Revolutionizing Blue OLED Efficiency with Non-Conjugated Aryl Boron Polymers
The landscape of organic light-emitting diodes (OLEDs) is undergoing a significant transformation driven by the urgent demand for higher efficiency and lower production costs in next-generation display technologies. Patent CN111378438A introduces a groundbreaking class of fluorescent polymer compounds that utilize aryl boron acceptors and space charge transfer luminescence to overcome the intrinsic limitations of conventional materials. Unlike traditional fluorescent materials that are restricted by spin quantum statistics to a maximum internal quantum efficiency of 25%, this novel approach leverages thermally activated delayed fluorescence (TADF) mechanisms to harvest both singlet and triplet excitons. The core innovation lies in the unique molecular architecture where electron donors and acceptors are spatially separated rather than connected through conjugated bonds, facilitating a reduction in HOMO and LUMO overlap. This structural design achieves a remarkably small singlet-triplet energy level difference, denoted as ΔEST, which is critical for activating the reverse intersystem crossing process required for high-efficiency light emission. Furthermore, the utilization of aryl boron units with weak electron-withdrawing properties allows for precise tuning of charge transfer intensity, making it exceptionally suitable for realizing stable blue light emission, a longstanding challenge in the industry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of polymer-based OLED materials has been dominated by conjugated main chain structures that rely on through-bond charge transfer (TBCT) mechanisms for light emission. While these materials have shown promise, they face severe inherent drawbacks that hinder their commercial viability for high-performance blue displays. The primary issue stems from the extensive delocalization of electron clouds along the conjugated backbone, which invariably leads to a red-shift in the emission spectrum, making it extremely difficult to achieve the deep blue color coordinates required by modern display standards. Additionally, controlling the electronic distribution between donor and acceptor moieties within a rigid conjugated system is notoriously challenging, often resulting in a trade-off where achieving a small ΔEST compromises the fluorescence quantum efficiency (PLQY). This imbalance prevents the simultaneous optimization of exciton utilization and radiative decay rates, ultimately capping the device efficiency. Moreover, the synthesis of complex conjugated polymers often involves harsh conditions and expensive transition metal catalysts, which complicates the supply chain and increases the overall cost of manufacturing for large-area panels.
The Novel Approach
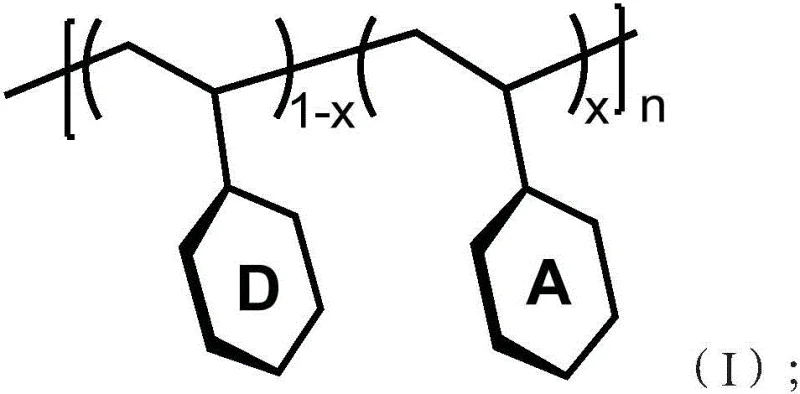
In stark contrast to the conventional paradigms, the technology disclosed in patent CN111378438A employs a non-conjugated main chain structure that facilitates space charge transfer (TSCT) between adjacent electron donors and acceptors. This architectural shift fundamentally decouples the electronic interaction from the backbone conjugation, allowing for independent optimization of the energy levels and spatial orientation of the functional units. By physically separating the donor and acceptor groups through saturated carbon linkers, the material minimizes the orbital overlap necessary to reduce the ΔEST to values below 0.1 eV, thereby unlocking nearly 100% theoretical internal quantum efficiency through triplet harvesting. Crucially, this spatial separation also suppresses the red-shifting effect typical of conjugated systems, enabling the material to maintain a blue emission peak in the desirable 444 nm to 480 nm range. The use of aryl boron as a weak electron acceptor further fine-tunes the charge transfer strength, ensuring that the material retains high fluorescence quantum efficiency while emitting in the blue region, effectively solving the dual challenges of efficiency and color purity that have plagued previous generations of TADF polymers.
