Scalable Synthesis of Heterocyclic Ortho-Iodo Sulfides via Metal-Free C-S Coupling for Pharmaceutical Applications
Introduction to Patent CN113683559A
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing carbon-sulfur bonds, a critical structural motif found in numerous bioactive molecules and active pharmaceutical ingredients (APIs). Patent CN113683559A introduces a groundbreaking approach for the preparation of heterocyclic ortho-iodo sulfides, utilizing a metal-free strategy that fundamentally shifts the paradigm of C-S bond formation. Unlike conventional cross-coupling reactions that rely heavily on transition metal catalysts, this invention employs sodium hydride (NaH) as a potent base to facilitate the nucleophilic substitution between heterocyclic thiophenols and o-diiodobenzene. This technical breakthrough addresses long-standing challenges regarding catalyst deactivation and harsh reaction conditions, offering a streamlined pathway for synthesizing complex 1,2-disubstituted benzenes. For R&D directors and process chemists, this represents a significant opportunity to simplify synthetic routes while maintaining high atom economy and product purity standards required for downstream drug development.
The core innovation lies in the ability to perform this transformation under exceptionally mild conditions, specifically at room temperature, which drastically reduces energy consumption and operational complexity. By leveraging the high reactivity of o-diiodobenzene in the presence of a strong base, the method avoids the need for pre-functionalized active species such as iodonium or sulfonium salts, which are often costly and difficult to handle on a large scale. This patent not only provides a specific synthetic protocol but also opens up a versatile platform for generating diverse heterocyclic scaffolds essential for modern medicinal chemistry. As a reliable pharma intermediate supplier, understanding and adopting such efficient technologies is crucial for maintaining competitiveness in the global supply chain, ensuring that high-purity building blocks are available for the synthesis of next-generation therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of aryl thioether compounds has been dominated by transition metal-catalyzed cross-coupling reactions, primarily utilizing palladium, copper, or nickel systems. While these methods are theoretically efficient, they suffer from significant practical drawbacks that hinder their widespread industrial application, particularly in the context of cost reduction in pharmaceutical manufacturing. A major issue is the propensity of sulfur-containing reagents to coordinate strongly with metal centers, leading to catalyst poisoning and deactivation. To counteract this, traditional protocols often necessitate the use of sophisticated, air-sensitive, and expensive ligands, alongside severe reaction conditions such as strong alkalis like sodium tert-butoxide and elevated temperatures ranging from 80°C to 140°C. These requirements not only inflate the raw material costs but also introduce complex purification steps to remove trace metal residues, which is a critical quality attribute for API intermediates. Furthermore, the sensitivity of these catalysts to moisture and oxygen demands rigorous inert atmosphere controls, adding to the operational burden and safety risks in a production environment.
The Novel Approach
In stark contrast to the cumbersome metal-catalyzed pathways, the novel approach disclosed in the patent utilizes a simple yet highly effective metal-free system driven by sodium hydride. This method capitalizes on the intrinsic nucleophilicity of thiolates generated in situ, allowing for a direct SNAr-type displacement on the electron-deficient o-diiodobenzene ring. The reaction proceeds smoothly at room temperature in a mixed solvent system of THF and DMA, eliminating the need for external heating and the associated energy costs. By removing transition metals entirely from the equation, the process inherently avoids the issues of catalyst poisoning and metal contamination, resulting in a cleaner crude product profile that simplifies downstream purification. 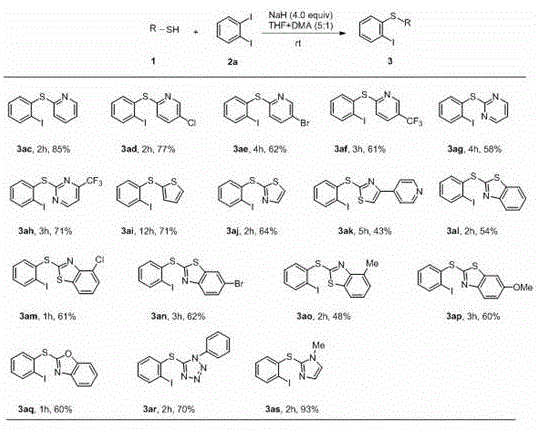 As illustrated in the reaction scheme, the conversion of pyridylthiol and o-diiodobenzene to the target heterocyclic o-iodo sulfide (3ac) is achieved with impressive efficiency using 4.0 equivalents of NaH. This simplicity translates directly into commercial advantages, as it allows for the use of standard stainless steel reactors without the need for specialized lining or passivation required for corrosive metal catalysts, thereby facilitating the commercial scale-up of complex pharmaceutical intermediates.
