Revolutionizing Fosfomycin Intermediate Production with Continuous Flow Hydrogenation Technology
The pharmaceutical industry is constantly seeking robust methodologies to enhance the safety and efficiency of antibiotic intermediate production, particularly for critical drugs like fosfomycin. A groundbreaking advancement in this domain is detailed in patent CN115417895A, which discloses a novel synthesis process for cis-propenyl phosphoric acid utilizing continuous flow microchannel technology. This innovation addresses the longstanding challenges associated with the traditional batch hydrogenation of allenyl phosphate, specifically targeting the issues of low selectivity, safety hazards, and excessive catalyst consumption. By transitioning from conventional stirred tank reactors to a sophisticated microchannel reactor system, the process achieves a dramatic reduction in residence time while simultaneously improving the stereochemical outcome of the reaction. For R&D directors and process engineers, this represents a paradigm shift towards inherently safer and more controllable chemical manufacturing, offering a reliable pathway to high-purity pharmaceutical intermediates that meet stringent global regulatory standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of cis-propenyl phosphoric acid has relied heavily on batch-wise hydrogenation processes conducted in standard laboratory or industrial kettles. These traditional methods suffer from significant inherent drawbacks, primarily stemming from poor heat and mass transfer characteristics which are typical of large-volume batch systems. As documented in prior art, such as the study by Su Yunlai, conventional hydrogenation using self-made palladium carbon often results in conversion rates as low as 52.58%, necessitating extensive downstream purification and leading to substantial yield losses. Furthermore, the hydrogenation reaction is highly exothermic, and in a batch setting, the inability to rapidly dissipate heat creates a severe risk of thermal runaway and potential explosion. This safety hazard mandates the use of specialized explosion-proof facilities and certified personnel, thereby inflating capital expenditure and operational overheads. Additionally, the lack of precise control over reaction parameters in batch reactors often leads to the formation of undesirable trans-isomers and over-hydrogenated by-products, compromising the overall purity and efficacy of the final antibiotic product.
The Novel Approach
In stark contrast to these legacy methods, the novel approach outlined in the patent leverages the superior engineering capabilities of microchannel reactor technology to redefine the synthesis landscape. By utilizing a continuous flow system, such as the Corning G1-10FM Hybrid reactor, the process achieves a mass transfer and mixing efficiency that is approximately 100 times greater than that of traditional tank reactors. This enhanced mixing ensures that the allenyl phosphate substrate interacts uniformly with the hydrogen gas and the palladium-carbon catalyst, driving the reaction to completion with remarkable speed. The residence time is drastically shortened to merely 4 minutes, compared to the 18 hours or more required in batch processes, which not only boosts throughput but also kinetically favors the formation of the desired cis-isomer. The modular nature of the microchannel system allows for precise tuning of temperature and pressure, effectively suppressing side reactions and ensuring a consistent product quality that is difficult to achieve in batch operations, making it an ideal solution for cost reduction in pharmaceutical intermediates manufacturing.
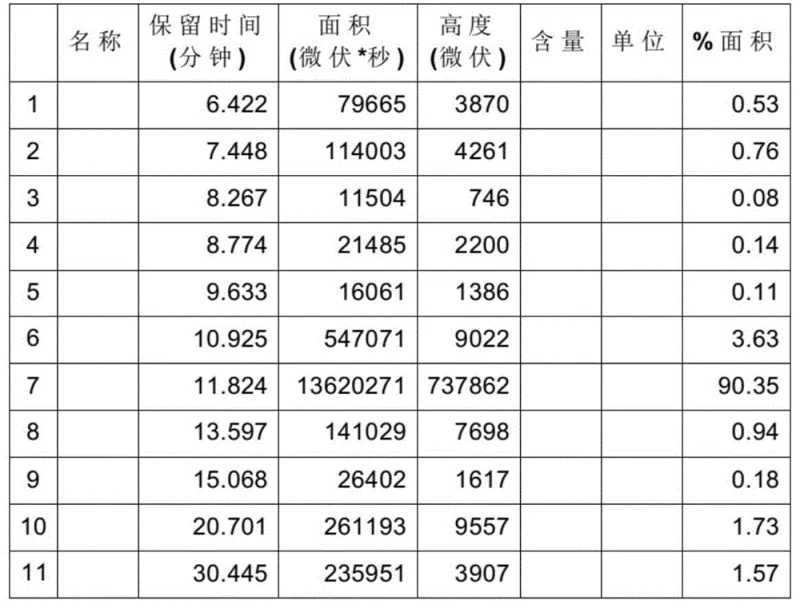
Mechanistic Insights into Microchannel-Catalyzed Hydrogenation
The core mechanism driving the success of this synthesis lies in the unique hydrodynamic environment created within the microchannels of the reactor. Unlike bulk fluid dynamics in a stirred tank, the flow within microchannels is typically laminar yet highly efficient due to the small diffusion distances. When the slurry containing allenyl phosphate and the palladium-carbon catalyst is pumped through the heated modules, the high surface-to-volume ratio facilitates instantaneous heat exchange, maintaining the reaction mixture at an optimal temperature range of 60 to 100°C. This thermal precision is critical because it prevents local hot spots that could otherwise trigger the isomerization of the double bond from the cis-configuration to the thermodynamically more stable but undesired trans-configuration. The rapid quenching of the reaction mixture immediately upon exiting the reactor further locks in the kinetic product, preserving the high cis-selectivity of greater than 97%. This mechanistic control is essential for R&D teams aiming to minimize impurity profiles and simplify the crystallization or extraction steps required to meet pharmacopeial specifications for fosfomycin precursors.