Mechanistic Insights into Space Charge Transfer Luminescence
The operational mechanism of this advanced material relies on the sophisticated interplay between spatially separated molecular orbitals rather than traditional covalent bond-mediated electron transport. In this system, the electron donor units, which can be selected from a diverse library of nitrogen-containing heterocycles such as acridine or carbazole derivatives, are pendant to a flexible non-conjugated polymer backbone. The electron acceptor units, characterized by the presence of aryl boron structures, are similarly attached but remain electronically isolated from the donors through the saturated backbone. When an electrical voltage is applied across the device, electrons and holes are injected from the respective electrodes and recombine within the emissive layer to form excitons. Due to the spatial proximity yet electronic isolation of the donor and acceptor, the excited state exhibits a charge transfer character where the highest occupied molecular orbital (HOMO) is localized on the donor and the lowest unoccupied molecular orbital (LUMO) is localized on the acceptor. This spatial separation drastically reduces the exchange energy between the singlet and triplet states, resulting in a minimal ΔEST that allows thermal energy at room temperature to promote triplet excitons back to the singlet state via reverse intersystem crossing (RISC). Consequently, both singlet and triplet excitons contribute to light emission, breaking the 25% efficiency barrier of conventional fluorescence without requiring expensive heavy metal complexes.
Beyond the fundamental photophysics, the impurity profile and stability of the material are significantly enhanced by the robust nature of the non-conjugated backbone and the specific choice of aryl boron acceptors. Traditional conjugated polymers are often susceptible to oxidative degradation and structural defects that act as quenching sites for excitons, reducing the overall device lifetime and efficiency. In this novel architecture, the saturated carbon chains provide greater chemical stability and flexibility, reducing the likelihood of aggregation-caused quenching which is common in rigid planar systems. The aryl boron acceptor units are specifically engineered to have weak electron-withdrawing capabilities, which prevents excessive charge transfer that could lead to non-radiative decay pathways. This careful balancing act ensures that the radiative decay rate remains high, leading to fluorescence quantum efficiencies (PLQY) that can reach up to 78% in film state. Furthermore, the modular nature of the synthesis allows for precise control over the ratio of donor to acceptor units, enabling manufacturers to fine-tune the emission color and efficiency profiles to meet specific application requirements without altering the core synthetic route, thus ensuring consistent quality and performance across different production batches.
How to Synthesize Aryl Boron TADF Polymer Efficiently
The preparation of these high-performance fluorescent polymer compounds is achieved through a straightforward yet highly controlled radical copolymerization process that is amenable to industrial scale-up. The synthesis begins with the preparation of two key vinyl-functionalized monomers: one carrying the electron donor moiety and the other carrying the aryl boron-based electron acceptor moiety. These monomers are then dissolved in an appropriate organic solvent such as tetrahydrofuran or toluene, along with a radical initiator like azobisisobutyronitrile (AIBN). The reaction mixture is subjected to heating under a strictly inert atmosphere, typically nitrogen or argon, to prevent oxygen inhibition which could terminate the growing polymer chains prematurely. The polymerization proceeds for a duration ranging from 8 to 72 hours at temperatures between 40°C and 120°C, allowing for the formation of long polymer chains with the desired donor-acceptor distribution. Following the reaction, the polymer is isolated by precipitation into a non-solvent such as methanol or acetone, followed by thorough purification steps including re-dissolution and re-precipitation to remove any unreacted monomers or oligomers. This solution-based synthesis route is particularly advantageous as it avoids the need for complex transition metal catalyzed coupling reactions often required for conjugated polymers, thereby simplifying the purification process and reducing the risk of metal contamination in the final electronic material.
- Mix vinyl-functionalized electron donor monomers and aryl boron acceptor monomers with a radical initiator like AIBN in an organic solvent such as THF.
- Conduct the reaction under a protective inert gas atmosphere at temperatures between 40°C and 120°C for 8 to 72 hours to ensure complete polymerization.
- Precipitate the resulting polymer solution into a non-solvent like methanol or acetone, followed by filtration and vacuum drying to obtain the pure solid product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement and supply chain perspective, the adoption of this non-conjugated TADF polymer technology offers substantial advantages over traditional phosphorescent and conjugated fluorescent materials. The most significant benefit arises from the elimination of scarce and expensive heavy metal catalysts such as iridium or platinum, which are traditionally required to harvest triplet excitons in phosphorescent OLEDs. By relying on purely organic components composed of abundant elements like carbon, hydrogen, nitrogen, and boron, the raw material costs are drastically reduced, and the supply chain becomes far less vulnerable to geopolitical fluctuations affecting precious metal markets. Additionally, the solution-processable nature of these polymers opens the door to low-cost manufacturing techniques such as inkjet printing and roll-to-roll coating, which stand in stark contrast to the high-vacuum thermal evaporation processes currently dominant in the industry. This shift not only lowers capital expenditure for manufacturing equipment but also significantly improves material utilization rates, as solution processing minimizes the waste associated with line-of-sight deposition methods. Consequently, manufacturers can achieve significant cost reductions in display panel manufacturing while maintaining high performance standards.
