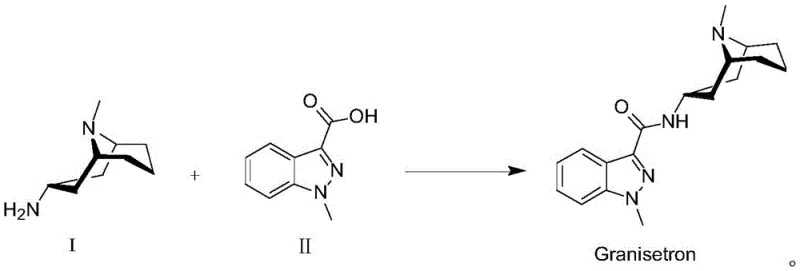
Advanced Manufacturing of Granisetron Intermediate 3α-Homotropylamine via Stereoselective Reduction
Advanced Manufacturing of Granisetron Intermediate 3α-Homotropylamine via Stereoselective Reduction
The pharmaceutical industry continuously seeks robust, scalable, and cost-effective pathways for synthesizing critical antiemetic agents like Granisetron. A pivotal breakthrough in this domain is detailed in patent CN110804051A, which discloses a superior preparation method for the key intermediate, 3α-homotropylamine. This innovation addresses long-standing challenges in stereoselectivity and process safety that have plagued previous manufacturing routes. By leveraging a unique combination of Red-Al reduction mediated by Lewis acids, the technology enables the production of high-purity intermediates under mild conditions, effectively bypassing the need for expensive noble metal catalysts and hazardous high-pressure hydrogenation. This report analyzes the technical merits and commercial implications of this advanced synthesis strategy for global supply chain stakeholders.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3α-homotropylamine has been fraught with significant technical and economic hurdles that impede efficient large-scale production. Prior art, such as the methods described in U.S. Pat. No. 4,470,585A, relies heavily on the use of expensive Platinum catalysts and highly reactive, dangerous reagents like Lithium Aluminum Hydride (LiAlH4). These processes not only demand specialized high-pressure equipment, increasing capital expenditure, but also introduce severe safety risks associated with handling pyrophoric materials and high-pressure hydrogen gas. Furthermore, alternative routes utilizing Raney Nickel for catalytic hydrogenation, as seen in CN1451660A, suffer from poor stereoselectivity. These conventional hydrogenation methods typically yield a complex mixture of 3α and 3β isomers, where the desired 3α-isomer constitutes only 60-70% of the product. The presence of substantial isomeric impurities complicates downstream purification, leading to significant yield losses and increased waste generation, thereby inflating the overall cost of goods sold for the final API.
The Novel Approach
In stark contrast to these legacy methods, the novel approach outlined in the patent introduces a streamlined and highly selective synthetic pathway that fundamentally alters the production landscape. The process initiates with a classic Mannich reaction involving acetone dicarboxylic acid to construct the tropane skeleton, followed by oxime formation. 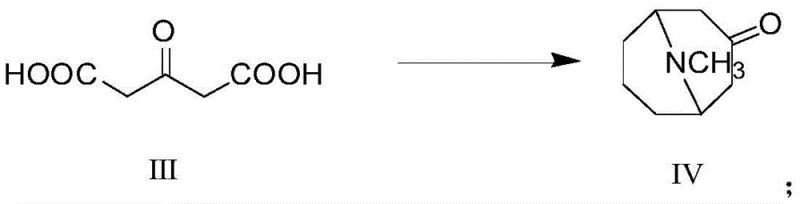
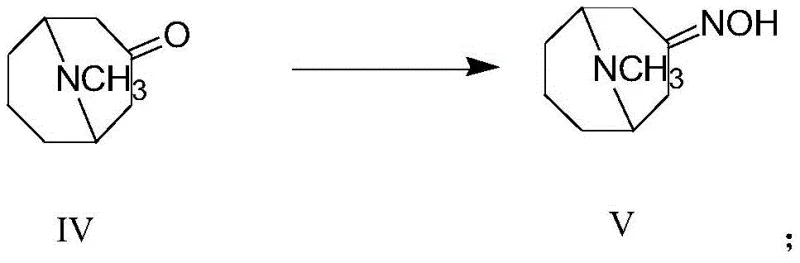 The core innovation lies in the subsequent reduction step, where 3-homotropinone oxime is treated with Red-Al (Sodium Bis(2-methoxyethoxy)aluminum Hydride) in the presence of a Lewis acid. This specific reagent combination facilitates a highly stereoselective reduction, yielding a crude product enriched with approximately 90% of the desired 3α-isomer. This dramatic improvement in selectivity means that the crude material can often be used directly in the final coupling step to produce Granisetron, or subjected to a mild purification protocol involving Boc-protection to achieve purity levels exceeding 99%. This eliminates the reliance on high-pressure infrastructure and precious metals, marking a significant leap forward in process efficiency.
