Advanced Manufacturing of 3,4-Dihydroxybenzonitrile via Efficient Vanillin Demethylation for Global Supply Chains
Introduction to High-Efficiency Nitrile Intermediate Synthesis
The global demand for high-purity heterocyclic intermediates continues to surge, driven by the expanding oncology and agrochemical sectors. A pivotal advancement in this domain is documented in patent CN107118128B, which outlines a robust preparation method for 3,4-dihydroxybenzonitrile. This specific compound serves as a critical building block for synthesizing quinazoline derivatives, which are essential scaffolds in modern anticancer drug development. Unlike traditional methodologies that rely on scarce or environmentally hazardous precursors, this innovation leverages vanillin, a ubiquitous and economically accessible starting material. The technical breakthrough lies in the strategic application of Lewis acid catalysis to achieve demethylation with exceptional efficiency. By optimizing reaction conditions such as temperature gradients and solvent systems, the process ensures that the final product achieves a purity threshold exceeding 99.0 percent. For R&D directors and procurement strategists, this represents a significant opportunity to streamline supply chains while adhering to stringent quality specifications required for pharmaceutical grade intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,4-dihydroxybenzonitrile has been plagued by significant logistical and economic hurdles. Early literature describes routes originating from 3,4-dihydroxybenzaldehyde, a raw material that is not only expensive but also involves a convoluted preparation process that generates substantial environmental pollution. Furthermore, alternative pathways utilizing m-aminobenzonitrile or 3,4-dimethoxybenzonitrile often require harsh reagents such as lithium diisopropamide or dimethyl imidazole diketone. These reagents are not only costly but also pose severe safety risks during handling and scale-up. Additionally, previous methods reported in patents like US2004/0024239 involved the use of highly toxic substances as catalysts, creating complex waste streams that complicate regulatory compliance. The reliance on such hazardous chemistry limits the feasibility of these processes for large-scale industrial adoption, forcing manufacturers to contend with low yields and inconsistent batch quality.
The Novel Approach
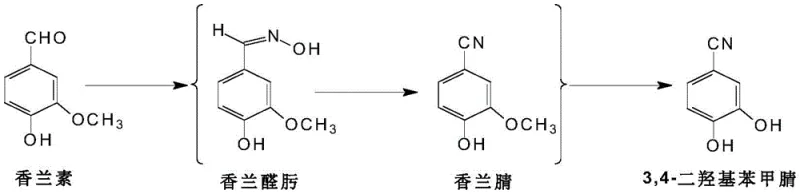
The methodology disclosed in the patent introduces a paradigm shift by utilizing vanillin as the primary feedstock, effectively bypassing the supply chain bottlenecks associated with dihydroxybenzaldehyde. As illustrated in the reaction scheme below, the process converts vanillin into vanillyl nitrile through a controlled oximation and dehydration sequence. This intermediate is then subjected to a Lewis acid-catalyzed demethylation, a step that is far more atom-economical and safer than previous demethylation strategies. The novelty extends to the flexibility of the process, which can be executed either as a stepwise synthesis with intermediate isolation or as a streamlined one-pot reaction. This adaptability allows manufacturers to choose the operational mode that best fits their existing infrastructure. By eliminating the need for exotic reagents and focusing on commodity chemicals like hydroxylamine hydrochloride and anhydrous aluminum trichloride, the novel approach drastically simplifies the manufacturing landscape while maintaining superior product integrity.
Mechanistic Insights into Lewis Acid-Catalyzed Demethylation
The core of this synthetic strategy relies on the precise manipulation of functional groups through a two-stage mechanistic pathway. Initially, the aldehyde group of vanillin undergoes condensation with hydroxylamine hydrochloride in a polar aprotic solvent, typically N,N-dimethylformamide (DMF). This reaction forms a vanillin aldoxime intermediate, which subsequently undergoes thermal dehydration at elevated temperatures ranging from 100 to 150 degrees Celsius to yield vanillyl nitrile. The second critical phase involves the cleavage of the aryl methyl ether bond. In this step, a Lewis acid, preferably anhydrous aluminum trichloride, coordinates with the oxygen atom of the methoxy group. This coordination weakens the carbon-oxygen bond, facilitating nucleophilic attack and subsequent removal of the methyl group as a chloride species. The reaction is carefully managed at temperatures between 120 and 140 degrees Celsius to ensure complete conversion without degrading the sensitive nitrile functionality. This mechanistic precision is vital for preventing the formation of mono-demethylated byproducts, ensuring that the final output is predominantly the desired 3,4-dihydroxy isomer.
Impurity control is intrinsically built into the reaction design through the selection of solvents and workup procedures. The use of polar aprotic solvents ensures that all reactants remain in solution during the critical heating phases, promoting homogeneous reaction kinetics. Following the demethylation, the reaction mixture is quenched with dilute hydrochloric acid, which hydrolyzes any remaining aluminum complexes and precipitates the product. The patent emphasizes the importance of recrystallization using an alcohol-water mixed solvent system. This purification step is highly effective at removing trace organic impurities and residual metal salts that might persist from the Lewis acid catalyst. By rigorously controlling the crystallization parameters, manufacturers can consistently achieve purity levels of 99.0 percent or higher. This high degree of purity is essential for downstream applications in medicinal chemistry, where even trace impurities can affect the efficacy and safety of the final active pharmaceutical ingredient.
