Advanced Synthesis of Indene Compounds for High-Performance Metallocene Catalyst Manufacturing
The landscape of fine chemical manufacturing for polymer additives is undergoing a significant transformation, driven by the need for more efficient and cost-effective synthetic routes for critical intermediates. Patent CN101318887B introduces a groundbreaking methodology for the preparation of indene compounds, which serve as essential ligands in the synthesis of metallocene catalysts used extensively in polyolefin production. This technology addresses long-standing inefficiencies in traditional synthesis by utilizing substituted benzoate compounds as readily available starting materials. By leveraging a novel combination of Lewis acids and inorganic or organic acids under dehydration cyclization conditions, this process converts benzoates directly into indenone intermediates with remarkable efficiency. The subsequent reduction and dehydration steps yield high-purity indene derivatives suitable for demanding applications in the petrochemical and advanced materials sectors. For R&D directors and procurement specialists, understanding the mechanistic advantages of this patent is crucial for optimizing supply chains and reducing the total cost of ownership for catalyst precursor manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of indene compounds has relied on complex multi-step pathways that often begin with substituted aromatic hydrocarbons or benzyl chlorides. As illustrated in the traditional reaction schemes, these methods typically involve Friedel-Crafts acylation followed by cyclization, hydrolysis, and subsequent functional group manipulations. 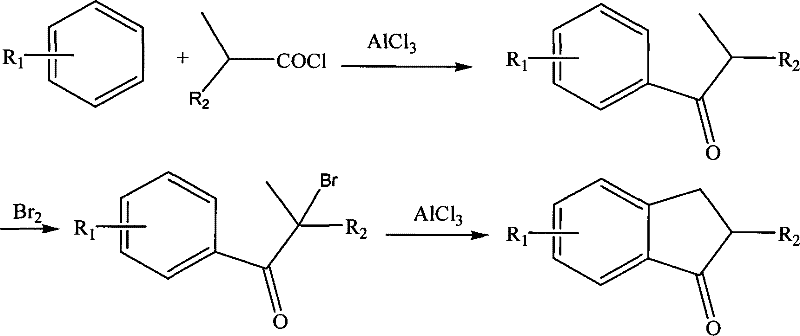 These conventional routes are plagued by several critical drawbacks that impact commercial viability. Firstly, the requirement for acyl chlorides introduces significant safety hazards and handling costs due to their corrosive nature and moisture sensitivity. Secondly, the multi-step nature of the synthesis leads to cumulative yield losses, where the overall efficiency is drastically reduced by the inefficiency of each individual transformation. Furthermore, the use of aryl-aryl coupling technologies to introduce specific substituents often requires expensive transition metal catalysts and rigorous purification steps to remove metal residues, which is detrimental to the performance of downstream metallocene catalysts. The time-consuming nature of these processes also results in extended lead times, creating bottlenecks in the supply chain for polymer manufacturers who require consistent and timely delivery of high-quality intermediates.
These conventional routes are plagued by several critical drawbacks that impact commercial viability. Firstly, the requirement for acyl chlorides introduces significant safety hazards and handling costs due to their corrosive nature and moisture sensitivity. Secondly, the multi-step nature of the synthesis leads to cumulative yield losses, where the overall efficiency is drastically reduced by the inefficiency of each individual transformation. Furthermore, the use of aryl-aryl coupling technologies to introduce specific substituents often requires expensive transition metal catalysts and rigorous purification steps to remove metal residues, which is detrimental to the performance of downstream metallocene catalysts. The time-consuming nature of these processes also results in extended lead times, creating bottlenecks in the supply chain for polymer manufacturers who require consistent and timely delivery of high-quality intermediates.
