry, particularly for the construction of kinase inhibitors and other targeted therapeutics. Patent CN102887851A introduces a robust and scientifically sound methodology for the preparation of 3,5-dimethyl-1H-pyrrole-2,4-dicarbaldehyde, a highly functionalized intermediate essential for diverse drug discovery programs. This technical disclosure outlines a novel three-step synthetic pathway that transforms simple, commercially available starting materials into a high-value dialdehyde scaffold with impressive efficiency. By leveraging classical organic transformations such as reductive cyclization and Vilsmeier-Haack formylation, the inventors have established a route that balances chemical elegance with practical manufacturability. For R&D directors and process chemists, this patent represents a significant advancement in accessing pyrrole-based architectures without relying on exotic reagents or prohibitively expensive catalytic systems. The detailed procedural parameters provided ensure that the transition from laboratory bench to pilot plant can be executed with minimal technical risk.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted pyrrole aldehydes has been fraught with challenges related to regioselectivity and harsh reaction conditions. Traditional approaches often necessitate the use of pre-functionalized pyrrole rings that are difficult to source or require multi-step protection and deprotection strategies to achieve the desired substitution pattern. Furthermore, many existing methodologies rely on stoichiometric amounts of toxic heavy metals or unstable organometallic reagents, which complicate waste disposal and increase the environmental footprint of the manufacturing process. Inefficient purification steps in older protocols frequently lead to poor yields and inconsistent batch-to-batch quality, creating bottlenecks for supply chain managers who require reliable volumes of high-purity intermediates. The lack of a streamlined, cost-effective route has often forced procurement teams to accept higher price points for these critical building blocks, thereby inflating the overall cost of goods for downstream API manufacturing.
The Novel Approach
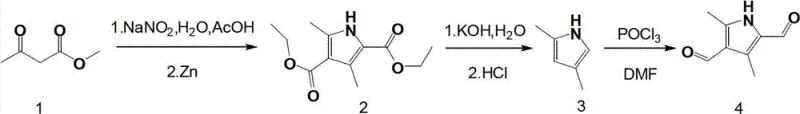
The methodology described in the patent data circumvents these historical hurdles by utilizing methyl acetoacetate as a foundational feedstock, a commodity chemical known for its availability and low cost. The innovation lies in the strategic sequence of nitrosation followed by zinc-mediated reduction to construct the pyrrole core, effectively installing the methyl groups and ester functionalities in a single operational sequence. This is subsequently followed by a hydrolysis-decarboxylation step to reveal the reactive pyrrole nucleus, which is then subjected to a double formylation. This logical progression minimizes the number of isolation steps and maximizes atom economy relative to alternative pathways. The process is designed to be scalable, utilizing standard solvents like acetic acid, ethanol, and DMF, which are familiar to any contract development and manufacturing organization (CDMO). By establishing a clear path from bulk chemicals to a sophisticated dialdehyde, this approach offers a compelling solution for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Vilsmeier-Haack Formylation and Reductive Cyclization
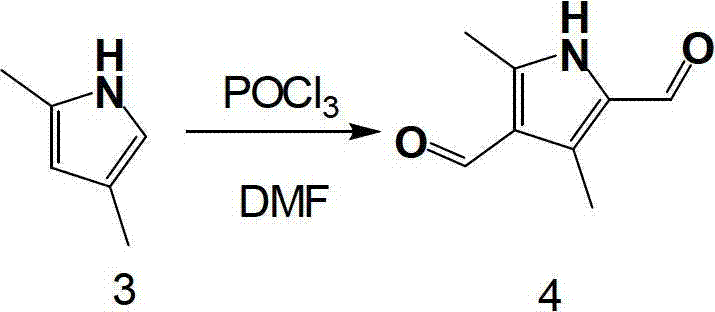
The heart of this synthetic strategy involves two distinct mechanistic phases that warrant close examination for process optimization. The initial formation of the pyrrole ring proceeds through the generation of an oximino intermediate from methyl acetoacetate using sodium nitrite in an acidic medium. This electrophilic nitrosation is followed by a critical reduction step using zinc powder, which facilitates the cyclization and aromatization to form the diethyl 2,4-dimethylpyrrole-3,5-dicarboxylate. The choice of zinc as the reductant is particularly advantageous from a safety and cost perspective, avoiding the pyrophoric nature of alternatives like lithium aluminum hydride. Following the hydrolysis of the ester groups, the resulting 2,4-dimethylpyrrole serves as the substrate for the final transformation. The Vilsmeier-Haack reaction, employing phosphorus oxychloride (POCl3) and dimethylformamide (DMF), generates a highly reactive chloroiminium ion species. This electrophile attacks the electron-rich positions of the pyrrole ring, specifically the 2 and 4 positions activated by the methyl substituents, to install the aldehyde groups with high regiocontrol.
Impurity control is inherently built into the physicochemical properties of the intermediates and the final product. The patent highlights that the final dialdehyde exhibits excellent crystallization behavior, which is a crucial parameter for purification. During the workup of the formylation reaction, the careful regulation of pH to neutrality using saturated potassium carbonate ensures the decomposition of excess Vilsmeier reagent and the protonation of basic impurities, allowing them to be separated into the aqueous phase. The subsequent extraction with ethyl acetate isolates the organic product, which can then be further purified by recrystallization. This reliance on crystallization rather than chromatography is a key indicator of industrial viability, as it allows for the processing of large batches with high throughput. The structural integrity of the final molecule is confirmed through spectroscopic analysis, ensuring that the complex substitution pattern is maintained without degradation of the sensitive aldehyde functionalities.
