Advanced Synthesis of Chlorobromoalkyl Phosphate Flame Retardants for Commercial Scale-Up
Advanced Synthesis of Chlorobromoalkyl Phosphate Flame Retardants for Commercial Scale-Up
The global demand for high-performance flame retardants has intensified as regulatory standards for polymer safety become increasingly stringent across the automotive, construction, and electronics sectors. Patent CN101016463A introduces a sophisticated methodology for preparing chlorobromoalkyl phosphate flame retardants, representing a significant technological leap in the field of polymer additives. This innovation leverages a synergistic mechanism involving chlorine, bromine, and phosphorus elements to achieve superior flame retardant efficiency while mitigating the environmental and stability drawbacks associated with traditional halogenated systems. For R&D Directors and Procurement Managers seeking a reliable polymer additive supplier, understanding the underlying chemical architecture of this patent is crucial for evaluating its potential integration into existing manufacturing lines. The process utilizes readily available raw materials such as pentaerythritol, phosphorus trichloride, liquid bromine, and epichlorohydrin, ensuring that the supply chain remains robust and cost-effective. By addressing the critical balance between flame retardancy, thermal stability, and hydrolysis resistance, this technology offers a compelling value proposition for the production of high-purity flame retardants intended for use in plastics, rubber, and synthetic fabrics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the flame retardant industry has relied heavily on organic brominated compounds due to their exceptional efficiency, which is generally 2.5 to 4 times higher than that of chlorine-based analogs. However, these conventional brominated flame retardants suffer from significant technical deficiencies that limit their long-term viability in high-performance applications. A primary concern is the reduction of anti-ultraviolet stability in the flame-retarded substrate, which leads to premature degradation of materials exposed to sunlight or harsh weather conditions. Furthermore, during combustion, traditional brominated systems tend to generate substantial amounts of smoke, corrosive gases, and toxic byproducts, posing serious environmental and safety hazards. The inherent weakness of the bromine-carbon bond, while beneficial for flame inhibition, also contributes to higher volatility and potential migration issues within the polymer matrix over time. These factors collectively create a pressing need for advanced formulations that can maintain high flame retardant performance without compromising the physical integrity or environmental profile of the final product. For Supply Chain Heads, relying on these legacy chemistries often means navigating complex regulatory landscapes and managing the risks associated with potential bans or restrictions on specific brominated compounds.
The Novel Approach
The methodology disclosed in patent CN101016463A presents a transformative solution by integrating chlorine, bromine, and phosphorus into a single molecular structure, thereby harnessing the synergistic effects of all three elements. This novel approach ensures that flame retardancy occurs simultaneously in both the gas phase and the condensed phase, significantly enhancing the overall protection efficiency compared to single-element additives. The presence of phosphorus promotes the formation of a protective carbonaceous char layer on the polymer surface, which acts as a physical barrier against heat and mass transfer, while the halogen components effectively scavenge free radicals in the flame zone. Crucially, the inclusion of chlorine helps to overcome the UV stability issues typically associated with pure brominated systems, resulting in a material that maintains its performance characteristics over extended periods of exposure. Additionally, the specific chemical structure designed in this patent exhibits low volatility and excellent hydrolysis resistance, addressing common failure modes found in earlier generations of phosphate esters. This comprehensive improvement in material properties makes the chlorobromoalkyl phosphate flame retardant an ideal candidate for cost reduction in flame retardant manufacturing, as it reduces the need for secondary stabilizers and extends the service life of the end products.