Furthermore, the interaction between the heterogeneous palladium-carbon catalyst and the gaseous hydrogen is significantly intensified in this flow regime. The patent specifies a mass ratio of allenyl phosphate to catalyst of 1:(0.0004-0.001), which is remarkably low compared to batch requirements. In the microchannel environment, the catalyst particles are suspended in a high-velocity stream that prevents settling and ensures maximum exposure of the active catalytic sites to the reactants. This efficient utilization of the catalyst surface area means that the reaction proceeds with high turnover frequency, allowing the same batch of catalyst to be recycled multiple times without significant deactivation. The ability to reuse the catalyst for up to 6 cycles is a direct consequence of the mild reaction conditions and the absence of mechanical shear forces that often degrade catalyst integrity in stirred tanks. This mechanistic advantage translates directly into economic value by reducing the dependency on expensive noble metals and minimizing the generation of hazardous metal-containing waste streams.
How to Synthesize Cis-Propenyl Phosphoric Acid Efficiently
The implementation of this continuous flow synthesis requires a systematic approach to ensure optimal performance and safety. The process begins with the preparation of a homogeneous feed solution where allenyl phosphate is dissolved in a lower alcohol solvent, with methanol being the preferred choice due to its cost-effectiveness and ease of recovery. This solution is then mixed with the palladium-carbon catalyst to form a pumpable slurry, which is filtered to remove any large agglomerates that could clog the microchannels. The clarified slurry is subsequently introduced into the preheating module of the reactor system, where it is brought to the target reaction temperature before entering the main reaction zone. Detailed standardized synthetic steps see the guide below.
- Dissolve allenyl phosphate in an organic solvent such as methanol to create a mixed solution with a concentration between 0.05 and 1.5 mol/L.
- Add a palladium-carbon catalyst to the mixture at a mass ratio of 1: (0.0004-0.001), filter the slurry, and pump it into the preheating module of the microchannel reactor.
- React the preheated slurry with hydrogen gas at 60-100°C and 1-10 bar pressure within the reaction module, then filter and concentrate the effluent to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this microchannel-based synthesis offers compelling strategic advantages that extend beyond mere technical performance. The primary benefit lies in the drastic simplification of the supply chain for raw materials, particularly the palladium-carbon catalyst. Since the catalyst can be reused multiple times without replenishment, the frequency of purchasing this high-value commodity is significantly reduced, leading to stabilized inventory levels and reduced working capital requirements. Moreover, the use of common organic solvents like methanol or ethanol, which are readily available in bulk quantities globally, eliminates the reliance on exotic or hard-to-source reagents that often plague complex pharmaceutical syntheses. This accessibility ensures a robust and resilient supply chain capable of withstanding market fluctuations and logistical disruptions, thereby guaranteeing the continuous availability of this critical antibiotic intermediate for downstream manufacturing.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, driven primarily by the elimination of expensive safety infrastructure and the optimization of catalyst usage. Traditional batch hydrogenation requires specialized explosion-proof workshops and certified operators, representing a significant fixed cost burden. By adopting the microchannel reactor, which operates with a minimal hold-up volume and intrinsic safety features, manufacturers can potentially operate in standard chemical processing facilities, thereby drastically lowering capital investment and insurance premiums. Additionally, the ability to recycle the palladium-carbon catalyst for up to six cycles means that the effective cost of the catalyst per kilogram of product is fractioned significantly. This reduction in consumable costs, combined with the higher yield and purity which reduces waste disposal fees, results in a leaner and more profitable manufacturing operation without compromising on quality standards.
