Revolutionizing 2-Ethylanthraquinone Production via Continuous Liquid-Phase Cyclization Technology
Revolutionizing 2-Ethylanthraquinone Production via Continuous Liquid-Phase Cyclization Technology
The global demand for high-purity 2-ethylanthraquinone, a critical precursor in hydrogen peroxide synthesis and specialty dye manufacturing, necessitates robust and scalable production methodologies. Patent CN103360229A introduces a transformative continuous process that addresses the inherent inefficiencies of traditional batch synthesis. This technology leverages the direct utilization of liquid 2-(4-alkylbenzoyl)benzoic acid, bypassing energy-intensive solidification and redissolution steps. By integrating precise thermal control with continuous flow reactors, the method achieves superior molecular mixing and reaction kinetics. For R&D directors and procurement specialists seeking a reliable 2-ethylanthraquinone supplier, understanding this technological leap is essential for securing long-term supply chain stability and cost efficiency in fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
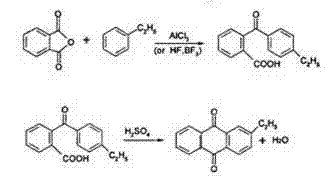
Traditional manufacturing routes for 2-ethylanthraquinone typically rely on a discontinuous batch process where the intermediate, 2-(4-ethylbenzoyl)benzoic acid (BE acid), is isolated as a solid. This solid intermediate requires extensive downstream processing, including drying, granulation, and subsequent dissolution in concentrated sulfuric acid before cyclization can occur. These additional unit operations introduce significant bottlenecks, increasing labor intensity and equipment footprint while creating opportunities for product degradation. Furthermore, the dissolution of solid BE acid in sulfuric acid is often kinetically limited, leading to incomplete mixing at the molecular level. This heterogeneity frequently results in localized hot spots or unreacted pockets, which not only depresses overall yield to the 75-79% range but also complicates impurity profiles, making purification more costly and technically challenging for large-scale operations.
The Novel Approach
In stark contrast, the patented continuous methodology eliminates the solid isolation step entirely by maintaining the BE acid intermediate in a molten liquid state at temperatures between 130-180°C. This liquid stream is immediately introduced into a high-efficiency mixer where it meets concentrated sulfuric acid at a controlled volume ratio of 1:1.5 to 1:2.5. The immediate contact between the liquid reactants ensures rapid, molecular-scale homogenization, which is critical for uniform reaction progression. Following mixing, the stream flows continuously through a series of stirred reactors maintained at 100-140°C, allowing for precise residence time control between 5-30 minutes. This seamless transition from acylation to cyclization drastically simplifies the process flow, reduces capital expenditure on drying equipment, and enhances the overall throughput capacity for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Acid-Catalyzed Dehydration Cyclization
The core chemical transformation in this process is the intramolecular Friedel-Crafts acylation, specifically the dehydration cyclization of 2-(4-alkylbenzoyl)benzoic acid. In the presence of concentrated sulfuric acid, the carboxylic acid group is protonated, facilitating the loss of water to generate a highly reactive acylium ion intermediate. This electrophilic species then attacks the electron-rich aromatic ring at the ortho position relative to the benzoyl group, closing the central quinone ring. The efficiency of this mechanism is heavily dependent on the acidity function of the medium and the availability of the substrate in solution. By feeding the substrate as a liquid, the patent ensures that the concentration of the reactive species remains optimal throughout the reactor train, minimizing the induction period often seen when dissolving solids. This kinetic advantage allows the reaction to proceed rapidly at moderate temperatures, preserving the integrity of the ethyl substituent against oxidative degradation.
Impurity control is another critical aspect governed by the reaction parameters outlined in the patent. A primary side reaction in anthraquinone synthesis is sulfonation, where the sulfuric acid acts as a sulfonating agent rather than just a dehydrating catalyst. This undesired pathway becomes thermodynamically favorable at temperatures exceeding 140°C. The patented process strictly limits the reactor temperature to the 100-140°C window and utilizes a series of reactors to approximate plug-flow behavior, thereby preventing back-mixing and over-exposure of the product to harsh acidic conditions. Additionally, the precise control of the acid-to-substrate ratio prevents the formation of polysulfonated by-products, which are difficult to remove and can poison downstream hydrogenation catalysts. This rigorous control strategy ensures that the final 2-ethylanthraquinone meets stringent purity specifications required for high-performance applications.
How to Synthesize 2-Ethylanthraquinone Efficiently
The implementation of this continuous synthesis route requires careful attention to thermal management and fluid dynamics to maximize yield and safety. The process begins with the maintenance of the BE acid feedstock in a molten state, avoiding the energy penalty of crystallization. Detailed operational parameters regarding flow rates, mixing Reynolds numbers, and specific reactor configurations are critical for replicating the laboratory success on an industrial scale. Operators must ensure that the mixing zone achieves turbulent flow to guarantee homogeneity before the mixture enters the heated reaction zone. The following guide outlines the standardized procedural framework derived from the patent examples, serving as a foundational reference for process engineers aiming to adopt this advanced manufacturing technique.