As illustrated in the reaction scheme, the conversion of pyridylthiol and o-diiodobenzene to the target heterocyclic o-iodo sulfide (3ac) is achieved with impressive efficiency using 4.0 equivalents of NaH. This simplicity translates directly into commercial advantages, as it allows for the use of standard stainless steel reactors without the need for specialized lining or passivation required for corrosive metal catalysts, thereby facilitating the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into NaH-Mediated Nucleophilic Substitution
The mechanistic foundation of this transformation rests on the powerful deprotonating capability of sodium hydride, which acts as a non-nucleophilic base to quantitatively generate the corresponding thiolate anion from the heterocyclic thiophenol substrate. Upon addition of the thiol to the suspension of NaH in THF/DMA, rapid deprotonation occurs, creating a highly nucleophilic sulfur species that is primed for attack. The choice of o-diiodobenzene as the electrophile is strategic; the presence of two iodine atoms creates a sufficiently electron-deficient aromatic system that is susceptible to nucleophilic aromatic substitution, even without the activation typically provided by a metal catalyst. The first iodine atom serves as the leaving group during the initial C-S bond formation, while the second ortho-iodine substituent remains intact, providing a valuable handle for further functionalization. This orthogonal reactivity is key to the utility of the product, as the remaining C-I bond can be exploited in subsequent palladium-catalyzed cross-couplings or cyclization reactions to build even more complex molecular architectures.
From an impurity control perspective, the mechanism offers distinct advantages over radical-based or metal-catalyzed pathways. Since the reaction proceeds via a concerted nucleophilic substitution rather than a radical chain mechanism, the formation of homocoupled disulfide byproducts is minimized, provided that the reaction is conducted under appropriate conditions. The use of a slight excess of o-diiodobenzene (2.0 equivalents relative to the thiol) ensures that the thiolate is fully consumed, preventing the accumulation of unreacted starting material which can be difficult to separate from the product. Furthermore, the absence of transition metals means there is no risk of metal-mediated side reactions such as Ullmann-type couplings or desulfurization, which often plague traditional C-S bond formations. The result is a reaction profile characterized by high selectivity and a single major product, as evidenced by the clean NMR spectra reported in the patent data, which significantly reduces the burden on analytical quality control teams during batch release.
How to Synthesize Heterocyclic Ortho-Iodo Sulfide Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to the stoichiometry and solvent composition to maximize yield and reproducibility. The patent outlines a straightforward procedure where sodium hydride is first suspended in anhydrous tetrahydrofuran (THF), followed by the dropwise addition of the heterocyclic thiophenol dissolved in dimethylacetamide (DMA). This specific solvent combination is critical, as DMA helps solubilize the polar thiolate intermediate while THF maintains a manageable viscosity for stirring and heat dissipation. The detailed standardized synthesis steps below provide a clear roadmap for chemists to replicate the high yields reported in the intellectual property, ensuring that the process can be transferred seamlessly from R&D to production scales.
- Suspend sodium hydride (4.0 equiv) in anhydrous THF at room temperature under magnetic stirring.
- Add heterocyclic thiophenol (1.0 equiv, dissolved in DMA) dropwise and stir for 3 minutes to generate the thiolate species.
- Add o-diiodobenzene (2.0 equiv, dissolved in THF) to the mixture, stir at room temperature until TLC indicates completion, then quench and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this metal-free C-S coupling technology presents a compelling value proposition centered on cost stability and supply security. Traditional methods relying on palladium or copper catalysts are subject to significant price volatility driven by the mining and refining of precious metals, whereas sodium hydride is a commodity chemical with a stable and abundant global supply. By eliminating the dependency on these fluctuating metal markets, manufacturers can achieve substantial cost savings in raw material procurement, leading to more predictable budgeting and pricing strategies for their customers. Additionally, the removal of expensive ligands and the reduction in reaction temperature contribute to a lower overall cost of goods sold (COGS), making the final intermediates more competitive in the marketplace without sacrificing quality or performance.
