Advanced Azobenzene Heterocyclic Amide Derivatives for Next-Generation Fungicide Manufacturing
Advanced Azobenzene Heterocyclic Amide Derivatives for Next-Generation Fungicide Manufacturing
Introduction to Novel Fungicidal Chemistry
The agricultural chemical industry is constantly evolving to meet the dual challenges of increasing crop yields and minimizing environmental impact. A significant breakthrough in this domain is documented in patent CN111087345A, which introduces a novel class of azobenzene heterocyclic amide derivatives. These compounds represent a strategic evolution in fungicidal chemistry, specifically designed to combat resistant fungal pathogens while maintaining a favorable safety profile. The core innovation lies in the molecular integration of an azobenzene group into a pyrazole amide scaffold, resulting in a structure that exhibits remarkable inhibitory activity against plant pathogenic bacteria. This technical advancement offers a promising solution for the development of high-efficiency, low-toxicity agrochemical products that align with modern regulatory standards.
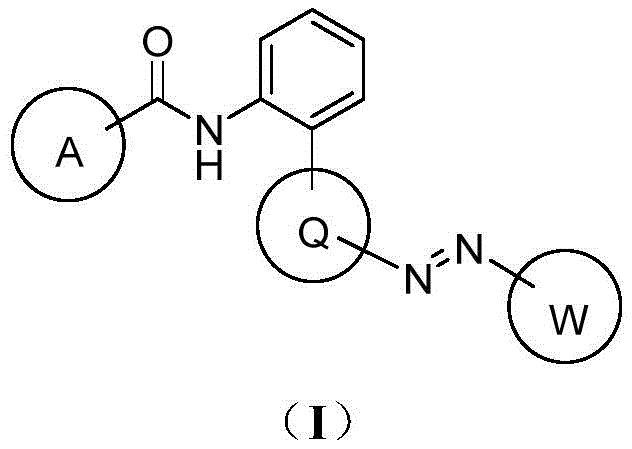
For R&D directors and technical procurement specialists, understanding the structural versatility of these derivatives is crucial. The general formula encompasses a wide range of substituents on rings A, Q, and W, allowing for fine-tuning of physicochemical properties such as solubility, systemic movement, and metabolic stability. This flexibility ensures that the technology can be adapted to various formulation requirements, from emulsifiable concentrates to water-dispersible granules. By leveraging the specific substitution patterns defined in the patent, manufacturers can optimize the active ingredient for specific crop protection scenarios, ensuring maximum efficacy against target diseases like Sclerotinia sclerotiorum and Botrytis cinerea.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional pyrazole amide bactericides have long served as a cornerstone in fungal disease management, primarily by acting on succinate dehydrogenase in the mitochondrial respiratory chain complex II. However, the extensive and often indiscriminate use of these legacy compounds has led to significant challenges, including the emergence of high-resistance risks among fungal populations. Furthermore, many conventional formulations pose potential hazards to aquatic organisms, creating regulatory hurdles and environmental liabilities for agricultural producers. The technical limitations of these older chemistries often necessitate higher application rates to achieve control, which inadvertently increases the chemical load on the ecosystem and accelerates the selection pressure for resistant strains.
The Novel Approach
The novel approach described in the patent addresses these critical pain points through rational molecular design. By introducing the azobenzene functionality, the new derivatives disrupt the binding interactions that typically lead to cross-resistance with existing SDHI fungicides. This structural modification not only restores sensitivity in resistant pathogen populations but also enhances the overall biological activity, allowing for effective control at potentially lower doses. The result is a next-generation active ingredient that combines high efficiency with improved environmental compatibility, offering a sustainable pathway for crop protection that mitigates the risks associated with aquatic toxicity and resistance management.
Mechanistic Insights into Palladium-Catalyzed Biaryl Construction
The synthesis of these advanced intermediates relies on robust and scalable organic transformations, prominently featuring palladium-catalyzed cross-coupling reactions. The construction of the biaryl amine scaffold, a key structural motif in the target molecule, is achieved through a Suzuki-Miyaura coupling reaction. This process involves the reaction of a brominated amide intermediate with an aminophenylboronic acid derivative in the presence of a palladium catalyst, such as [1,1'-bis(diphenylphosphino)ferrocene]palladium(II)dichloride. The reaction is typically conducted in inert solvents like 1,4-dioxane or tetrahydrofuran at elevated temperatures ranging from 60°C to 120°C, ensuring high conversion rates and excellent regioselectivity.