How to Synthesize 3,5-Dimethyl-1H-Pyrrole-2,4-Dicarbaldehyde Efficiently
Executing this synthesis requires precise control over reaction temperatures and stoichiometry to maximize the reported 38% overall yield. The process begins with the careful addition of sodium nitrite to a cooled solution of methyl acetoacetate in acetic acid, maintaining the temperature between 5°C and 15°C to prevent side reactions. Following the reduction and cyclization, the intermediate diester is subjected to vigorous hydrolysis conditions using potassium hydroxide, followed by acidification to induce decarboxylation. The final formylation step demands anhydrous conditions and slow addition of phosphorus oxychloride to manage the exotherm effectively. For a comprehensive understanding of the specific operational parameters, including exact molar ratios and stirring times, please refer to the standardized protocol outlined below.
- Synthesize diethyl 2,4-dimethylpyrrole-3,5-dicarboxylate by reacting methyl acetoacetate with sodium nitrite in acetic acid followed by zinc powder reduction.
- Perform hydrolysis and decarboxylation of the diester intermediate using potassium hydroxide and hydrochloric acid to obtain 2,4-dimethylpyrrole.
- Execute Vilsmeier-Haack formylation on 2,4-dimethylpyrrole using phosphorus oxychloride and DMF to yield the final dialdehyde product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers substantial benefits for organizations looking to optimize their supply chains for pyrrole-based intermediates. The reliance on methyl acetoacetate as the primary starting material decouples production from volatile markets for specialized heterocycles, ensuring a stable and predictable supply of raw materials. Since the reagents used—such as zinc powder, potassium hydroxide, and phosphorus oxychloride—are standard industrial chemicals available from multiple global vendors, the risk of supply disruption is significantly minimized. This diversification of the supply base allows procurement managers to negotiate better terms and secure long-term contracts with confidence. Furthermore, the elimination of precious metal catalysts removes the need for expensive scavenging steps to meet residual metal specifications, which is a common regulatory requirement in pharmaceutical manufacturing.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of low-cost commodity feedstocks and the avoidance of proprietary catalysts. By utilizing zinc for reduction instead of noble metals, the direct material costs are drastically lowered, and the associated waste treatment costs are simplified. The high purity achieved through crystallization reduces the need for resource-intensive chromatographic purification, leading to significant savings in solvent consumption and labor hours. These factors combine to create a highly competitive cost structure for the final dialdehyde, enabling downstream partners to reduce their overall cost of goods sold for complex API synthesis.
- Enhanced Supply Chain Reliability: The robustness of the chemical transformations ensures consistent batch quality, which is critical for maintaining continuous production schedules. The intermediates generated in the early steps are stable and can be stockpiled if necessary, providing a buffer against short-term demand fluctuations. Additionally, the simplicity of the workup procedures, involving standard liquid-liquid extractions and filtrations, means that the process can be easily transferred between different manufacturing sites without requiring specialized equipment. This flexibility enhances the resilience of the supply chain, allowing for rapid scaling in response to market needs.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing reaction conditions that are safe and manageable in large reactors. The waste streams generated are primarily aqueous salts and organic solvents that can be treated using conventional effluent treatment protocols, ensuring compliance with stringent environmental regulations. The high atom efficiency of the formylation step minimizes the generation of byproduct waste, aligning with green chemistry principles. This environmental compatibility not only reduces disposal costs but also supports the sustainability goals of modern pharmaceutical companies, making it an attractive option for eco-conscious sourcing strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 3,5-dimethyl-1H-pyrrole-2,4-dicarbaldehyde. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a transparent view of the technology's capabilities. Understanding these details helps stakeholders make informed decisions about integrating this intermediate into their development pipelines.
Q: What is the primary advantage of this synthesis route for 3,5-dimethyl-1H-pyrrole-2,4-dicarbaldehyde?
A: The primary advantage is the use of inexpensive, commodity raw materials like methyl acetoacetate and the avoidance of precious metal catalysts, which significantly reduces production costs while maintaining high purity through effective crystallization.
Q: How is the purity of the final dialdehyde controlled in this process?
A: Purity is controlled through a rigorous workup procedure involving pH regulation and multiple extractions, followed by recrystallization using a petroleum ether and ethyl acetate solvent system, which effectively removes organic impurities.
Q: Is this synthetic route suitable for large-scale industrial manufacturing?
A: Yes, the process utilizes standard chemical reagents and conventional unit operations such as reflux, filtration, and distillation, making it highly adaptable for commercial scale-up from kilogram to multi-ton production levels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,5-Dimethyl-1H-Pyrrole-2,4-Dicarbaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in accelerating drug discovery and development. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale synthesis to industrial manufacturing is seamless. We are committed to delivering 3,5-dimethyl-1H-pyrrole-2,4-dicarbaldehyde with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical instrumentation. Whether you require custom synthesis or bulk supply, our infrastructure is designed to meet the demanding requirements of the global pharmaceutical industry.
We invite you to collaborate with us to optimize your supply chain for this valuable building block. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing expertise can support your project timelines and quality objectives.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