Mechanistic Insights into Halogen-Phosphorus Synergistic Esterification
The core of this technological advancement lies in a meticulously engineered three-step synthesis route that maximizes yield and purity while minimizing waste. The process begins with the esterification of pentaerythritol and phosphorus trichloride in a toluene solvent system, where precise control of the molar ratio at 1:2.5 is essential to drive the reaction towards the formation of Intermediate I, identified as 3,9-dichloro-2,4,8,10-tetraoxo-3,9-diphosphaspiro[5.5]undecane. This initial step establishes the phosphorus-rich backbone of the molecule, which is critical for the condensed-phase flame retardant mechanism. Following the removal of solvents and excess reagents, the Intermediate I undergoes a bromination reaction in carbon tetrachloride, where liquid bromine is introduced at a molar ratio of 1:2.1. This step is highly exothermic and requires careful temperature management between 45°C and 50°C to ensure selective substitution without degrading the sensitive phosphorus ester linkages. The resulting Intermediate II, 2,2-dibromomethylpropylbromodiphosphoryl chloride, serves as the key precursor that introduces the bromine atoms necessary for gas-phase radical scavenging. 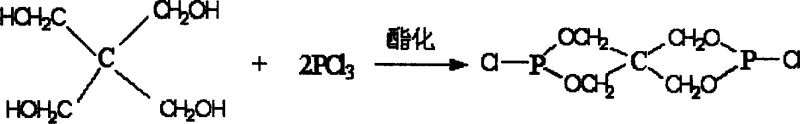
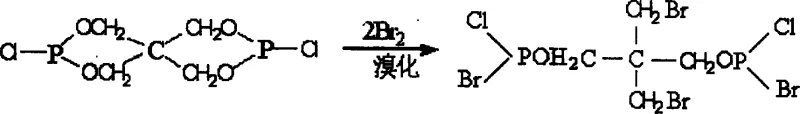
The final and most critical stage involves the ring-opening condensation of Intermediate II with epichlorohydrin, catalyzed by anhydrous aluminum trichloride. This reaction is conducted with a molar ratio of 1:4.2 (Intermediate II to epichlorohydrin) and involves a temperature ramp from 50°C to 65°C over a period of 3 to 7 hours. The use of epichlorohydrin not only introduces the chlorine component but also extends the alkyl chains, improving the compatibility of the flame retardant with various polymer matrices such as polyvinyl chloride, polystyrene, and polyurethane foams. The anhydrous aluminum trichloride catalyst facilitates the nucleophilic attack of the phosphorus-chlorine bonds by the epoxide ring, ensuring high conversion rates and minimizing the formation of unwanted byproducts. The final product, 2,2-dibromomethyl-1,3-bis[2-bromo-3-chloropropyl-2-chloro-3-chloropropyl phosphate]propane, is isolated as a yellow transparent viscous liquid with a crude yield reaching up to 94.1%. 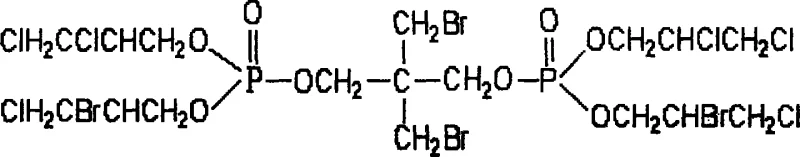 This high yield and structural integrity are paramount for R&D Directors focusing on the purity and impurity profile of the commercial scale-up of complex polymer additives, as it ensures consistent performance in downstream applications without the need for extensive purification steps that would otherwise drive up costs.
This high yield and structural integrity are paramount for R&D Directors focusing on the purity and impurity profile of the commercial scale-up of complex polymer additives, as it ensures consistent performance in downstream applications without the need for extensive purification steps that would otherwise drive up costs.
How to Synthesize Chlorobromoalkyl Phosphate Efficiently
The synthesis of this advanced flame retardant requires precise adherence to the reaction conditions outlined in the patent to ensure safety and reproducibility on an industrial scale. The process involves handling reactive chemicals such as phosphorus trichloride and liquid bromine, necessitating robust engineering controls and specialized equipment capable of managing exothermic events and corrosive byproducts like hydrogen chloride gas. Detailed standardized synthesis steps are essential for maintaining batch-to-batch consistency and achieving the target physical properties, including acid value, water content, and viscosity, as specified in the patent documentation. The operational simplicity of the third step, utilizing epichlorohydrin as a cyclization reagent, further enhances the feasibility of this route for large-scale manufacturing, reducing the complexity typically associated with multi-step organophosphorus synthesis.
- Esterification of pentaerythritol with phosphorus trichloride in toluene to form Intermediate I.
- Bromination of Intermediate I with liquid bromine in carbon tetrachloride to generate Intermediate II.
- Ring-opening condensation of Intermediate II with epichlorohydrin using anhydrous aluminum trichloride catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this chlorobromoalkyl phosphate synthesis route offers substantial strategic benefits for procurement and supply chain operations within the fine chemical sector. The reliance on commodity chemicals such as pentaerythritol, phosphorus trichloride, and epichlorohydrin ensures that raw material sourcing is not constrained by geopolitical risks or limited availability of exotic precursors. This accessibility translates directly into enhanced supply chain reliability, allowing manufacturers to maintain continuous production schedules even during periods of market volatility. Furthermore, the elimination of expensive transition metal catalysts in favor of anhydrous aluminum trichloride, which is used in catalytic amounts and can be managed effectively during workup, contributes to significant cost optimization in the overall production budget. The high crude yield of 94.1% reported in the patent examples indicates a highly efficient atom economy, reducing the volume of waste generated per unit of product and lowering the burden on waste treatment facilities. For Supply Chain Heads, these factors collectively reduce lead time for high-purity flame retardants by streamlining the manufacturing process and minimizing the need for complex purification stages that often bottleneck production capacity.