The Novel Approach
In stark contrast to the cumbersome traditional methods, the technology disclosed in CN101318887B offers a streamlined and robust alternative that fundamentally reimagines the synthetic logic. 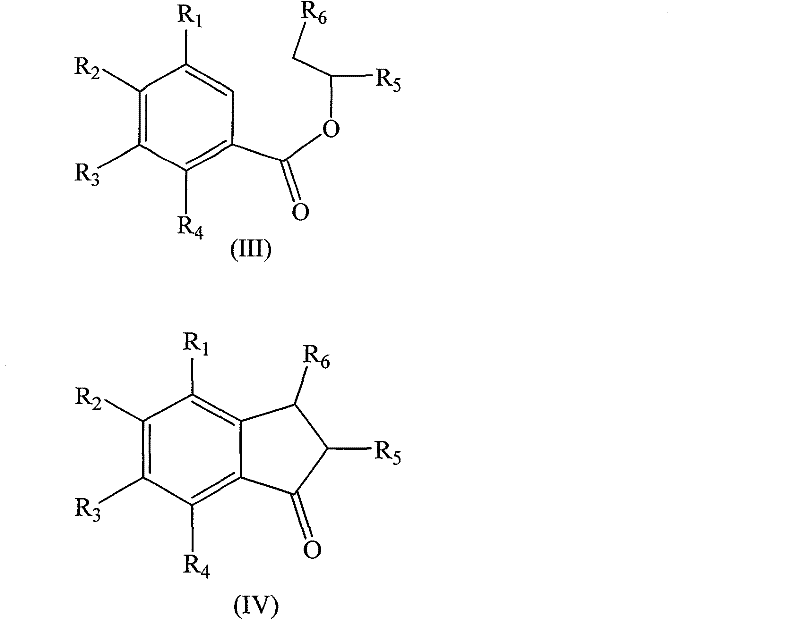 This novel approach utilizes substituted benzoate compounds, which are commercially available and cost-effective, as the primary feedstock. The core innovation lies in the one-pot cyclodehydration step where a Lewis acid, such as aluminum chloride, is combined with a strong acid like polyphosphoric acid or trifluoromethanesulfonic acid. This combination facilitates the direct intramolecular cyclization of the benzoate ester to form the indanone skeleton without the need for prior activation of the aromatic ring via hazardous acyl chlorides. This simplification not only reduces the number of unit operations but also minimizes the generation of chemical waste, aligning with modern green chemistry principles. The subsequent conversion of the indanone to the final indene product via reduction and dehydration is highly efficient, utilizing standard reagents like sodium borohydride and acid-catalyzed azeotropic distillation. This route significantly lowers the barrier to entry for manufacturing these high-value intermediates, offering a compelling value proposition for supply chain managers seeking to optimize production costs and reliability.
This novel approach utilizes substituted benzoate compounds, which are commercially available and cost-effective, as the primary feedstock. The core innovation lies in the one-pot cyclodehydration step where a Lewis acid, such as aluminum chloride, is combined with a strong acid like polyphosphoric acid or trifluoromethanesulfonic acid. This combination facilitates the direct intramolecular cyclization of the benzoate ester to form the indanone skeleton without the need for prior activation of the aromatic ring via hazardous acyl chlorides. This simplification not only reduces the number of unit operations but also minimizes the generation of chemical waste, aligning with modern green chemistry principles. The subsequent conversion of the indanone to the final indene product via reduction and dehydration is highly efficient, utilizing standard reagents like sodium borohydride and acid-catalyzed azeotropic distillation. This route significantly lowers the barrier to entry for manufacturing these high-value intermediates, offering a compelling value proposition for supply chain managers seeking to optimize production costs and reliability.
Mechanistic Insights into Lewis Acid Catalyzed Cyclodehydration
The chemical elegance of this process is rooted in the synergistic interaction between the Lewis acid and the protic acid during the cyclization phase. The Lewis acid, typically aluminum chloride or zinc chloride, coordinates with the carbonyl oxygen of the benzoate ester, increasing the electrophilicity of the carbonyl carbon. Simultaneously, the strong inorganic or organic acid, such as polyphosphoric acid, acts as a dehydrating agent and a proton source, facilitating the cleavage of the ester bond and the subsequent electrophilic aromatic substitution on the ortho-position of the benzene ring. This dual-activation mechanism ensures that the cyclization proceeds under relatively mild thermal conditions, typically ranging from 50°C to 130°C, which helps to preserve the integrity of sensitive functional groups on the aromatic ring. The reaction mixture is carefully quenched with ice water and extracted using non-polar solvents like n-heptane, allowing for the efficient isolation of the indanone intermediate. This mechanistic pathway avoids the formation of poly-acylated byproducts often seen in traditional Friedel-Crafts reactions, thereby enhancing the selectivity of the process.