- Cost Reduction in Manufacturing: The economic benefits of this process extend beyond simple reagent costs to include significant savings in operational expenditures. The elimination of transition metal catalysts removes the need for costly metal scavenging resins or complex recrystallization steps designed to meet strict residual metal specifications (e.g., ICH Q3D guidelines). This simplification of the purification train reduces solvent consumption and waste generation, directly lowering the environmental compliance costs associated with hazardous waste disposal. Furthermore, the ability to run the reaction at room temperature eliminates the energy demand associated with heating large reactor vessels for extended periods, contributing to a greener and more energy-efficient manufacturing footprint that aligns with modern sustainability goals.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available starting materials such as o-diiodobenzene and various heterocyclic thiols, which are produced by multiple vendors globally. Unlike specialized catalysts that may have long lead times or single-source dependencies, the reagents for this process can be sourced competitively, reducing the risk of production stoppages due to material shortages. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality or environmental factors, ensuring consistent batch-to-batch performance. This reliability is crucial for maintaining continuous supply to downstream API manufacturers, who depend on uninterrupted delivery of high-quality intermediates to meet their own production schedules and regulatory commitments.
- Scalability and Environmental Compliance: From a scale-up perspective, the simplicity of the NaH-mediated protocol makes it highly amenable to large-scale production in multi-purpose chemical plants. The absence of air-sensitive catalysts simplifies the engineering requirements, allowing the reaction to be performed in standard glass-lined or stainless steel reactors without the need for exotic materials of construction. The workup procedure, involving a simple aqueous quench and extraction, is easily adaptable to continuous processing or large batch operations, facilitating the rapid expansion of capacity to meet market demand. Moreover, the reduced use of heavy metals and the potential for solvent recycling contribute to a lower environmental impact, helping companies meet increasingly stringent regulatory standards for emissions and effluent treatment while enhancing their corporate social responsibility profiles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis method. These answers are derived directly from the experimental data and technical disclosures within the patent, providing clarity on the feasibility and advantages of the process for potential partners and licensees. Understanding these details is essential for evaluating the fit of this technology within existing manufacturing portfolios and for assessing the potential return on investment for process optimization initiatives.
Q: Why is the NaH-mediated method superior to traditional palladium-catalyzed C-S coupling?
A: Traditional methods often suffer from catalyst poisoning due to sulfur coordination, requiring expensive ligands and high temperatures. The NaH method operates at room temperature without transition metals, eliminating metal residue concerns and reducing raw material costs significantly.
Q: What is the role of the solvent mixture THF and DMA in this reaction?
A: The combination of THF and DMA optimizes the solubility of both the inorganic base (NaH) and the organic substrates. A volume ratio of roughly 3:1 to 8:1 ensures efficient mass transfer and stabilizes the reactive thiolate intermediate, leading to higher isolated yields.
Q: Can this process be scaled for industrial production of API intermediates?
A: Yes, the process uses commercially available, low-cost reagents like o-diiodobenzene and sodium hydride. The mild room temperature conditions and simple aqueous workup make it highly suitable for large-scale manufacturing without specialized high-pressure or high-temperature equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Heterocyclic Ortho-Iodo Sulfide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of metal-free coupling technologies in the synthesis of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of heterocyclic ortho-iodo sulfide delivered meets the highest standards of quality and consistency required by global regulatory agencies. We are committed to leveraging our technical expertise to optimize this NaH-mediated route, delivering cost-effective solutions that accelerate our clients' drug development timelines.
We invite forward-thinking R&D and procurement leaders to collaborate with us to explore the full capabilities of this synthesis platform. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and profitability in your supply chain. Let us be your trusted partner in navigating the complexities of fine chemical manufacturing and securing a competitive edge in the global marketplace.