- Enhanced Supply Chain Reliability: The continuous nature of the flow chemistry process inherently supports a more reliable supply chain compared to batch processing. Batch operations are prone to variability between runs, often requiring lengthy cleaning and setup times that create bottlenecks in production scheduling. In contrast, the microchannel system can run continuously for extended periods, providing a steady stream of product that aligns perfectly with just-in-time manufacturing principles. This consistency reduces the need for large safety stocks of finished goods, freeing up warehouse space and reducing the risk of product degradation during storage. Furthermore, the scalability of the technology is linear; increasing production capacity simply involves running the reactor for longer durations or numbering up the reactor modules, rather than building entirely new large-scale vessels. This flexibility allows suppliers to respond rapidly to spikes in demand for fosfomycin, ensuring that downstream drug manufacturers never face shortages of this vital intermediate.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this process offers a cleaner and more sustainable production route. The high selectivity of the reaction means that fewer by-products are generated, which simplifies the wastewater treatment process and reduces the load on effluent treatment plants. The closed-loop nature of the continuous flow system also minimizes the emission of volatile organic compounds (VOCs) and hydrogen gas, enhancing workplace safety and ensuring compliance with increasingly strict environmental regulations. The ability to scale up without the 'amplification effect'—where reaction outcomes change unpredictably as vessel size increases—is a key advantage for commercial expansion. Manufacturers can confidently scale from pilot plant quantities to multi-ton annual production capacities knowing that the reaction parameters remain constant, ensuring that the environmental footprint per unit of product remains low even as total output increases significantly.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this microchannel hydrogenation technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this process compares to existing industry standards. Understanding these details is crucial for stakeholders evaluating the feasibility of adopting this advanced synthesis route for their own production lines or supply networks.
Q: How does the microchannel reactor improve safety compared to traditional batch hydrogenation?
A: Traditional batch hydrogenation carries a high risk of explosion due to large reactant volumes and poor heat dissipation. The microchannel reactor minimizes the holding volume of reactive materials and enhances heat transfer efficiency by approximately 1000 times, effectively eliminating the risk of thermal runaway and ensuring intrinsic safety during the exothermic hydrogenation process.
Q: What is the selectivity advantage of this new synthesis process?
A: The process achieves a cis-selectivity greater than 97%, significantly higher than conventional methods. The precise control over residence time (approx. 4 minutes) and temperature in the microchannel reactor prevents over-hydrogenation and suppresses the formation of the unwanted trans-isomer, resulting in a crude purity exceeding 85%.
Q: Can the palladium-carbon catalyst be reused in this continuous flow system?
A: Yes, the palladium-carbon catalyst demonstrates excellent reusability. It can be recycled and reused for up to 6 cycles without significant loss in catalytic activity or conversion rates. This reusability drastically reduces the consumption of precious metal catalysts, contributing to substantial raw material cost optimization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cis-Propenyl Phosphoric Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the global antibiotic supply chain. Our team of expert chemists and engineers possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are fully equipped to implement advanced continuous flow technologies like the one described in CN115417895A, leveraging our state-of-the-art facilities to deliver cis-propenyl phosphoric acid with stringent purity specifications. Our rigorous QC labs employ advanced analytical techniques to verify every batch, guaranteeing that the cis-selectivity and impurity profiles meet the exacting demands of pharmaceutical grade manufacturing.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. By partnering with us, you gain access to specific COA data and comprehensive route feasibility assessments that demonstrate the tangible benefits of switching to our superior production methods. Contact us today to discuss how we can support your fosfomycin production goals with reliable, safe, and cost-effective solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