- Cost Reduction in Manufacturing: The process design inherently lowers production costs by utilizing a straightforward three-step sequence that avoids the need for high-pressure or cryogenic conditions, thereby reducing energy consumption and capital expenditure on specialized reactor systems. The use of toluene and carbon tetrachloride as solvents, while requiring careful environmental management, allows for efficient recovery and recycling, further driving down operational expenses. By achieving a synergistic flame retardant effect, the additive loading levels in the final polymer can potentially be optimized, meaning less additive is required to achieve the same fire safety rating, which directly impacts the bill of materials for downstream customers. This logical deduction of cost efficiency is based on the chemical mechanism rather than arbitrary financial projections, providing a solid foundation for long-term pricing strategies.
- Enhanced Supply Chain Reliability: The raw materials identified in the patent are produced globally in large volumes for various chemical industries, ensuring a stable and diversified supply base that mitigates the risk of single-source dependency. The robustness of the reaction conditions, particularly the tolerance for standard industrial temperature ranges between 50°C and 80°C, means that the process can be easily transferred between different manufacturing sites without requiring extensive requalification or equipment modification. This flexibility is crucial for maintaining business continuity and meeting the just-in-time delivery expectations of major polymer manufacturers. Additionally, the stability of the intermediates allows for potential decoupling of the synthesis steps, enabling the stocking of key precursors to buffer against short-term supply disruptions of the final reagents.
- Scalability and Environmental Compliance: The synthesis route is designed with scalability in mind, as evidenced by the successful demonstration of the reaction in standard laboratory glassware which translates well to stirred tank reactors used in commercial plants. The workup procedure involving alkaline washing with sodium carbonate and water washing to neutrality is a standard unit operation in the chemical industry, facilitating easy integration into existing wastewater treatment protocols. While the use of halogenated solvents requires adherence to strict environmental regulations, the closed-loop nature of the recommended process minimizes emissions and exposure risks. The resulting product's low toxicity and hydrolysis resistance also align with the growing demand for sustainable and safe chemical additives, reducing the regulatory burden on customers who incorporate this flame retardant into consumer-facing products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this flame retardant technology, based on the specific data points and claims made within the patent documentation. Understanding these details is essential for stakeholders evaluating the feasibility of adopting this new chemical route for their specific application needs. These answers provide a clear framework for assessing the technical merits and operational requirements associated with the production and use of chlorobromoalkyl phosphate flame retardants.
Q: What is the primary advantage of the Cl-Br-P synergistic mechanism?
A: The combination of chlorine, bromine, and phosphorus provides simultaneous gas-phase and condensed-phase flame retardancy, overcoming the UV instability of pure brominated systems while maintaining high efficiency.
Q: How does this process improve supply chain reliability?
A: By utilizing readily available commodity chemicals like pentaerythritol and epichlorohydrin, the process reduces dependency on scarce specialty reagents, ensuring consistent production continuity.
Q: Is the synthesis scalable for industrial production?
A: Yes, the patent describes a robust three-step sequence with manageable exothermic controls and standard workup procedures, achieving crude yields up to 94.1% suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chlorobromoalkyl Phosphate Flame Retardant Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating innovative patent technologies like CN101016463A into reliable commercial realities for our global partners. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory synthesis to industrial manufacturing is seamless and efficient. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch against the high standards required for high-purity flame retardants. We understand that the consistency of the chlorine, bromine, and phosphorus ratio is vital for the synergistic performance of the additive, and our advanced analytical capabilities guarantee that this balance is maintained throughout the production process. By partnering with us, you gain access to a supply chain that is not only robust but also deeply knowledgeable about the nuances of organophosphorus chemistry and polymer additive formulation.
We invite you to engage with our technical procurement team to discuss how this specific chlorobromoalkyl phosphate technology can be integrated into your product portfolio to achieve superior flame retardancy and cost efficiency. We are prepared to provide a Customized Cost-Saving Analysis that evaluates the specific economic benefits of switching to this synergistic additive based on your current formulation and volume requirements. Please contact us to request specific COA data and route feasibility assessments, allowing us to demonstrate our capability to deliver this complex intermediate with the reliability and precision your business demands. Our team is ready to support your R&D and supply chain objectives with tailored solutions that drive value and innovation.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