The core innovation lies in the subsequent reduction step, where 3-homotropinone oxime is treated with Red-Al (Sodium Bis(2-methoxyethoxy)aluminum Hydride) in the presence of a Lewis acid. This specific reagent combination facilitates a highly stereoselective reduction, yielding a crude product enriched with approximately 90% of the desired 3α-isomer. This dramatic improvement in selectivity means that the crude material can often be used directly in the final coupling step to produce Granisetron, or subjected to a mild purification protocol involving Boc-protection to achieve purity levels exceeding 99%. This eliminates the reliance on high-pressure infrastructure and precious metals, marking a significant leap forward in process efficiency.
Mechanistic Insights into Lewis Acid-Mediated Stereoselective Reduction
The success of this synthetic route hinges on the precise mechanistic interaction between the reducing agent and the Lewis acid promoter during the conversion of the oxime to the amine. When Red-Al is combined with Lewis acids such as tetraisopropyl titanate, titanium tetrachloride, or concentrated sulfuric acid, it generates a modified hydride species with distinct steric and electronic properties. This activated complex coordinates with the nitrogen atom of the oxime intermediate, directing the hydride attack specifically from the less hindered face of the bicyclic tropane ring system. This directed delivery is crucial for establishing the correct stereochemistry at the C3 position, favoring the formation of the 3α-amine over the 3β-epimer. The reaction is typically conducted in tetrahydrofuran (THF) at controlled temperatures ranging from -10°C to 20°C, ensuring that the kinetic control of the reduction is maintained throughout the process. 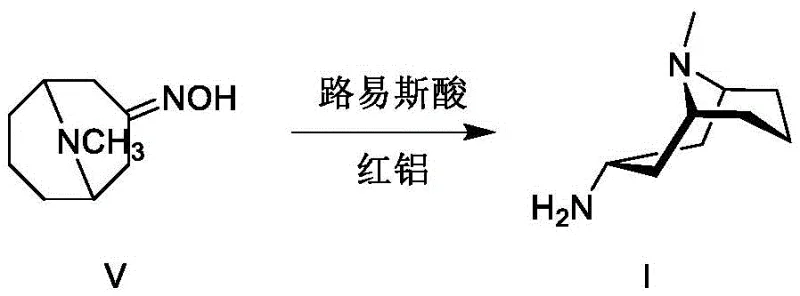 By carefully modulating the molar ratios of Red-Al to Lewis acid (typically 2:1 to 5:1), the reaction environment is optimized to maximize the yield of the target isomer while minimizing side reactions.
By carefully modulating the molar ratios of Red-Al to Lewis acid (typically 2:1 to 5:1), the reaction environment is optimized to maximize the yield of the target isomer while minimizing side reactions.
Furthermore, the mechanism includes a robust strategy for impurity management, which is critical for pharmaceutical grade intermediates. Even in scenarios where the alternative Raney Nickel reduction is employed, the patent discloses a sophisticated purification mechanism to rescue the stereochemical purity. This involves a selective protection-deprotection sequence using di-tert-butyl dicarbonate (Boc2O) in the presence of copper sulfate and sodium bicarbonate. The specific interaction between the copper ions and the amine isomers likely facilitates a differential solubility or reactivity that allows for the effective separation of the 3α-isomer from the 3β-impurity. 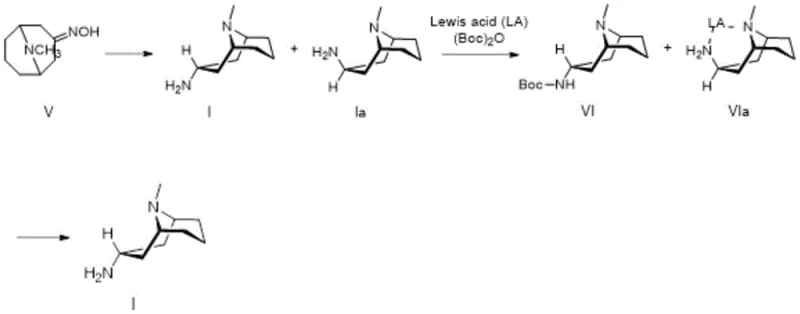 Following the formation of the Boc-protected intermediate, acidic hydrolysis regenerates the free amine with exceptional purity (>99%). This dual-pathway capability—either direct use of the highly selective Red-Al product or rigorous purification of the Raney Nickel product—provides manufacturers with flexible options to balance cost and purity requirements based on their specific quality control protocols.
Following the formation of the Boc-protected intermediate, acidic hydrolysis regenerates the free amine with exceptional purity (>99%). This dual-pathway capability—either direct use of the highly selective Red-Al product or rigorous purification of the Raney Nickel product—provides manufacturers with flexible options to balance cost and purity requirements based on their specific quality control protocols.
