Revolutionizing UV Absorber Production: Advanced Catalytic Strategies for Commercial Scale Manufacturing
The global demand for effective ultraviolet protection has surged dramatically in recent years, driven by increasing awareness of skin health and the deleterious effects of UV-A radiation on human DNA. At the heart of this market lies the critical class of 1,3-diphenylpropanedione compounds, most notably Avobenzone (Parsol 1789), which stands as one of the few FDA-approved organic filters capable of absorbing the entire UV-A spectrum. However, the industrial manufacturing of these high-value cosmetic intermediates has historically been plagued by low yields, hazardous reagents, and complex purification protocols that strain supply chains. A groundbreaking technical solution detailed in patent CN109305897B offers a transformative approach to this challenge, introducing a high-yield production process that leverages a sophisticated mixed-alkali catalytic system combined with continuous reactive distillation. This innovation not only addresses the long-standing purity issues associated with traditional synthesis but also establishes a new benchmark for cost-effective and environmentally compliant manufacturing of fine chemical intermediates.
For procurement leaders and R&D directors seeking a reliable UV absorber intermediate supplier, understanding the mechanistic superiority of this patented route is essential for securing long-term supply continuity. The technology replaces dangerous and expensive reagents with a optimized blend of sodium and potassium methoxides, achieving yields exceeding 85.0% while significantly reducing the generation of hazardous three-waste streams. By shifting the reaction equilibrium through the continuous removal of byproducts, the process ensures a lighter product color and characteristic aromatic odor, qualities that are paramount for downstream formulation in premium sunscreen and skincare applications. This report delves deep into the technical architecture of this process, analyzing how it overcomes the limitations of legacy methods to deliver substantial value across the entire chemical value chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 1,3-diphenylpropanedione scaffolds relied heavily on aggressive strong bases such as sodium amide or sodium hydride, which introduced severe operational complexities including rigorous anhydrous requirements and significant safety hazards associated with violent hydrolysis. The use of sodium amide often leads to undesirable ammonolysis side reactions that degrade product quality and lower overall yield, necessitating extensive and costly purification steps to remove nitrogenous impurities. Furthermore, sodium hydride reacts explosively with trace moisture, imposing exorbitant demands on reactor drying and inert atmosphere maintenance, which drastically inflates capital expenditure and operational risk profiles for manufacturers. Alternative routes involving halogenation and dehalogenation steps, while offering moderate yields, introduce toxic halogenated solvents and corrosive reagents that create massive environmental liabilities and complicate waste disposal compliance. These legacy methodologies collectively result in a fragile supply chain characterized by batch-to-batch variability, high production costs, and significant safety vulnerabilities that are increasingly unacceptable in modern green chemistry standards.
The Novel Approach
In stark contrast to these hazardous legacy protocols, the novel approach disclosed in the patent utilizes a carefully engineered mixed-alkali catalyst system comprising sodium methoxide and a catalytic amount of potassium methoxide to drive the Claisen condensation with exceptional efficiency. This method operates under milder conditions in hydrocarbon solvents like toluene, eliminating the need for cryogenic temperatures or strictly anhydrous environments required by metal hydrides. The integration of a high-efficiency rectification column equipped with theta-ring stainless steel packing allows for the continuous removal of methanol byproduct, effectively shifting the chemical equilibrium towards the desired diketone product according to Le Chatelier's principle. As illustrated in the specific synthesis of Avobenzone below, this dynamic removal of byproducts prevents the reverse reaction and minimizes thermal degradation, resulting in a robust process that consistently delivers yields greater than 85%.
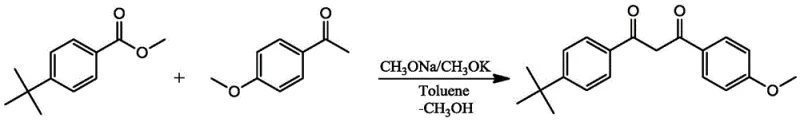
The visual representation of the Avobenzone synthesis highlights the elegance of this transformation, where p-tert-butyl methyl benzoate and p-methoxyacetophenone converge under the influence of the dual-catalyst system to form the target UV filter with minimal side products. This streamlined workflow simplifies post-treatment to basic acidification, washing, and recrystallization, thereby reducing solvent consumption and energy usage compared to multi-step halogenation routes. For cost reduction in cosmetic intermediate manufacturing, this elimination of toxic reagents and complex workup procedures translates directly into improved margins and a smaller environmental footprint, making it an ideal candidate for sustainable industrial scale-up.