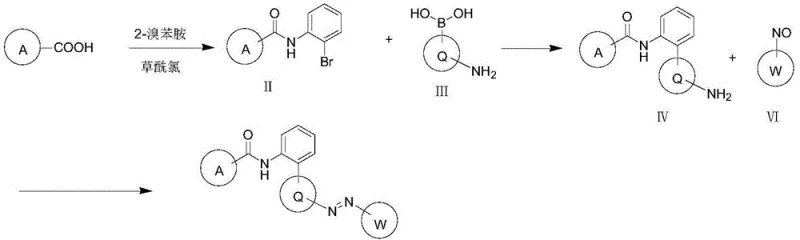
Following the formation of the biaryl amine, the final azobenzene linkage is established through a coupling reaction with a nitroso compound. This step is critical for imparting the unique biological properties of the derivative. The nitroso species, generated in situ via the oxidation of the corresponding aniline using oxidizing agents like potassium hydrogen persulfate, reacts with the amine intermediate under mild acidic conditions. This sequence of reactions demonstrates a high degree of chemical precision, allowing for the incorporation of diverse substituents on the aromatic rings. Such modularity is essential for optimizing the impurity profile and ensuring that the final active ingredient meets the stringent purity specifications required for global agrochemical registration.
How to Synthesize Azobenzene Heterocyclic Amide Efficiently
Implementing this synthesis route on an industrial scale requires careful attention to reaction parameters and purification strategies. The process begins with the activation of the heterocyclic carboxylic acid using oxalyl chloride to generate the reactive acyl chloride species, which is then immediately coupled with 2-bromoaniline. This initial acylation step sets the foundation for the subsequent carbon-carbon bond-forming reactions. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- React heterocyclic carboxylic acid with oxalyl chloride to form acyl chloride, then couple with 2-bromoaniline to obtain the brominated amide intermediate.
- Perform a palladium-catalyzed Suzuki coupling reaction between the brominated amide and an aminophenylboronic acid derivative to construct the biaryl amine scaffold.
- Oxidize the corresponding aniline to a nitroso compound, then couple with the biaryl amine intermediate under acidic conditions to form the final azobenzene derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers substantial benefits for procurement managers and supply chain heads seeking to optimize their sourcing strategies. The reliance on readily available starting materials, such as substituted anilines and heterocyclic acids, significantly reduces the risk of raw material shortages and price volatility. Furthermore, the use of common industrial solvents like dichloromethane and toluene simplifies the solvent recovery and recycling processes, contributing to a more sustainable and cost-effective manufacturing operation. These factors collectively enhance the reliability of the supply chain, ensuring consistent availability of high-quality intermediates for downstream formulation.
- Cost Reduction in Manufacturing: The streamlined synthetic pathway eliminates the need for exotic reagents or complex multi-step sequences that often drive up production costs. By utilizing efficient catalytic systems and avoiding expensive transition metal removal steps where possible, the overall cost of goods sold can be significantly optimized. This economic efficiency allows for competitive pricing in the global agrochemical market without compromising on the quality or performance of the final fungicidal product.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which tolerate a wide range of temperatures and solvent systems, ensures high process reliability. This stability minimizes the risk of batch failures and production delays, which are critical concerns for supply chain planners. Additionally, the modular nature of the synthesis allows for flexible manufacturing schedules, enabling producers to respond quickly to fluctuating market demands and seasonal peaks in agrochemical consumption.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing unit operations that are easily transferred from laboratory to pilot and commercial scales. The generation of waste streams is minimized through efficient atom economy in the coupling steps, and the use of less hazardous oxidants supports compliance with increasingly strict environmental regulations. This alignment with green chemistry principles facilitates smoother regulatory approvals and enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these azobenzene derivatives. The answers are derived directly from the patented technology and are intended to provide clarity for stakeholders evaluating the feasibility of integrating this chemistry into their product portfolios. Understanding these details is essential for making informed decisions about investment and development timelines.
Q: What is the primary biological target of these azobenzene derivatives?
A: These derivatives act as succinate dehydrogenase inhibitors (SDHIs) in the mitochondrial respiratory chain complex II, effectively inhibiting fungal pathogen spore germination and mycelial growth.
Q: How does this new structure address resistance issues in existing fungicides?
A: By introducing an azobenzene moiety into the pyrazole amide structure, the molecular design creates a novel steric and electronic environment that overcomes existing resistance mechanisms found in traditional pyrazole amide bactericides.
Q: Are the starting materials for this synthesis commercially viable?
A: Yes, the synthesis utilizes readily available starting materials such as heterocyclic carboxylic acids, bromoanilines, and substituted anilines, which supports scalable commercial production and supply chain stability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Azobenzene Heterocyclic Amide Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this patented chemistry in the agrochemical sector. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from concept to market. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of azobenzene heterocyclic amide derivative meets the highest international standards for efficacy and safety.
We invite you to collaborate with our technical procurement team to explore how this innovative route can enhance your product lineup. By requesting a Customized Cost-Saving Analysis, you can gain valuable insights into the economic viability of this synthesis. We encourage you to contact us today to obtain specific COA data and comprehensive route feasibility assessments tailored to your specific manufacturing requirements.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