How to Synthesize 3,4-Dihydroxybenzonitrile Efficiently
The synthesis protocol described in the patent offers a clear roadmap for producing this valuable intermediate with high reproducibility. Whether opting for the stepwise isolation of vanillyl nitrile or the direct one-pot conversion, the key lies in strict temperature control and stoichiometric precision. The process begins with the reaction of vanillin and hydroxylamine hydrochloride, followed by the introduction of the Lewis acid catalyst for demethylation. Detailed operational parameters, including specific molar ratios and heating ramps, are critical for maximizing yield and minimizing side reactions. For technical teams looking to implement this route, adherence to the standardized steps outlined in the intellectual property is essential for success.
- React industrial-grade vanillin with hydroxylamine hydrochloride in a polar aprotic solvent like DMF at controlled temperatures (50-120°C) to form vanillyl nitrile.
- Without isolation in the one-pot method, or after isolation in the stepwise method, introduce a Lewis acid catalyst such as anhydrous aluminum trichloride.
- Heat the mixture to facilitate demethylation, followed by hydrolysis, extraction, and recrystallization to achieve purity exceeding 99.0%.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthesis route offers compelling advantages that directly address the pain points of modern chemical procurement. The shift from specialized, high-cost starting materials to commodity-grade vanillin fundamentally alters the cost structure of the supply chain. Vanillin is produced on a massive global scale for the flavor and fragrance industry, ensuring a stable and continuous supply that is less susceptible to the volatility seen with niche pharmaceutical intermediates. This abundance translates into significant cost reduction in pharmaceutical intermediate manufacturing, as the raw material expenditure constitutes a smaller fraction of the total production cost. Furthermore, the elimination of highly toxic catalysts reduces the burden on waste management systems, lowering the overall environmental compliance costs associated with production.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of inexpensive and readily available reagents. By replacing costly demethylating agents with common Lewis acids like aluminum trichloride, the direct material costs are substantially lowered. Additionally, the ability to perform the reaction in a one-pot fashion eliminates the need for intermediate isolation, filtration, and drying steps. This consolidation of unit operations reduces energy consumption, labor hours, and solvent usage, leading to a leaner manufacturing process. The cumulative effect of these efficiencies is a drastic simplification of the production workflow, which inherently drives down the cost per kilogram of the final product without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply chain resilience is a top priority for multinational corporations, and this method enhances reliability by diversifying the source of critical inputs. Since vanillin is a bulk chemical with multiple global suppliers, the risk of supply disruption due to single-source dependency is minimized. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, further stabilizing production schedules. For supply chain heads, this translates to reduced lead time for high-purity pharmaceutical intermediates, as the manufacturing cycle is shorter and more predictable. The scalability of the process ensures that sudden increases in demand can be met without the need for extensive process re-engineering or capital investment.
- Scalability and Environmental Compliance: The transition from laboratory scale to commercial production is often fraught with challenges, but this patent demonstrates successful pilot test verification. The use of standard industrial solvents like DMF and common reactor materials compatible with Lewis acids facilitates easy scale-up from 100 kgs to 100 MT annual commercial production. Moreover, the process avoids the generation of persistent organic pollutants associated with older synthetic routes. The waste streams generated are easier to treat, aligning with increasingly stringent global environmental regulations. This environmental compatibility not only mitigates regulatory risk but also enhances the corporate sustainability profile of the manufacturer, a factor that is becoming increasingly important in vendor selection criteria for major pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 3,4-dihydroxybenzonitrile. These insights are derived directly from the experimental data and claims presented in the patent documentation. Understanding these nuances is crucial for technical procurement teams evaluating the feasibility of integrating this intermediate into their supply networks. The answers provided reflect the specific advantages of the vanillin-based route over legacy technologies.
Q: What are the primary advantages of using vanillin over 3,4-dihydroxybenzaldehyde for this synthesis?
A: Vanillin is significantly more abundant and cost-effective than 3,4-dihydroxybenzaldehyde. The patent highlights that traditional routes using 3,4-dihydroxybenzaldehyde involve complex preparation processes and serious environmental pollution, whereas the vanillin route utilizes a widely available starting material with a simpler, more industrially viable workflow.
Q: How does the Lewis acid demethylation step impact product purity?
A: The use of Lewis acids like anhydrous aluminum trichloride allows for efficient removal of the methyl group under controlled conditions. When combined with standard recrystallization techniques described in the patent, this method consistently yields 3,4-dihydroxybenzonitrile with purity levels reaching up to 99.5%, effectively minimizing impurities common in high-temperature dehydration methods.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the patent explicitly states that both the stepwise and one-pot preparation modes have been verified through pilot tests to be suitable for industrial production. The process avoids highly toxic substances found in earlier methods and simplifies the operational steps, making it robust for scaling from kilogram to multi-ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4-Dihydroxybenzonitrile Supplier
The technological potential of the vanillin-to-nitrile conversion route is immense, offering a sustainable and cost-effective solution for the global market. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of critical intermediates. Our facility is equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch. We understand that in the pharmaceutical and agrochemical industries, the reliability of the supply chain is just as important as the chemical quality itself. Our commitment to process excellence allows us to deliver high-purity 3,4-dihydroxybenzonitrile that meets the exacting standards required for the synthesis of next-generation therapeutics.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific projects. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages of switching to this vanillin-based supply chain. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your volume requirements. Let us collaborate to enhance your supply chain efficiency and drive innovation in your product development pipeline.