Mechanistic Insights into Mixed-Alkali Catalyzed Claisen Condensation
The core innovation of this process lies in the synergistic interaction between sodium and potassium cations within the reaction medium, which modulates the solubility and reactivity of the enolate intermediates formed during the condensation. Sodium methoxide serves as the primary base to induce the initial deprotonation of the acetophenone derivative, generating the nucleophilic enolate species that attacks the carbonyl carbon of the benzoate ester. However, the exclusive use of sodium bases often leads to the precipitation of the intermediate sodium enolate salt, which can create a heterogeneous solid-liquid system that impedes mass transfer and slows reaction kinetics in the later stages. The strategic addition of potassium methoxide, which possesses higher alkalinity and forms more soluble potassium salts, acts as a phase-transfer promoter that keeps the reactive intermediates in solution, ensuring sustained catalytic activity throughout the entire reaction duration. This delicate balance prevents the accumulation of inactive solid precipitates and maintains a homogeneous reaction environment that facilitates rapid conversion of starting materials.
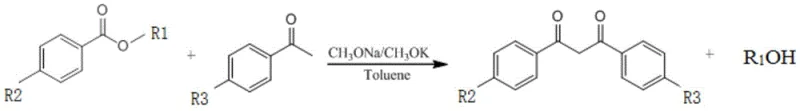
Furthermore, the control of impurity profiles is meticulously managed through the precise regulation of dropping rates and temperature gradients within the rectification column. By maintaining the reaction temperature between 105°C and 108°C, the process avoids the thermal thresholds where self-condensation of the acetophenone starting material becomes kinetically favorable, a common source of difficult-to-remove oligomeric impurities. The continuous distillation of methanol not only drives the forward reaction but also removes low-boiling volatile impurities that could otherwise contaminate the final crystalline product. This results in a crude product of such high purity that subsequent recrystallization yields white needle-like crystals with a GC purity exceeding 98%, meeting the stringent specifications required for high-purity UV absorber applications in sensitive dermal formulations. The mechanistic clarity provided by this patent offers R&D teams a reliable blueprint for optimizing similar condensation reactions across a broad range of fine chemical intermediates.
How to Synthesize Avobenzone Efficiently
The implementation of this high-yield synthesis route requires precise adherence to the sequential addition of catalysts and the maintenance of specific thermal conditions to maximize the benefits of the mixed-alkali system. Operators must first establish the reaction environment by charging the benzoate ester and solid sodium methoxide into the reactor, allowing the initial induction period to proceed before introducing the acetophenone component. The critical step involves the dropwise addition of the acetophenone over a controlled period of 1 to 1.5 hours, synchronized with the continuous fractionation of methanol to prevent local concentration spikes that could trigger exotherms or side reactions. Detailed standardized operating procedures regarding catalyst ratios, solvent volumes, and distillation rates are essential for replicating the >85% yield reported in the patent examples.
- Charge the reactor with benzoate compound A, solid sodium methoxide catalyst, and toluene solvent, then heat to initiate methanol removal.
- Dropwise add acetophenone compound B while maintaining temperature at 105-108°C and continuously distilling off methanol byproduct via a rectification column.