How to Synthesize 3α-Homotropylamine Efficiently
The synthesis of 3α-homotropylamine via this patented methodology offers a practical and scalable solution for industrial chemists aiming to optimize their production lines. The process is designed to be operationally simple, utilizing readily available starting materials and standard laboratory equipment, which significantly lowers the barrier to entry for contract manufacturing organizations. The initial steps involve the construction of the pseudopelletierine core followed by oximation, both of which are high-yielding transformations that set the stage for the critical reduction step. Operators must pay close attention to temperature control during the addition of the Lewis acid and the subsequent reduction to ensure optimal stereoselectivity. The detailed standardized synthesis steps, including specific reagent quantities, reaction times, and workup procedures, are provided in the technical guide below to facilitate immediate technology transfer and pilot plant trials.
- Perform a Mannich reaction on acetone dicarboxylic acid with glutaraldehyde and methylamine to obtain pseudopelletierine (Formula IV).
- React pseudopelletierine with hydroxylamine to synthesize 3-homotropinone oxime (Formula V).
- Subject the oxime to catalytic reduction using Red-Al and a Lewis acid (e.g., tetraisopropyl titanate) at controlled temperatures to yield 3α-homotropylamine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route presents a compelling value proposition centered on risk mitigation and cost optimization. The primary economic driver is the complete elimination of expensive noble metal catalysts like Platinum, which are subject to volatile market pricing and supply constraints. By substituting these with abundant and cost-effective Lewis acids and Red-Al, the raw material cost structure is significantly improved. Additionally, the removal of high-pressure hydrogenation steps reduces the dependency on specialized, high-CAPEX reactor vessels, allowing production to be scaled in standard glass-lined or stainless steel reactors. This flexibility enhances supply chain resilience, as the process can be easily transferred between different manufacturing sites without requiring extensive infrastructure modifications, thereby ensuring consistent supply continuity for downstream API production.
- Cost Reduction in Manufacturing: The transition away from hazardous reagents like LiAlH4 and expensive Platinum catalysts results in substantial direct cost savings. The milder reaction conditions also translate to lower energy consumption for heating and cooling, as the process does not require extreme temperatures or pressures. Furthermore, the high stereoselectivity of the Red-Al method minimizes the formation of isomeric impurities, which reduces the burden on purification units and decreases solvent usage for chromatography or recrystallization. This holistic reduction in material and utility consumption drives down the overall cost of goods, making the final Granisetron API more competitive in the generic pharmaceutical market.
- Enhanced Supply Chain Reliability: Reliance on a single source for critical catalysts or high-pressure processing capabilities creates a bottleneck in the supply chain. This new method utilizes commodity chemicals that are widely available from multiple global suppliers, diversifying the supply base and reducing the risk of disruption. The robustness of the chemistry ensures high batch-to-batch consistency, which is essential for maintaining regulatory compliance and avoiding costly production delays due to out-of-specification results. The ability to produce high-purity intermediates reliably strengthens the entire value chain, from raw material sourcing to final drug product delivery.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this process offers significant advantages that align with modern green chemistry principles. The avoidance of heavy metal catalysts simplifies waste treatment protocols and reduces the environmental footprint associated with heavy metal disposal. The mild operating conditions enhance workplace safety by eliminating the risks associated with high-pressure hydrogen gas and pyrophoric reagents. These factors facilitate easier regulatory approval for new manufacturing sites and support the long-term sustainability goals of pharmaceutical companies, ensuring that the production of this critical antiemetic intermediate remains viable and compliant with increasingly stringent global environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing a clear understanding of the process capabilities and limitations. Stakeholders are encouraged to review these details to assess the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does this new method improve stereoselectivity compared to traditional Raney Nickel hydrogenation?
A: Traditional Raney Nickel hydrogenation often yields a difficult-to-separate mixture of 3α and 3β isomers with low selectivity. The patented Red-Al and Lewis acid method achieves approximately 90% 3α-isomer content directly, significantly reducing downstream purification burdens.
Q: What are the safety advantages of avoiding Platinum catalysts and LiAlH4?
A: Conventional methods utilizing Platinum catalysts and Lithium Aluminum Hydride (LiAlH4) require high-pressure equipment and pose significant safety risks due to pyrophoric reagents. This novel route operates under mild conditions with safer reagents, lowering operational hazards and equipment costs.
Q: Can the crude product be used directly for Granisetron synthesis?
A: Yes, the crude 3α-homotropylamine obtained via the Red-Al reduction has sufficient purity (85-90%) to be used directly in subsequent coupling reactions, although an optional Boc-protection purification step is available to achieve >99% purity if required.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3α-Homotropylamine Supplier
The technological advancements detailed in patent CN110804051A represent a significant opportunity for optimizing the supply chain of Granisetron intermediates. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to leverage these innovative synthetic routes for our global partners. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 3α-homotropylamine meets the highest pharmaceutical standards required for API synthesis.
We invite procurement leaders and R&D directors to collaborate with us to evaluate the potential for cost savings and process improvements in your current supply chain. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced manufacturing capabilities can enhance your operational efficiency and reduce time-to-market for your final pharmaceutical products.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