Following the formation of the indanone, the reduction step employs hydride reducing agents such as sodium borohydride or lithium aluminum hydride in polar solvents like tetrahydrofuran or methanol. This transformation selectively reduces the ketone moiety to a secondary alcohol (indanol) without affecting other potential substituents on the ring system. The final dehydration step is equally critical, utilizing acid catalysts like p-toluenesulfonic acid in refluxing toluene to remove water azeotropically. This drives the equilibrium towards the formation of the double bond, yielding the target indene compound. The ability to control the ratio of isomeric indene products (such as 1-substituted vs. 2-substituted indenes) through the choice of starting benzoate and reaction conditions provides R&D teams with the flexibility to tailor the ligand structure for specific metallocene catalyst performance requirements. This level of control over the impurity profile and isomer distribution is paramount for ensuring the consistent activity and stereoselectivity of the final polymerization catalysts.
How to Synthesize Indene Compounds Efficiently
The implementation of this synthesis route in a pilot or production setting requires strict adherence to the optimized reaction parameters defined in the patent data. The process begins with the preparation of the acid catalyst mixture, followed by the controlled addition of the substituted benzoate substrate under an inert atmosphere to prevent moisture interference. Detailed standard operating procedures regarding temperature ramps, quenching protocols, and work-up sequences are essential to maximize yield and safety.
- Cyclodehydration of substituted benzoates using Lewis acid and inorganic/organic acid mixtures to form indanone intermediates.
- Reduction of the resulting indanone compounds using hydride reducing agents to generate indanol derivatives.
- Acid-catalyzed dehydration of indanols in azeotropic solvents to yield the final indene compounds.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic methodology translates into tangible strategic advantages that extend beyond simple chemical yield. The shift from complex, multi-step traditional routes to this streamlined benzoate-based process fundamentally alters the cost structure of indene compound manufacturing. By eliminating the need for expensive and hazardous acyl chlorides and reducing the number of isolation steps, the overall consumption of raw materials and solvents is drastically reduced. This simplification of the bill of materials allows for significant cost savings, which can be passed down through the supply chain to benefit polymer producers. Furthermore, the use of commercially available benzoate starting materials ensures a stable and reliable supply source, mitigating the risks associated with sourcing specialized or custom-synthesized precursors that often suffer from long lead times and availability issues.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and hazardous acylating agents removes the need for expensive metal scavenging steps and specialized waste treatment protocols. This qualitative reduction in process complexity directly correlates to lower operational expenditures and reduced capital investment in safety infrastructure. The streamlined workflow also reduces labor hours and energy consumption per kilogram of product, contributing to a more competitive pricing model for high-purity indene derivatives.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like substituted benzoates rather than custom intermediates significantly de-risks the supply chain. These starting materials are produced by multiple global suppliers, ensuring continuity of supply even in the face of market fluctuations or logistical disruptions. The robustness of the reaction conditions also means that production schedules are less prone to delays caused by sensitive reaction failures, ensuring consistent on-time delivery for downstream catalyst manufacturers.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing standard reactor types and separation techniques that are common in fine chemical plants. The reduction in hazardous waste generation, particularly the avoidance of chlorinated byproducts and heavy metal residues, simplifies environmental compliance and waste disposal. This aligns with increasingly stringent global environmental regulations, future-proofing the manufacturing process against regulatory changes and enhancing the sustainability profile of the final polymer products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this indene synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and chemical capabilities of this route for potential partners and technical stakeholders.
Q: What are the primary advantages of using substituted benzoates over traditional aromatic hydrocarbons for indene synthesis?
A: Using substituted benzoates eliminates the need for hazardous acyl chlorides and complex multi-step Friedel-Crafts acylation, significantly simplifying the workflow and reducing raw material costs.
Q: How does this method improve the purity profile for metallocene catalyst production?
A: The direct cyclization route minimizes side reactions and byproduct formation common in traditional coupling methods, resulting in a cleaner impurity profile essential for high-performance catalyst ligands.
Q: Is this synthesis route scalable for industrial polymer additive manufacturing?
A: Yes, the process utilizes commercially available reagents and standard unit operations like extraction and distillation, making it highly suitable for large-scale commercial production without specialized equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indene Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality indene compounds play in the development of next-generation metallocene catalysts and advanced polymer materials. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative chemistry described in CN101318887B can be seamlessly transferred from the laboratory to industrial manufacturing. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs which utilize advanced analytical techniques to verify impurity profiles and isomer ratios. Our capability to handle complex synthetic routes allows us to offer customized solutions that meet the specific performance requirements of your catalyst systems.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains for polymer additives. By leveraging our expertise in this efficient synthesis route, we can provide a Customized Cost-Saving Analysis tailored to your specific volume needs. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your target indene derivatives. Together, we can drive innovation in the polyolefin sector through reliable, cost-effective, and high-performance chemical solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