- Introduce liquid potassium methoxide co-catalyst during the reaction, followed by acidification, washing, and recrystallization to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this mixed-alkali catalytic process offers profound advantages that extend far beyond simple yield improvements, fundamentally altering the cost structure and reliability of the supply chain for UV absorber intermediates. The replacement of expensive and hazardous reagents like sodium hydride with commodity alkoxides significantly lowers the raw material cost basis, while the simplified workup procedure reduces the consumption of acids, water, and crystallization solvents. This reduction in material intensity directly contributes to cost reduction in fine chemical manufacturing, allowing suppliers to offer more competitive pricing without sacrificing margin, a critical factor in the highly price-sensitive cosmetics and personal care markets. Moreover, the inherent safety of the process reduces insurance premiums and regulatory compliance costs associated with handling pyrophoric materials, further enhancing the economic viability of large-scale production campaigns.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and toxic halogenated reagents removes the need for expensive heavy metal scavenging steps and specialized waste treatment facilities, leading to substantial operational savings. By utilizing a mixed alkali system that operates efficiently at atmospheric pressure with standard glass-lined or stainless steel reactors, the process minimizes capital depreciation and maintenance costs associated with high-pressure or exotic alloy equipment. The high atom economy of the direct condensation route ensures that a greater proportion of raw material mass is converted into saleable product, drastically reducing the cost per kilogram of the final active ingredient. Additionally, the ability to recycle the toluene solvent through simple distillation further compounds these savings, creating a closed-loop system that maximizes resource efficiency.
- Enhanced Supply Chain Reliability: The reliance on widely available commodity chemicals such as sodium methoxide, potassium methoxide, and toluene ensures that production is not vulnerable to the supply bottlenecks often seen with specialized reagents like sodium amide. The robustness of the reaction conditions, which tolerate minor variations in moisture content better than hydride-based routes, reduces the frequency of batch failures and off-spec material, guaranteeing consistent delivery schedules for downstream formulators. This stability is crucial for reducing lead time for high-purity UV absorber intermediates, as it allows manufacturers to maintain lean inventory levels without risking stockouts due to unpredictable synthesis outcomes. The scalability of the process from pilot plant to multi-tonne commercial reactors ensures that supply can be rapidly ramped up to meet seasonal demand surges in the sunscreen industry.
- Scalability and Environmental Compliance: The process generates significantly less hazardous waste compared to traditional methods, as the primary byproduct is methanol, which can be easily recovered and reused or disposed of with minimal environmental impact. The absence of heavy metals and halogenated organics simplifies the regulatory approval process for new manufacturing sites and reduces the burden of environmental reporting and monitoring. This alignment with green chemistry principles enhances the brand reputation of suppliers and meets the increasingly strict sustainability criteria imposed by major multinational cosmetic brands. The commercial scale-up of complex cosmetic intermediates is facilitated by the use of standard unit operations like rectification and crystallization, which are well-understood by plant operators and easy to optimize for continuous processing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this advanced synthesis technology, providing clarity for stakeholders evaluating its adoption. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, ensuring accuracy and relevance for industrial application. Understanding these nuances is vital for making informed decisions about process validation and supplier qualification.
Q: How does the mixed alkali catalyst improve yield compared to traditional sodium methoxide methods?
A: The synergistic use of sodium methoxide and a small amount of potassium methoxide optimizes alkalinity and solubility dynamics. This combination prevents the formation of insoluble salt precipitates that often hinder reaction kinetics in single-catalyst systems, driving yields above 85% compared to traditional methods.
Q: What safety advantages does this process offer over sodium hydride or sodium amide routes?
A: Unlike sodium hydride or sodium amide, which pose severe explosion risks upon contact with moisture and require strictly anhydrous conditions, the methoxide-based system operates safely in alcohol or hydrocarbon solvents. This significantly reduces equipment costs and eliminates the potential for violent hydrolysis incidents during large-scale manufacturing.
Q: Why is the rectification column critical for this specific Claisen condensation?
A: The rectification column continuously removes low-boiling methanol byproduct from the reaction equilibrium. According to Le Chatelier's principle, this shifts the reversible condensation reaction towards product formation, shortening reaction times and minimizing side reactions like self-condensation of the acetophenone starting material.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Avobenzone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to superior synthetic routes requires a partner with deep technical expertise and the infrastructure to support rigorous process validation. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the mixed-alkali catalytic process are fully realized in a GMP-compliant manufacturing environment. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify that every batch of Avobenzone meets the exacting standards required for global cosmetic registration. Our commitment to quality assurance means that we do not just supply chemicals; we deliver validated solutions that integrate seamlessly into your existing formulation workflows.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can optimize your supply chain and reduce your total cost of ownership. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic impact of switching to our high-yield process, backed by specific COA data and route feasibility assessments tailored to your volume requirements. Let us demonstrate how our advanced manufacturing capabilities can secure your supply of critical UV absorbers while driving value through superior quality and operational excellence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
