Scalable Manufacturing Of Spiro Diazepine Intermediates Via Optimized Catalytic Hydrogenation
The pharmaceutical industry constantly seeks robust synthetic routes for complex heterocyclic scaffolds that serve as privileged structures in drug discovery. Patent CN101875658B introduces a significant methodological advancement in the preparation of 3-carbonyl-2,8-diaza spiro[4.5]decane-8-carboxylic acid tert-butyl ester, a valuable template molecule for generating diverse compound libraries. This specific spirocyclic framework is increasingly recognized for its potential in modulating biological targets, yet its synthesis has historically been plagued by purification bottlenecks. The disclosed technology addresses these challenges by replacing labor-intensive chromatographic separations with efficient crystallization and filtration protocols. By leveraging a cascade of Michael addition, catalytic hydrogenation, and thermal decarboxylation, the process achieves a streamlined workflow that enhances both chemical purity and operational throughput. For R&D teams evaluating scaffold accessibility, this patent offers a compelling alternative to legacy methods, promising a more sustainable and economically viable pathway to high-value spiro intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of spiro-fused diazepine derivatives has relied on methodologies that, while chemically valid, present severe obstacles for industrial application. The seminal work by Paul W. Smith, often cited as a reference standard, utilizes a two-step sequence involving 1,1,3,3-tetramethylguanidine (TMG) as a catalyst. A critical drawback of this conventional approach is the absolute dependence on column chromatography for product isolation at every stage. In a commercial setting, column chromatography is a major bottleneck; it consumes vast quantities of silica gel and organic solvents, generates substantial hazardous waste, and limits batch sizes due to column loading capacity. Furthermore, the reported yield for the initial step in the legacy route is merely 35%, indicating significant material loss and inefficient atom economy. These factors collectively inflate the cost of goods sold (COGS) and extend the lead time for producing research quantities, making the traditional route unsuitable for the rapid iteration required in modern medicinal chemistry campaigns.
The Novel Approach
In stark contrast, the methodology outlined in CN101875658B fundamentally reengineers the purification strategy to eliminate chromatography entirely. The new route initiates with a Michael addition that allows the product to be isolated simply by filtration and ethanol beating, bypassing the need for silica-based separation. Subsequent steps utilize catalytic hydrogenation with Raney Nickel, a robust and inexpensive heterogeneous catalyst, to effect both nitro reduction and lactamization in a single pot. The final decarboxylation step employs high-boiling polar solvents like DMSO, facilitating thermal elimination without complex workups. This strategic shift from chromatographic purification to crystallization-driven isolation represents a paradigm shift in process chemistry. It not only improves the overall mass balance but also aligns the synthesis with Green Chemistry principles by reducing solvent intensity and waste generation. The result is a process that is inherently more scalable and cost-effective, directly addressing the pain points of supply chain managers and procurement specialists.
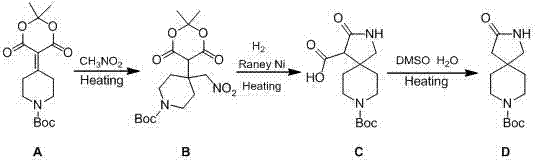
Mechanistic Insights into Raney Nickel Catalyzed Lactamization
The core innovation of this synthetic pathway lies in the second step, where a nitro-methyl piperidine derivative is converted into the spiro-lactam structure. This transformation is driven by Raney Nickel under hydrogen pressure, functioning through a dual-mechanism pathway. Initially, the catalyst facilitates the reduction of the nitro group (-NO2) to a primary amine (-NH2). In the presence of the adjacent ester functionality within the dioxane ring system, the newly formed amine acts as a nucleophile. It attacks the carbonyl carbon, triggering a cyclization event that forms the seven-membered diazepine ring while simultaneously cleaving the dioxane protecting group. This tandem reduction-lactamization sequence is highly efficient because it avoids isolating the unstable amino-intermediate, which could otherwise undergo side reactions or polymerization. The use of Raney Nickel is particularly advantageous here due to its high surface area and activity, allowing the reaction to proceed at moderate temperatures (15°C to 78°C) and pressures (20-60 psi), ensuring safety and energy efficiency during scale-up.
Impurity control is another critical aspect managed by the specific reaction conditions described in the patent. The Michael addition step utilizes sodium methoxide in dioxane, creating a basic environment that favors the formation of the nitro-Michael adduct while minimizing self-condensation of the starting ketone. The subsequent filtration and ethanol beating effectively remove unreacted starting materials and soluble byproducts before they can interfere with the hydrogenation step. In the final decarboxylation phase, the addition of water and sodium chloride in DMSO plays a crucial role in stabilizing the transition state and facilitating the removal of the carboxyl group as carbon dioxide. This careful modulation of ionic strength and solvent polarity ensures that the final spiro product precipitates with high purity (reported up to 99%) upon extraction with toluene, eliminating the need for further recrystallization or chromatographic polishing.