- Cost Reduction in Manufacturing: The economic impact of switching to this aryl boron-based polymer system is profound, primarily driven by the simplification of the synthetic route and the avoidance of precious metals. Traditional phosphorescent emitters require multi-step syntheses involving costly palladium or iridium catalysts, which not only increase the bill of materials but also necessitate expensive purification steps to remove trace metal residues that could degrade device performance. In contrast, the radical polymerization method described in the patent utilizes common organic initiators and solvents, streamlining the production workflow and reducing the overall complexity of the supply chain. Furthermore, the high fluorescence quantum efficiency achieved without heavy metals means that less material is required to achieve the same brightness levels, directly lowering the consumption cost per unit area of the display. This combination of cheaper raw materials, simpler synthesis, and higher material efficiency translates into substantial cost savings for mass production facilities aiming to compete in the price-sensitive consumer electronics market.
- Enhanced Supply Chain Reliability: Ensuring a stable and continuous supply of critical materials is a top priority for supply chain heads, and this technology offers a more resilient alternative to metal-dependent emitters. The reliance on organic synthesis using widely available chemical feedstocks reduces the risk of supply disruptions caused by mining constraints or export restrictions on rare earth elements and precious metals. The synthetic pathway is robust and scalable, utilizing standard chemical engineering unit operations that are well-understood and easily replicated across different manufacturing sites globally. This geographical flexibility allows companies to diversify their supplier base and mitigate risks associated with single-source dependencies. Moreover, the stability of the non-conjugated polymer backbone suggests a longer shelf life and easier handling requirements compared to sensitive organometallic complexes, reducing losses during storage and transportation. These factors collectively contribute to a more predictable and reliable supply chain, ensuring that production schedules for downstream display manufacturers can be met consistently without unexpected delays.
- Scalability and Environmental Compliance: As the industry moves towards more sustainable manufacturing practices, the environmental profile of this polymer synthesis offers distinct advantages over conventional methods. The solution-based processing eliminates the need for high-energy vacuum chambers and reduces the carbon footprint associated with the physical vapor deposition processes used for small molecules. The use of common organic solvents allows for established recycling and recovery protocols, minimizing hazardous waste generation. Additionally, the absence of toxic heavy metals in the final product simplifies end-of-life disposal and recycling of electronic devices, aligning with increasingly stringent global environmental regulations such as RoHS and REACH. The scalability of the radical polymerization process is well-proven in the broader polymer industry, meaning that scaling from laboratory grams to commercial tons does not require fundamental changes to the chemistry or equipment. This ease of scale-up ensures that supply can rapidly expand to meet growing market demand for OLED TVs, smartphones, and lighting applications without encountering the bottlenecks often seen with novel nanomaterials or complex organometallics.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of integrating this material into their product roadmaps, we have compiled answers to common inquiries regarding the performance and processing characteristics of the aryl boron TADF polymer. These questions address the critical technical hurdles often encountered when transitioning from small molecule evaporated materials to solution-processed polymers, focusing on efficiency, color stability, and compatibility with existing device architectures. Understanding these nuances is essential for R&D teams planning pilot runs and for procurement officers assessing the long-term viability of the supply source. The answers provided are derived directly from the experimental data and technical disclosures within the patent documentation, ensuring accuracy and relevance to real-world application scenarios.
Q: Why do traditional conjugated TADF polymers struggle with blue emission?
A: Traditional conjugated polymers often suffer from significant electron cloud delocalization along the main chain, which inevitably causes a red-shift in the emission spectrum, making it difficult to achieve pure blue light required for high-quality displays.
Q: How does the space charge transfer mechanism improve efficiency?
A: By spatially separating the electron donor and acceptor units without conjugated links, the overlap between HOMO and LUMO orbitals is minimized, resulting in a very small singlet-triplet energy gap that facilitates efficient reverse intersystem crossing.
Q: Is this material suitable for low-cost manufacturing processes?
A: Yes, the polymer is designed for solution processing techniques such as spin coating and inkjet printing, which significantly reduces equipment costs compared to traditional vacuum thermal evaporation methods used for small molecule OLEDs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Boron TADF Polymer Supplier
The transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and proven manufacturing capabilities. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, offering comprehensive support for the scale-up and production of advanced OLED materials like the aryl boron TADF polymers described in patent CN111378438A. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from gram-scale synthesis to industrial manufacturing is seamless and efficient. We understand the critical importance of material purity in electronic applications and operate stringent purity specifications backed by rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom modification of the donor-acceptor ratio to tune emission colors or need assistance with optimizing the formulation for inkjet printing, our CDMO experts are ready to collaborate on developing tailored solutions that meet your specific performance targets.
We invite you to engage with our technical procurement team to discuss how this breakthrough technology can enhance your product portfolio and reduce your overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic benefits of switching to our solution-processable TADF polymers compared to your current material stack. We encourage you to contact us to obtain specific COA data for our pilot batches and to schedule a consultation for route feasibility assessments tailored to your production constraints. Let us help you navigate the complexities of next-generation display material sourcing and secure a competitive advantage in the rapidly evolving optoelectronics market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