- Maintain liquid 2-(4-alkylbenzoyl)benzoic acid at 130-180°C and mix with concentrated sulfuric acid at a ratio of 1: 1.5 to 1:2.5 in a high-efficiency mixer.
- Transfer the mixture immediately into a series of stirred reactors maintained between 100-140°C for a residence time of 5-30 minutes to effect ring closure.
- Directly hydrolyze the reaction effluent, followed by solvent extraction with benzene or toluene, washing, and distillation to isolate high-purity 2-ethylanthraquinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this continuous liquid-phase technology offers profound strategic benefits beyond mere technical novelty. The elimination of solid handling steps such as drying and granulation translates directly into reduced utility consumption and lower labor costs, as fewer manual interventions and batch turnovers are required. The continuous nature of the process also implies a smaller physical footprint for the production facility, allowing for higher output density within existing infrastructure. Furthermore, the improved yield profile means that less raw material is wasted per kilogram of finished product, effectively lowering the cost of goods sold (COGS) without compromising quality. These factors collectively enhance the competitiveness of suppliers who utilize this methodology in the global market for fine chemical intermediates.
- Cost Reduction in Manufacturing: The removal of the drying and redissolution stages significantly cuts energy consumption associated with heating and cooling cycles typical of batch processing. By processing the intermediate directly from the upstream acylation unit in its liquid form, the facility avoids the capital and operational expenses of solid-liquid separation equipment. This streamlined workflow reduces the overall cycle time per batch, effectively increasing asset utilization rates. Consequently, manufacturers can offer more competitive pricing structures for high-purity 2-ethylanthraquinone while maintaining healthy margins, providing a distinct economic advantage in cost reduction in agrochemical intermediates manufacturing and related sectors.
- Enhanced Supply Chain Reliability: Continuous processing inherently offers greater predictability and stability compared to batch operations, which are prone to variability between runs. The steady-state operation of the reactor train ensures a consistent output rate, allowing for more accurate forecasting and inventory planning. This reliability is crucial for downstream customers in the hydrogen peroxide industry who require uninterrupted feedstock supplies to maintain their own continuous production lines. By mitigating the risks of batch failures and extended turnaround times, this technology strengthens the resilience of the supply chain, ensuring reducing lead time for high-purity 2-ethylanthraquinone derivatives and fostering stronger long-term partnerships.
- Scalability and Environmental Compliance: The process design facilitates straightforward scalability through the addition of parallel reactor trains or increasing the diameter of continuous units, avoiding the non-linear challenges often faced when scaling batch vessels. Environmentally, the optimized acid ratio minimizes the volume of spent acid waste generated, reducing the burden on wastewater treatment facilities and lowering disposal costs. The closed-system nature of continuous flow chemistry also limits fugitive emissions and operator exposure to hazardous chemicals, aligning with increasingly strict global environmental, health, and safety (EHS) regulations. This commitment to sustainable manufacturing practices positions the supplier as a responsible partner in the value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the continuous synthesis of 2-ethylanthraquinone. These insights are derived directly from the experimental data and process descriptions found in the referenced patent literature. Understanding these nuances helps stakeholders evaluate the feasibility and benefits of integrating this technology into their sourcing strategies. The answers reflect the consensus on best practices for managing reaction conditions and optimizing product quality in an industrial setting.
Q: How does the continuous liquid-phase method improve yield over traditional batch processes?
A: By utilizing liquid feedstock directly without drying or granulation, the process ensures molecular-scale mixing with sulfuric acid, reducing side reactions and increasing yield by 5-10% compared to conventional solid-dissolution methods.
Q: What are the critical temperature controls to prevent sulfonation by-products?
A: The cyclization reaction temperature must be strictly controlled between 100-140°C. Temperatures exceeding this range promote unwanted sulfonation of the anthraquinone ring, significantly lowering product purity and recovery rates.
Q: Does this process reduce environmental waste compared to older technologies?
A: Yes, the optimized acid-to-substrate ratio (1:1.5-1:2.5) and continuous flow design minimize excess acid usage and facilitate easier recycling, resulting in substantially reduced waste acid discharge compared to traditional batch operations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Ethylanthraquinone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced continuous manufacturing processes is key to meeting the evolving demands of the global pharmaceutical and chemical industries. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the liquid-phase cyclization of BE acid are translated into robust, GMP-compliant supply chains. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-ethylanthraquinone meets the exacting standards required for hydrogen peroxide catalysis and specialty applications. Our commitment to technological excellence ensures that our clients receive products of consistent quality, backed by a deep understanding of reaction engineering and process optimization.
We invite procurement leaders and R&D directors to collaborate with us on optimizing their supply chains for critical intermediates. Whether you are looking to secure a stable source of existing materials or develop custom synthetic routes, our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a versatile manufacturing platform capable of adapting to complex chemical challenges, ensuring your projects move forward without interruption.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