How to Synthesize 3-carbonyl-2,8-diaza spiro[4.5]decane-8-carboxylic acid tert-butyl ester Efficiently
The synthesis of this complex spiro intermediate is achieved through a logical three-step sequence that prioritizes operational simplicity and high purity. The process begins with the activation of nitromethane followed by conjugate addition, proceeds through a catalytic hydrogenation cascade, and concludes with a thermal decarboxylation. Each step has been optimized to allow for direct telescoping or simple filtration workups, avoiding the complexity of traditional multi-step syntheses. For detailed laboratory protocols and specific stoichiometric ratios, please refer to the standardized guide below.
- Perform Michael addition between tert-butyl-4-(2,2-dimethyl-4,6-dicarbonyl-1,3-dioxan-5-ylene)piperidine-1-carboxylate and nitromethane using sodium methoxide in dioxane at 30-65°C.
- Execute catalytic hydrogenation and lactamization using Raney Nickel catalyst under 20-60 psi hydrogen pressure in alcohol solvent to form the spiro lactam intermediate.
- Conduct thermal decarboxylation in DMSO with water and sodium chloride additives at 120-172°C to yield the final spiro diazepine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers tangible strategic benefits beyond mere chemical yield. The most significant advantage is the drastic simplification of the manufacturing process, which directly translates to lower operational expenditures. By removing the requirement for column chromatography, the facility saves on the recurring costs of silica gel, high-grade elution solvents, and the labor hours associated with packing and running columns. This reduction in unit operations also decreases the turnaround time between batches, allowing for faster fulfillment of internal R&D requests or external customer orders. Furthermore, the reliance on commodity chemicals such as nitromethane, Raney Nickel, ethanol, and toluene ensures that raw material sourcing is stable and不受 geopolitical supply shocks that often affect specialized reagents.
- Cost Reduction in Manufacturing: The elimination of chromatographic purification is the primary driver for cost savings in this process. Chromatography is notoriously expensive due to the high volume of solvents required and the low throughput of the equipment. By switching to filtration and crystallization, the process significantly reduces solvent consumption and waste disposal costs. Additionally, the use of Raney Nickel, a non-precious metal catalyst, avoids the high capital tie-up associated with precious metal catalysts like palladium or platinum, and removes the need for expensive metal scavenging steps to meet residual metal specifications in the final API.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain continuity. The reagents used are bulk industrial chemicals with multiple global suppliers, mitigating the risk of single-source dependency. The process conditions, such as moderate hydrogen pressure and temperatures below 200°C, are compatible with standard glass-lined or stainless steel reactors found in most multipurpose pharmaceutical plants. This compatibility means that the process can be easily transferred to contract manufacturing organizations (CMOs) without requiring specialized equipment investments, ensuring a flexible and resilient supply network.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this route is superior. The reduction in solvent usage directly lowers the facility's environmental footprint, aiding in compliance with increasingly strict environmental regulations regarding volatile organic compound (VOC) emissions. The solid waste generated is primarily spent catalyst and filter cakes, which are easier to manage and dispose of compared to the liquid waste streams from chromatography. The high purity achieved through crystallization (99%) ensures that the material meets stringent quality standards for downstream applications without additional refining, streamlining the path from kilogram-scale development to commercial tonnage production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a reliable basis for feasibility assessments.
Q: How does this new synthesis route improve purification compared to traditional methods?
A: Unlike the conventional Smith method which requires tedious column chromatography for purification, this novel route utilizes filtration and recrystallization techniques. This eliminates the need for large volumes of silica gel and eluents, drastically simplifying the downstream processing and reducing waste generation.
Q: What are the critical reaction conditions for the hydrogenation step?
A: The hydrogenation and simultaneous lactamization step utilizes Raney Nickel as a heterogeneous catalyst. Critical parameters include maintaining hydrogen pressure between 20-60 psi and controlling the temperature between 15°C and 78°C in an alcoholic solvent to ensure high conversion to the spiro lactam intermediate without over-reduction.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is highly amenable to scale-up. The avoidance of chromatography, the use of robust heterogeneous catalysis (Raney Ni), and the reliance on common industrial solvents like ethanol, methanol, and toluene make it ideal for multi-kilogram to ton-scale manufacturing in standard pharmaceutical reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-carbonyl-2,8-diaza spiro[4.5]decane-8-carboxylic acid tert-butyl ester Supplier
At NINGBO INNO PHARMCHEM, we understand that accessing complex spirocyclic intermediates is critical for advancing your drug discovery programs. Our technical team has extensively analyzed the route described in CN101875658B and possesses the expertise to execute this chemistry with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our facilities are equipped with state-of-the-art hydrogenation reactors and rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 3-carbonyl-2,8-diaza spiro[4.5]decane-8-carboxylic acid tert-butyl ester meets the highest industry standards.
We invite you to collaborate with us to optimize your supply chain for this key intermediate. By leveraging our process optimization capabilities, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you accelerate your project timelines with a reliable, high-quality supply of this essential pharmaceutical building block.
